Pancreatic ductal adenocarcinoma (PDAC) represents the fourth leading cause of cancer-related mortality,
associated with an extremely poor outcome and 5% of five-year survival rate due to its aggressiveness.
Early diagnosis is crucial for patient's clinical history,
leading to surgical treatment in case of resectable PDAC or neoadjuvant chemotherapy in the case of locally advanced cancer.
If early surgical and chemotherapy treatment can determine survival improvement,
on the other hand,
misdiagnosis of benign or non-neoplastic entities that mimic PDAC may lead to diagnostic error and use of unnecessary invasive procedures.
PANCREATIC DUCTAL ADENOCARCINOMA
PDAC represents about 90% of pancreatic masses and is most commonly located in the head of the pancreas (75%).
In the early stages of the disease,
PDAC is usually asymptomatic and frequently undetectable with US or CECT.
The onset symptoms depend directly on the size and localization of the mass: head masses will determine compression symptoms of the biliary tract and duodenum,
while body-tail masses are frequently associated with a delayed set of systemic symptoms.
Most common symptoms are jaundice,
ill-defined localized abdominal pain,
weight loss,
asthenia,
and anorexia.
US is frequently the first-line diagnostic exam for patients presenting with jaundice or abdominal pain.
Typical findings on US are the presence of a hypoechoic and hypovascular mass,
associated with dilated intra/extrahepatic bile duct,
if the mass is located in the head of the pancreas,
due to compression.
If the body of the pancreas is visible we can detect associated dilatation of main pancreatic duct (double duct sign).
Pancreatic body and tail masses are quite difficult to detect because of the absence of direct and indirect signs.
US sensitivity and accuracy are also highly dependent on the operator’s experience,
the degree of disease progression,
and patient's habitus,
and reported as anywhere between 50-90%.
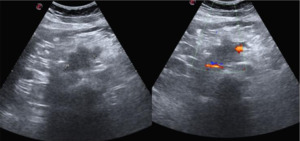
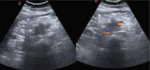
Fig. 4: 56-year-old woman accessing the ER for abdominal pain, with high hepatic enzymes.
Trans-abdominal US detects a hypoechoic and hypovascular mass (ø 3,8 cm) in the pancreatic head.
References: Department of Radiology, AOUP Paolo Giaccone, University of Palermo / Italy 2018
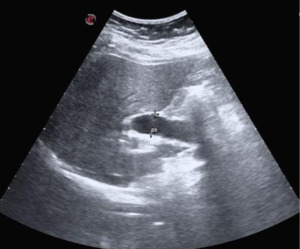
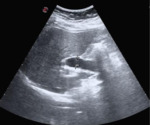
Fig. 5: Trans-abdominal US associated dilatation of common bile duct (ø 1,5 cm) in same patient with pancreatic head mass. Not showed: hydropic gallbladder, hepatic metastasis.
References: Department of Radiology, AOUP Paolo Giaccone, University of Palermo / Italy 2018

Fig. 6
Contrast-enhanced Computed Tomography (CECT) is the most widely used imaging modality for pancreatic tumors evaluation with a sensitivity between 76% and 92%,
permitting accurate surgical planning and staging assessing morphology,
ductal anatomy,
and relationship with surrounding organs and vascular structures.

