62 patients were eligible to participate in a prospective study from February to October 2018.
Mean patient age was 56 (range,
45-85).
The sex-ratio was 1,58.
They presented with abdominal pain,
weight loss,
anorexia,
asthenia,
and/or jaundice.
Ultrasound revealed only secondary signs of pancreatic cancer: pancreatic duct (PD) dilatation and/or contour abnormalities (n=41),
and/or an hypoechoic pancreatic mass (n=12),
or was normal (n=9).
- MULTIDETECTOR COMPUTED TOMOGRAPHY (MDCT)
All the patients underwent CT scan using a 16 slice machine.
Immediately before scanning,
the patient was asked to ingest 900–1,000 mL of water as a negative intraluminal contrast agent.
MDCT included a three-phase helical CT protocol: a phase without injection,
a pancreatic (delayed arterial) phase,
an hepatic (portal) phase after the injection of 1.5 mL/kg of an iodinated contrast agent,
at a rate of 4–5 mL/sec with thin-section imaging (≤3 mm) and multi-planar reconstructed images.
- The pancreatic phase
This phase is acquired 35–50 seconds after the start of the injection and is timed for peak parenchymal enhancement to maximize the difference in enhancement of the hypoenhancing tumor and background pancreas in order to increase conspicuity of the mass.
Tumor-pancreas contrast was significantly greater during the "pancreatic parenchymal" phase than the "maximal hepatic enhancement" phase due to both greater enhancement of normal pancreas during the earlier acquisition and gradually increasing tumor enhancement.
Arterial and venous enhancement was significantly greater during pancreatic parenchymal phase,
including the celiac,
hepatic,
superior mesenteric artery and vein (SMA,
SMV),
and portal vein.
This phase provides a detailed analysis of spread to local and regional blood vessels,
particularly the arteries,
and also ensures the identification of arterial anatomic variants that may influence the treatment strategy. It allows a better detection of vascular invasion.
2. The hepatic phase
The hepatic (portal) phase acquired 70–80 seconds after the start of the injection provides optimal evaluation for involvement of adjacent veins (mesenteric,
portal and splenic) because of flow artifacts in the superior mesenteric vein,
observed in the pancreatic phase.
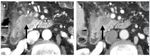
Fig. 17
Also,
this phase serves to detect peritoneal and liver metastases.
Small liver metastases can be missed on CT resulting in unresectable disease being found at surgery.
Routine acquisition of images in the arterial phase is unnecessary for detection of pancreatic tumors because tumor-to-gland attenuation differences are smaller than that obtained with pancreatic and hepatic phases.
- MAGNETIC RESONANCE IMAGING (MRI)
MRI was indicated when the tumor was occult on CT (n=11) and was particularly useful for the detection and characterization of isoenhancing pancreatic masses that are not directly seen on CT.
In our study,
MRI was performed with a 1.5-T system.
The protocol included : fat-saturated transverse and coronal T2-weighted,
fat-saturated transverse T1-weighted,
diffusion-weighted and contrast-enhanced fat-saturated transverse T1-weighted sequences after intravenous injection of gadobenate dimeglumine at a dose of 0.1 mmol/kg.
Findings were considered to be positive if images from at least one sequence revealed the lesion.