Patients Selection
Two-hundred thirteen patients that underwent SABR at our Institution from January 2013 to January 2017 were retrospectively evaluated.
Inclusion criteria were: an available pre-treatment contrast-enhanced computed tomography (CT) and a minimum imaging follow-up of 6 months at our Institution.
Fifty patients (N = 50) were enrolled.
Demographical and clinical information were collected for all patients,
including age,
tumor size,
tumor site,
histological subtype (Table 1),
number of treated nodules (Table 2) and SABR delivery technique.
Nine patients presented synchronous nodules and were treated concomitantly; 3 patients were treated on a site already irradiated (“retreatment”).
CT Technique
All CT scan were performed on two multidetector CT scanners (GE Lightspeed 16 Ultra and Lightspeed Pro32,
General Electric,
Milwaukee,
USA). Iodinated contrast media dose was proportional to patient’s body weight and was administered at a flow rate of 2-3 ml/s followed by a bolus of saline solution.
Acquisition was performed with a 70 seconds delay time after contrast injection.
CT scan data were acquired using the following parameters: 120 kVp,
100-300 mA,
5 mm slice thickness with 1.25 mm collimation.
Image acquisition was performed with Kernel and 5mm slice thickness,
standard automatic mA modulation.
Image reconstructions at 1.25 mm were obtained and used for multiplanar reformations in order to better analyze small lung nodules and bone structures.
Images were then transferred to PACS for evaluation,
reporting and archieving.
CT evaluation
Image interpretation was performed by a single radiologist that analyzed pre-treatment and post treatment CT.
For all the target lesions the radiologist analyzed all the HRFs of recurrence at follow up according to standard criteria and then according to Nguyen system (5); the final decision of response or local recurrence was then evaluated considering all the informations obtained from clinical evaluation,
laboratory testing,
tumoral markers and further follow up,
by the multidisciplinary lung disease team.
During image evaluation the radiologist was unaware of patient clinical status.
Nguyen and several international opinion leaders in SABR,
using the Delphi method,
identified the following HRFs of local recurrence: Infiltration into adjacent organs/structures,
sustained growth over serial scans,
bulging margins,
mass-like growth,
predominantly spherical growth,
craniocaudal growth,
air space obliteration/loss of air bronchograms.
Several opinion leaders in SABR follow up evaluation such as Nguyen et al (5) and Huang et al (12) suggested to use a cut off of 3 combined HRFs of LR to have a better diagnostic accuracy.
The radiologist furthermore analyzed acute and late lung injury post SABR.
Acute changes present within the first 6 months while late radiological changes manifest after 6 months (6,8).
Acute radiological changes on computed tomography (CT) scan were subclassified into the following categories: no changes,
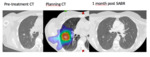
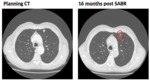
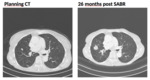
diffuse consolidation (fig.1),
patchy consolidation (fig.2),
diffuse ground glass opacity (GGO) (fig.3),
and patchy GGO (fig.4).
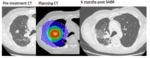
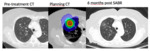
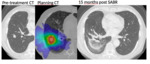
Late radiological changes are categorized into: modified conventional pattern of fibrosis (fig.5),
scar-like fibrosis (fig.6),
mass-like fibrosis (fig.7) and no changes.
Patient setup and planning.
In order to account for nodule motion during all phases of the respiratory cycle,
each patient underwent a slow CT scan simulation in the supine position.
The All-in-One (AIO) system (Orfit Industries n.v.
Antwerp,
Belgium),
which includes thermoplastic masks or abdominal compression,
was used for immobilization.
Permanent marks on the skin and mask were used to align patients to the laser and AIO systems.
The Eclipse™ (Varian Medical Systems,
Inc.,
Palo Alto,
CA,
USA) treatment planning system was used to contour target volume and organs at risk (OARs).
The clinical target volume (CTV) corresponded to the gross target volume (GTV).
The planning target volume (PTV) was obtained by adding a 3-5-mm margin isotropically from the internal target volume (ITV) that encompass the hysteresis movement.
SBRT delivery:Total doses and fractionations.
Patients were treated with different fractionation schemes depending on the tumor stage (T),
tumor location (peripheral vs.central),
and evidence of a primary lung tumor or metastatic disease (Table 3).
The median number of fraction was 4 (range 4-8) delivered twice a week up to a total dose of 48 Gy (range 48-60).
Central tumors (less than 2 cm from mediastinum) were treated with 50 Gy in 5 fraction once a week,
due to their higher risk of major toxicity (9).
The biological effective dose (BED) for tumor and normal tissues was estimated for each adopted fractionation schedule according to the linear-quadratic formula .
An α/β ratio equal to 8-10 Gy was used for primary and secondary tumors,
while an α/β equal to 3 Gy was used for late-responding normal tissues (10). Patients were treated with Helical Tomotherapy (HT Accuracy Sunnyvale California,
USA) and by Volumetric Modulated Arc Therapy (VMAT Varian Medical Systems Palo Alto,
California,
USA).
Statistic analysis
Outcomes as local control (LC) and overall survival (OS) have been predicted by Kaplan-Meyer analysis with JMP V 13.2 SAS.
The LC has been evaluated from the clinical reports of the patients,
assessed by the radiation oncologist (or multidisciplinary team) and integrated by functional and morphological imaging (FDG-PET and/or CT scans).
We called this type of local control evaluation as “type A”.
Based on imaging features we differentiated the LC defined strictly by Nguyen HRFs as “type B” and LC combining Nguyen criteria with radiologist expertise in lung SABR follow up
as “Type C”.
Sensitivity,
specificity and predictive values of HRFs were calculated on the basis of clinical outcomes.