Motion phantom model and CCTA scanning protocol
We used a cardiac motion phantom (20) coupled to a three-dimensional (3D) motion simulator (QRM-Sim4D-Cardio; Quality Assurance in Radiology and Medicine,
Moehrendorf,
Germany).
The motion simulator permits a range of motion of 80×40×80 mm (x-,
y- and z-axis) and a maximum frequency of 3 Hz,
thereby creating motion sequences simulating 3D heart motion with corresponding simulated ECG,
which can be fed out to the CT machine’s ECG control panel (figure 1).
This allows for an ECG-synchronised CT image resembling physiological coronary artery movement.
Attached to the 3D motion simulator,
there is the motion arm with a probe holder fitted for simulated stenotic vessels.
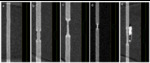
Five coronary arteries phantom models (ranging from 1.18 to 6.4mm diameter and from 42 to 71% stenosis) were filled with iodine,
diluted with deionized water at a concentration of 25:1,
resulting in target attenuation of approximately 350 Hounsfield units (figure 2).
We have found this dilution with a local cohort of spectral CCTA,
which give us the concentration of iodine in coronary during CCTA.
The vessel was placed in a water tank.
The anthropomorphic thorax phantom consists of artificial lungs,
spine,
and soft tissue equivalent material who simulated the tissues in the thorax in terms of density and attenuation.
At the position of the heart is a cylindrical hole in which the water tank can be placed.
We positioned all the phantom on the multidetector computed tomography table to move into the scan field of view during image acquisition (figure 3).
The phantom model was scanned by a multidetector row CT scanner (GE Healthcare,
Optima CT660,
Milwaukee,
WI,
USA).
Scanner parameters included 256×0.625 collimation,
and detector coverage was 40 mm,
with a reconstruction slice thickness of 0.6 mm.
Gantry rotation time was 350 ms per rotation and the maximum tube current was 450 mA with a tube voltage of 120 kVp.
The initial scan was conducted without motion for reference,
followed by motion that simulated HR values of 50,
60,
70,
80,
90,
100,
110 BPM and one arrhytmic with HR variability about 15 BPM.
For each HR values,
we used three motion profiles : left anterior descending,
circumflex and right coronary artery (figure 4).
Image reconstruction and analysis
All images were reconstructed using conventional algorithm (CA) and MCA techniques (SnapShot Freeze®; GE Healthcare).
We used a retrospective ECG gating with CA and MCA reconstructions images generated at 5% intervals at approximately 45%,
60% and 75% of the R-R interval.
This led to the inclusion of 720 vessel reconstructions in each group.
The principles of the MCA we applied have been published previously (12,16,21).
MCA requires image data sampling acquisition at one particular time point within the cardiac cycle that corresponds to the target cardiac phase at ECG-triggered CCTA,
and at adjacent time points 80 ms prior to and after that time point,
which is referred to as “padding”.
Based on this tri-phasic data set,
the MCA first performs automatic coronary artery tree segmentation and subsequently analyzes direction and velocity of motion of each coronary artery using a fast and accurate,
three dimensional,
nonrigid,
bidirectional,
labeled point matching approach.
The MCA then calculates a motion-corrected data set at the target cardiac phase to restore the vessel lumen.
This requires few minutes off-line computation time.
This approach works on per-vessel and per-segment bases to correct for differing degrees of motion for each voxel of the coronary vessel adaptively compressing the temporal window within those localized regions where most needed.
When the MCA failed,
we corrected the vessel tracking manually to allow the success of the MCA.
Image quality (qualitative score,
signal-to-noise and contrast-to-noise ratios,
phase quality,
interpretability,
artefacts) as well as diagnostic accuracy (% of error of: coronary diameters,
area and level of stenosis measurements,
compared to static reference acquisition),
were evaluated for both MCA and conventional algorithm reconstructions.
Qualitative score (12 points) included diagnostic confidence,
outline quality,
homogeneity and artifacts.
The type of artifacts were noted: stair-step artifact,
motion artifact and blooming artifact.
The signal-to-noise ratio (SNR) was measured by placing a circular region of interest within the contrast-enhanced lumen of the tube for determining the mean attenuation by the standard deviation.
The contrast-to-noise ratio (CNR) was calculated by dividing the contrast between tube lumen and surrounding water by the image noise.
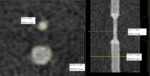
Area,
minimal and average diameters were measured with an electronic caliper (figure 5).
Statistical analysis
All comparisons were performed using standard paired T-Test and McNemar test for qualitative values.
P < 0.05 indicated a significant difference.