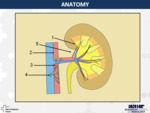
IMAGING PROTOCOLS:
The modality techniques for imaging acquisition and evaluation of the renal sinus can vary from:
US. Basic distinction of cystic from solid space-occupying lesions.
Doppler: Study of renal vascular lesions and vascularization of the kidney.
Multidetector CT. Most sensitive,
efficient and fastest technique.
It provides high temporal and spatial resolution.
Multiplanar reconstructions.
- Non-enhanced CT (NECT): Detection of calcium and blood.
It is used as a baseline in order to check the contrast enhancement.
- Corticomedullary phase (35-40’’): Establishes the relation with vascular structures and the degree of enhancement after the administration of contrast.
- Nephrographic phase (90’’): It has the best sensibility to detect and characterize renal lesions.
- Excretory phase (5-15’): Detection of urothelial abnormalities and its dependence to the collecting system.
MR. It provides detailed tissue characterization of the lesions due to a better soft tissue resolution.
It is also an alternative for patients with renal failure or contrast allergies.
- Anatomical sequences: T2-weighted HASTE,
T1-weighted In-phase/out of phase and T1-weighted fat-suppressed sequences.
- Functional sequences: Diffusion-weighted and dynamic contrast-enhanced images.
Angiography. Evaluation and treatment of renal vascular lesions.
Excretory urography. Evaluation of the renal collecting system involvement.
PATHOLOGIES
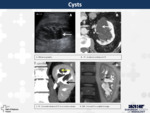
1. Cysts (Fig.
2)
Renal cysts have a high prevalence among population.
Peripelvic cysts are benign extraparenchymal cysts located in the renal sinus thought to have a lymphatic origin.
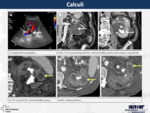
2. Calculi (Fig.
3)
Calculi are normally located in the calyx.
They can have different type of shapes and sizes.
When they move into the pelvis or ureter they can cause obstruction and retrograde dilatation of the excretory system.
Sometimes the obstruction without a correct treatment can be complicated with fornix rupture or infection.
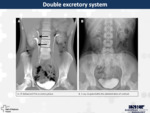
3. Double excretory system (Fig.
4)
Duplicated collecting systems is one of the most common congenital renal tract abnormalities.
It can be defined as incomplete fusion of upper and lower pole moieties of the kidney resulting in two pyelocaliceal systems that are associated with a single or double ureters.
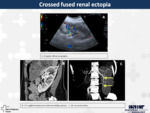
4. Crossed fused renal ectopia (Fig.
5)
It is a congenital disorder,
usually asymptomatic,
in which a kidney is crossed and fused with the contralateral kidney.
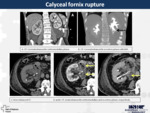
5. Calyceal fornix rupture (Fig.
6)
It is mainly caused secondary to hydronephrosis in cases of urinary tract obstruction.
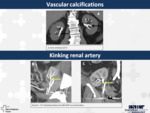
6. Vascular calcifications (Fig.
7)
Vascular calcifications are quite prevalent in the population,
especially as a complication in patients with chronic kidney disease undergoing dialysis.
There has been demonstrated that the extent and histological type of vascular calcification are predictors of vascular mortality.
Some vascular lesions can mymic parapelvic masses or peripelvic lesions.
It is important to demonstrate its vascular nature through the different imaging techniques.
7. Kinking renal artery (Fig.
7)
Kinking of the renal artery in transplants is usually due to malposition of the graft.
It occurs when the vascular anastomoses are inappropriately placed,
and is more common with the right kidney as the right renal vein is much shorter than the artery.
It is a rare cause of early graft dysfunction.
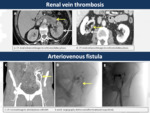
8. Renal vein thrombosis (Fig.
8)
The most common cause of renal vein thrombosis is hypercoagulation states (in adults for example in a nephrotic syndrome).
It can also be secondary to tumours (direct invasion,
coagulation alteration,
or vein compression),
to drugs or systemic diseases.
9. Arteriovenous fistula (Fig.
8)
The main cause of arteriovenous fistulas in the kidney is iatrogenic due to interventional procedures such as biopsies.
Others can be congenital.
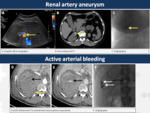
10. Renal artery aneurysm (Fig.
9)
Renal artery aneurysms are uncommon and most of them will be discovered as an incidental finding mainly caused by atherosclerosis.
However,
symptomatic aneurysms can cause hypertension,
pain,
hematuria,
and renal infarction.
They can be dangerous due to a potential risk of rupture and fistulization.
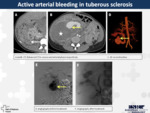
11. Active arterial bleeding (Fig.
9 and 10)
It can be caused due to renovascular or iatrogenic injuries (most commonly secondary to biopsies).
There are less common causes of renal bleeding,
as multiple and voluminous angiomyolipomas,
for example in tuberous sclerosis.
