MRI can be considered a challenge for radiologists,
especially in pediatric patients because it requires accurate preparation and great collaboration from the patient before and during the exam.
Moreover in younger children,
parental participation is essential to achieve greater compliance.
It is important to explain the methods of examination and the function of an adequate preparation.
In our experience their presence is fundamental to reassure the child and to administer the oral contrast.
For the preparation patients should not eat any solid food and drink any fluid for 4-6 hours.
Oral biphasic contrast materials (hypointense in T1w,
hyperintense in T2w) are administrated.
In the literature different oral contrast are mentioned as contrast material with barium,
polyethylene glycol,
solution with polysaccharide (mannose and galactose),
sorbitole,
simple water etc.
with
different patients response due to taste and texture.
We choose PEG solution as oral contrast because children favor its taste.
According to ESGAR/ ESPR consensus statement dosage is 20ml/kg with a maximum of 25ml/kg.
Cold water is preferred to favor progression of oral contrast medium.
Furthermore,
when possible,
parents are advised to move the child (slow walk) to stimulate the passage of oral contrast medium to obtain better bowel distension.
In pediatric patients unable to swallow (for example in general anesthesia) oral contrast is administered via nasogastric tube,
even if this condition takes more time and peristaltic pump may interfere with regular progression.
Administration of a spasmolytic is very useful to decrease peristaltic artifacts,
as GRE T1 sequences may be highly compromised by them.
In literature,
the most frequently used spasmolytic medications in pediatric patients are glucagon and hyoscine-N-butylbromide (butylscopolamine).
We use the latter that is administered intravenously at a dose of 0.5 mg/kg,
with a maximum dose of 20 mg.
Generally half the dose is given before the exam and half before the intravenous contrast medium.
Gadolinium contrast media is administrated in dose 0.1 mmol/kg,
in patients without renal impairment or previous allergic adverse reaction to gadolinium.
Sequence:
In MRI enterography to study pediatric bowel the sequences are the same as in the adult.
TSE-T2 +/- Fat sat,
balanced SSFP not Fat sat and SSFSE,
in axial and coronal plane.
Balanced SSFP is a rapid sequence,
useful to evaluate bowel distention.
If it is adequate,
we proceed with SSFSE in axial and coronal plane.
It provides a good anatomical detail and is less influenced by movement artifacts.
With TSE-T2 +/- Fat sat we assess mural inflammation or edema and changes in peri-enteric fat.
T1w FSPGR sequence is acquired with breath hold before and after intravenous medium contrast.
It discloses pathological contrast enhancement of bowel wall,
lymph nodes,
swelling of mesenteric vessels and complications as fistulous or abscess.
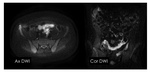
DWI can be used to localize active inflammatory bowel tract,
lymph nodes and eventually complications as fistula and abscess.
It is acquired with lower and highest b-value (50,
600,
900 seconds/mm2).
It could be a marker of activity disease without use of contrast medium.
There is yet standard quantitative parameter in ADC (Fig.
1).
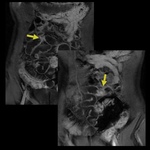
CINE sequence are useful to evaluate peristalsis and to show pathologic dilatations of bowel loop otherwise not visible on static images (Fig.
2-3).
The majority of pediatric CD small bowel findings are similar to adult patients’.
In pediatric CD pan-enteric onset is more typical,
mainly in VEO IBD.
In patients younger than 10 years old colic and upper gastrointestinal tract involvement defines 80% of cases and decreases with age; ileum involvement is rare; perianal involvement is reported in 13%-62%.
it comprendes anal fissures,
perirectal abscesses and anorectal fistulas.
The typical inflammatory bowel features include skip or continuous lesions with bowel wall and fold thickening,
wall hyper- enhancement (stratified or diffuse),
mucosal ulceration,
intramural and mesenteric oedema and increase mesenteric vascularity.
Abnormal wall thickening is over 3 mm.
It is measured in extended small bowel.
It may be either concentric or eccentric and either smooth or nodular.
It is often accompained by wall enhancement,
that may be stratified or diffuse (Fig.
4).
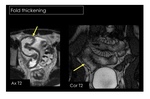
Diffuse thickening fold appears as reduction or distortion folds (Fig.
5).
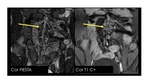
Acute wall edema is detected with high signal intensity within thickened bowel wall and,
sometimes,
in the adjacent mesenter.
Best seen in Fat saturated sequences (Fig.
6).
Also lymph nodes appear enlarged,
hyperenhanced,
typically along the vascular supply of the affected segment.
Best seen on T2w SSFP and on T1w post contrast (Fig.
7).
Increased mesenteric vascularity appears with classical comb sign.
It is observed as high-signal intensity parallel lines on contrast enhanced sequences (Fig.
8).
Ulcers appear as thin lines of high signal,
oriented longitudinally or transversally within the thickened wall (Fig.
9).
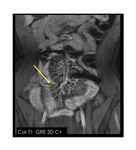
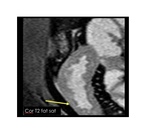
The fistulizing/perforating subtype is caused by transmural inflammation,
it is characterized by deep ulcers.
Complications are sinus tracts,
fistulas and/or abscesses,
they generally develop several years after diagnosis.
Fistulas are transmural ulcers that communicate with adjacent epithelial surface,
while sinuses are blind-ending tracts arising from bowel and does not reach other epithelial surfaces.
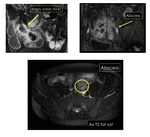
Abscesses are often seen in severe active forms.
Best seen in T2w and post contrast medium (Fig.
10).
In chronic forms,
inflammation progresses towards fibro-stenotic complications.
MRI is less sensitivity to detect mural fibrosis.
Hypointensity of bowel wall on T2-w images,
little mucosal enhancement,
and minimal transmural enhancement suggest chronic fibrosis in the bowel segment.
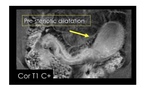
Bowel narrowing is a unusual feature at onset.
In the progressive form it could determine a SBO.
Strictures are defined as luminal narrowing with pre-stenotic dilatation (>3 cm).
It is determined by inflammatory and fibrotic components.
It is important to identify the major components to find the most adequate therapy; several ways (DWI,
late enhancement sequences etc) to differentiate them are being used; still,
as 2018,
no global consensus has been reached (Fig.
11).