LV diastolic dysfunction occurs in several cardiac and systemic diseases which involve abnormalities of active ventricular relaxation and passive ventricular compliance,
leading to ventricular stiffness.
Several factors can cause LV stiffness including age,
increased LV wall thickness relative to cavity size (such as in hypertension,
aortic stenosis or hypertrophic cardiomyopathy),
intracellular changes in microtubules,
extracellular changes in collagen and infiltration (such as in amyloidosis or sarcoidosis).
Other less common causes of stiffness and impaired LV relaxation are idiopathic restrictive myocarditis,
storage disease (hemochromatosis,
Fabry disease) and endomyocardial disease (such as endomyocardial fibrosis,
effects of radiation or anthracycline therapy).
Furthermore,
a constrictive pericarditis must always be considered in the differential diagnosis of diastolic dysfunction.
All these factors cause abnormal LV diastolic distensibility,
impaired filling,
and slow or delayed relaxation.
In fact,
while in normal hearts most of the LV filling (70-80 %) occurs in early diastole at a normal pressure,
in cases with impaired LV relaxation,
filling progressively shifts to the later part of diastole and left atrial (LA) contribution to cardiac output increases.
Abnormal relaxation and increased LV stiffness cause progressively higher diastolic pressures,
which are transmitted through atrial and pulmonary venous systems,
reducing lung compliance.
This cascade of events leads to a decreased cardiac output and to a consequent inability to augment end-diastolic volume despite increasing LV diastolic pressure (as normally occurs during exercise).
Therefore,
physiologic or pathologic stressors,
such as a hypertensive crisis,
can overcome compensatory mechanisms and even result in pulmonary edema.
Diastolic dysfunction can be classified into four grades (Fig 1): in grade I,
the impaired LV relaxation cause a reduction of the early diastole ventricular filling with a relative compensatory increase of atrial contribution during the late diastole.
As diastolic dysfunction worsens,
the contribution of atrial contraction decreases at cost of an increase in LV pressure,
with a pseudo-normalization of ventricular filling pattern (grade II).
Grade IIIis characterized by a severe impairment of LV compliance and relaxation,
with progressive increase of LA pressure and LV diastolic filling pressures and a very abrupt flow of blood from LA into LV,
which is partially reversible with the Valsalva maneuver.
Finally,
in Grade IVthese mechanisms become irreversible.
Cardiac MR evaluation
LV diastolic function is assessable with invasive or noninvasive imaging techniques; although left-sided cardiac catheterization is considered the reference standard for diastolic impairment assessment,
it is an invasive tool and it’s not always feasible for a routine use.
Echocardiography is the most widely used non-invasive method for the evaluation of LV dysfunction,
revealing the typical structural and functional abnormalities in this condition,
giving also information about its severity.
However,
thanks to its excellent spatial resolution,
CMR imaging has gained an important role in the assessment of diastolic function,
and several imaging tools are currently used in clinical practice.
(1) Left Atrial (LA) volume. LA dilatation can be considered a useful marker for chronic diastolic dysfunction because it represents a direct consequence of elevated LV filling,
being primary involved by LV filling pressure during diastole.
Its enlargement is an important predictor of adverse events,
such as Atrial Fibrillation and stroke and it is associated with a poor prognosis.
LA quantification techniques rely on measurements of LA area or volume,
which have to be performed at end-systole (maximal LA size) and it is assessed with SSFP cine sequences.
The most used measurement methods are:
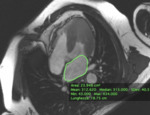
- the 4-chamber planimetry with a manual contouring (area > 24 cm2 means dilation) (Fig 2);
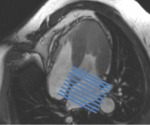
- Simpson volumetric method,
with a manual or semi quantitative delineation at the short-axis sequences (Fig 3);
- Biplane area-length method,
in which LA volume is obtained by defining LA area and anterior-posterior LA length,
on both 4- and 2-chamber views.
(2) LV filling curve.
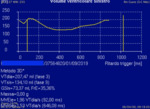
It was the first tool to be developed and it allows the evaluation of LV volume during cardiac cycle.
It is assessed using SSFP cine sequence in short axis,
countouring manually all cardiac phases of all LV slices (200-300 images).
Volume data are plotted in a volume/time curve ant its first derivate (LV dV/dt),
results in a curve similar to TMF curve (but with a much longer post-processing time).
(Fig 4a; Fig 5)
(3) Transmitral Flow (TMF).
Mitral inflow velocities from LA to LV (Transmitral flow,
TMF) is a marker of LA/LV gradient filling which is determined from through-plane phase-contrast (PC) imaging,
with a section positioned parallel to the mitral annulus and perpendicularly to the LV inflow.
The TMF curves obtained from the analysis of these images are composed of two waves: E wave,
which represents the rapid ventricular filling and depends on the pressure gradient between LA and LV,
and the A wave,
which corresponds to the late LV filling after LA contraction.
Normally the E/A ratio is >1 because of the majority of LV filling is due to the early rapid filling.
(Fig 4b) This ratio usually decreases in the I grade diastolic dysfunction (in which abnormal relaxation prevails) while it drastically increases in restrictive types (grade III and IV).
(Fig 6a)
(4) Pulmonary venous Flow (PVF).
It represents an imaging tool which allows the differentiation between normal and pseudo-normal TMF pattern and it is assessed with phase contrast cine sequences using a through-plane positionated perpendicularly to the right upper lobe pulmonary vein,
about 1 cm from the ostium.
The PVF is usually composed of three waveforms: the systolic wave (S) represents the LA inflow during ventricular systole,
the diastolic wave (D) which corresponds to the rapid filling during the early diastole and the A wave,
which is due to late filling during the LA systole.
(Fig 6b)
(5) Late Gadolinium enhancement (LGE),
T1 mapping and Extracellular Volume (ECV). LGE sequences are critically important in the overall assessment of diastolic dysfunction,
allowing the identification of specific patterns of fibrosis and scarring in many of the cardiomyopathies that may initially present with HFpEF.
However,
a condition of diffuse interstitial fibrosis,
which typically occurs in infiltrative cardiomyopathies,
can remain undetected using conventional LGE; in these cases,
the use of T1 mapping (both pre- and post-contrast) and the calculation of extracellular volume (ECV),
are essential for early diagnosis and for the subsequent follow-up.
(6) CMR research techniques.
- Myocardial Tagging: it’s a cine sequence with saturation lines or grids overimposed to the myocardium; diastolic function is assessed by calculation of strain,
strain rate,
twisting and untwisting movements during the cardiac cycle,
which are useful markers od myocardial deformation;
- Myocardial Tissue Phase-contrast: diastolic function is assessed by evaluation of myocardial velocities during diastole and systole,
similarly to echocardiography;
- Myocardial feature tracking: using the SSFP cine sequences feature tracking post-processing tool allow strain and strain rate calculation;
- MR Elastography (MRE): allows the evaluation of myocardial stiffness (similarly to extra-cardiac conditions);
MR Spectroscopy (MRS): active myocardial relaxation is assessed by means of its adenosine triphosphate-dependent energetic phenomenon.