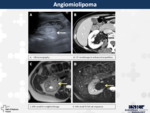
12. Angiomiolipoma (Fig.
11)
It is the most common benign solid renal mass and is composed of angiomatous tissue,
smooth muscle and fat.
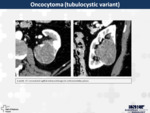
13. Oncocytoma (Fig.
12)
Oncocytoma is the second most common benign solid renal mass.
They are normally ill-defined lesions with uniform enhancement,
and sometimes a central scar (similar to the one of chromophobe RCC and to the central necrosis in clear cell RCC).
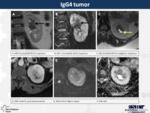
14. IgG4 tumour (Fig.
13)
IgG4-related systemic disease is a chronic inflammatory condition characterized by tissue infiltration with lymphocytes and IgG4 plasma cell,
various degrees of fibrosis and a usually prompt response to oral steroids.
They tend to form tumourous masses that have contrast enhancement.
They usually show hyperintensity in T2 and restricted diffusion.
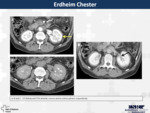
15. Erdheim Chester or Polyostotic sclerosing histiocytosis (Fig.
14)
It is a rare disease characterized by abnormal multiplication of histiocytes (non-Langerhans-cell histiocytosis) and other inflammatory cells and most commonly musculoskeletal involvement.
Extraskeletal involvement can be seen in 30-50% of patients (more commonly in lung,
kidney,
skin and heart-great vessels).
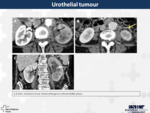
16. Urothelial tumour (Fig.
15)
The majority of the pelvicaliceal cancers are transitional cell carcinomas (90%),
and the rest squamous cell carcinomas.
They are both indistinguishable though imaging.
These tumours have normally a centrifugally growth,
having an origin in the renal pelvis and secondarily invading the renal sinus fat and finally the renal parenchyma (which corresponds to a T3 in the TNM classification).
They can be seen as intraluminal filling defects in excretory urography,
CT or MR.
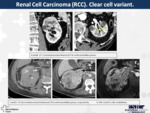
17. Renal Cell Carcinoma (RCC).
Clear cell variant (Fig.
16)
Clear cell is the most common subtype of RCC (70% of all).
It is a hypervascular lesion,
frequently heterogeneous due to necrosis,
hemorrhage,
cystic components or calcifications,
intracellular fat and rarely extracellular fat (differential diagnose with angiomyolipoma).
It has usually a strong enhancement in the corticomedullary phase,
being the nephrogenic phase the most sensitive for the detection,
because the renal parenchyma enhances more intensely than the tumour.
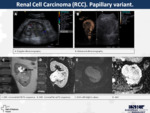
18. Renal Cell Carcinoma (RCC).
Papillary variant (Fig.
17)
Papillary RCC are typically homogeneous lesions mymicing cysts,
but can be heterogenous when they are large (due to necrosis,
hemorrhage or calcifications).
In contrast with clear cell carcinoma,
the lesions are normally hypovascular with a very subtle enhancement.
They are also more frequently bilateral and multifocal.
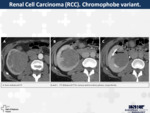
19. Renal Cell Carcinoma (RCC).
Chromophobe variant (Fig.
18)
Chromophobe type RCC it is usually a solid well-defined lesion that can have a central scar or wheel pattern of contrast enhancement (difficult to differentiate from oncocytomas on imaging).
The enhancement is homogeneous and less intense than in clear cell RCC.
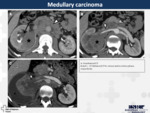
20. Medullary carcinoma (Fig.
19)
A medullary carcinoma has to be suspected when an infiltrative and aggressive renal mass is seen in a young patient with sickle cell disease.
The case showed in Fig.
19 corresponds to a patient that surprisingly,
did not have the history of sickle cell disease.
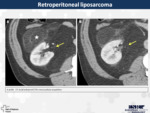
21. Retroperitoneal liposarcoma (Fig.
20)
It is a malignant tumour of mesenchymal origin and the most common primary retroperitoneal neoplasm.
They are more frequently well-differentiated and less commonly myxoid,
pleomorphic,
round cell or dedifferentiated subtypes.
At the CT it is usually an heterogeneous mass with a varying amount of fat and soft tissue,
multiple septae and different levels of enhancement of the components.
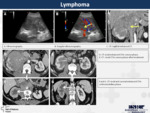
22. Lymphoma (Fig.
21)
Normally it is seen as a retroperitoneal mass with contiguous extension into the renal sinus and to the perinephric space.
These tumours characteristically make a vessel and collecting system encasement without invasion, without secondary thrombosis or hydronephrosis.
They are typically homogeneous masses with less enhancement compared to the normal parenchyma.
At MR they tend to be hypointense on T1-weighted sequences and heterogeneously hypo or isointense on T2-weighted sequences.