Fig. 7
Typical findings on CT include detection of ill-defined mass hypodense on non-contrast,
pancreatic parenchymal/late arterial (40-45 sec) and portal phase (65-70 sec) in 75-90% of cases.
10-15% of PDAC appear as isodense mass.
In these cases,
indirect signs such as double duct sign,
atrophy of the pancreatic tail,
or fullness of the pancreatic head (loss of the lobular appearance of the pancreatic parenchyma) may be helpful.
Dilatation of intrahepatic biliary tracts and hydropic gallbladder are other indirect signs that may help to orient the suspect.
Main limits of CECT are in the depiction of small metastases to the liver or peritoneum or primary pancreatic tumor <1 cm showing iso-attenuated.
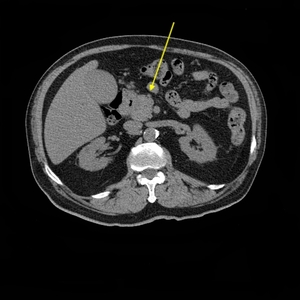
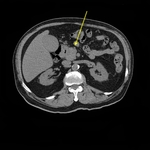
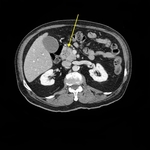
Fig. 8: 72-year-old man presenting with abdominal pain, dysphagia and weight loss.
Axial CT, non-enhanced phase. Enlarged pancreatic head.
References: Department of Radiology, AOUP Paolo Giaccone, University of Palermo / Italy 2018
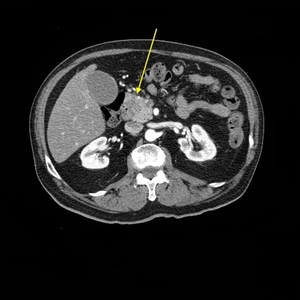
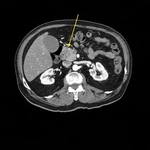
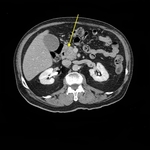
Fig. 9: Axial CT pancreatic late arterial phase, hypodense lesion of pancreatic head, ø 3cm, regular opacification, caliber and course of the mesenteric vessels; lesion appears not dissociable from duodenal second portion; patient underwent Whipple procedute. Histology confirmed suspicion of pancreatic adenocarcinoma.
References: Department of Radiology, AOUP Paolo Giaccone, University of Palermo / Italy 2018
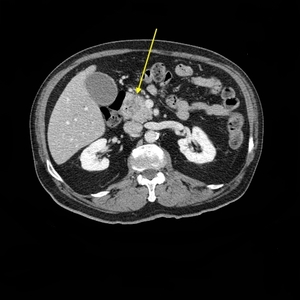
Fig. 10: Axial CT, portal venous phase, hypodense PDAC of pancreatic head.
References: Department of Radiology, AOUP Paolo Giaccone, University of Palermo / Italy 2018
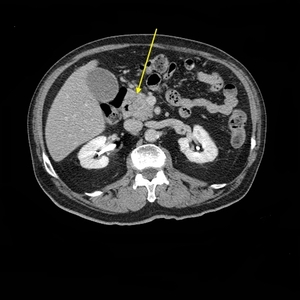
Fig. 11: Axial CT, delayed phase, inhomogeneous hypodense PDAC of the pancreatic head.
References: Department of Radiology, AOUP Paolo Giaccone, University of Palermo / Italy 2018
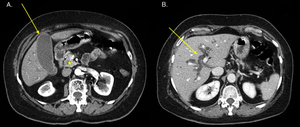
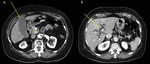
Fig. 12: 70-year-old woman, presenting whit jaundice and elevated liver function tests.
Axial CT, pancreatic phase. (A) Hypodense small mass (ø 1,5 cm) of the head of the pancreas (star), associated with indirect signs: hydropic gallbladder. (B) Dilatation of intrahepatic biliary tracts, due to tightened stenosis of common bile duct. Underwent Whipple procedure, biopsy-proven.
References: Department of Radiology, AOUP Paolo Giaccone, University of Palermo / Italy 2018
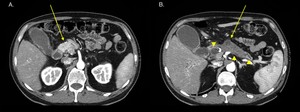
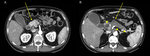
Fig. 13: 60-year-old man presenting with jaundice and dysphagia.
Axial CT, pancreatic phase. (A) Hyperdense pancreatic head parenchima, hydropic gallbladder. (B) Indirect signs of PDAC: atrophic pancreatic tail, dilatation of main pancreatic duct (arrow, arrowhead), dilatation of common bile duct (arrowhead).
References: Department of Radiology, AOUP Paolo Giaccone, University of Palermo / Italy 2018

Fig. 14
MRI may be performed as a second-line method when there's a high clinical suspect but CT does not detect the lesion or when CT with iodine contrast agent is contraindicated.
MRI can also clarify the characteristics of hepatic lesions indeterminate for metastasis in the staging phase.
MRCP can easily identify indirect double duct sign.
Typical findings on MRI are detection of distinct,
well-defined margins low signal intensity and decreased,
homogeneous,
enhancement compared to background normal parenchyma on post-contrast T1-weighted imaging (sensitivity 93% and specificity 75%).

Fig. 15