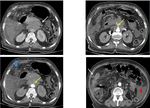
The typical appearance of pancreatic tumors on MDCT was an ill-defined mass which was hypoenhancing relative to the avidly-enhancing non-tumoral pancreatic parenchyma.
Approximately 3% of tumors dis not appear as a focal mass but as diffuse gland enlargement/involvement.
5% of adenocarcinomas was isoenhancing to the pancreatic parenchyma and were occult on CT,
particularly when small.
In these cases, CT showed at least one on indirect findings.
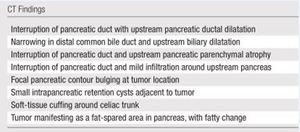
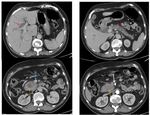
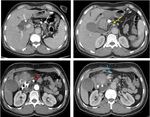
Fig. 19: CT Findings in Visually Isoattenuating Pancreatic Adenocarcinoma Patients
References: Visually Isoattenuating Pancreatic Adenocarcinoma at Dynamic- Enhanced CT: Frequency, Clinical and Pathologic Characteristics, and Diagnosis at Imaging Examinations, Jin Hee Kim et al, Radiology 2010; 257:87–96
The most common finding was the interruption of the main pancreatic duct and upstream pancreatic ductal dilatation but without any visible mass or nodule.
Visually isoattenuating pancreatic adenocarcinoma was defined as such when both of the following criteria were fulfilled:
- No pancreatic lesion compared with the pancreatic parenchyma
- No CT findings of advanced chronic pancreatitis or severe obstructive pancreatitis (ie,
pancreatitis secondary to pancreatic ductal obstruction by tumor) were observed.
The attenuation difference between visually isoattenuating pancreatic adenocarcinoma and both the pancreatic parenchyma was within 10 HU,
demonstrating absence of severe obstructive pancreatitis.
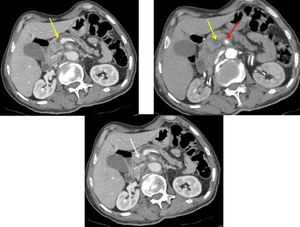
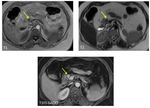
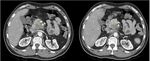
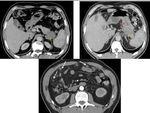
Fig. 16: An unresecable pancreatic ductal adenocarcinoma of the neck and the body of the pancreas. CT images show an isoattenuating tumor (white arrow) with dilatation of upstream pancreatic duct (yellow arrows) and narrowing of SMA (red arrow).
- MAGNETIC RESONANCE IMAGING
The typical appearance of tumors on MRI was an ill-defined T1 hypointense,
T2 hypointense,
relatively hypoenhancing mass.
They demonstrated restricted diffusion,
which may allow for increased detection of tumors even in the unenhanced state or isoattenuating on MDCT.
The most clinically relevant diseases to be distinguished from visually isoattenuating tumors would include benign focal pancreatic ductal stricture and the focal type of autoimmune pancreatitis.
Considering the highly specific nature of MRI,
the distinction between visually isoattenuating tumors and benign focal pancreatic ductal stricture by using these examinations would be straightforward.
On the other hand,
the distinction between visually isoattenuating tumor and autoimmune pancreatitis can be problematic,
as autoimmune pancreatitis can manifest as a mass or nodule at MRI.
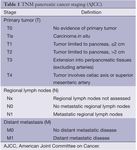
The TNM staging system from the American Joint Committee on Cancer is the most commonly used to assess the extent of disease.
Fig. 1
The resectability of a tumor is dependent on its:
1. Tumor location
Location of the tumor determines the route of local spread of disease and thus,
whether the patient would be treated with a pancreaticoduodenectomy (Whipple procedure) or distal pancreatectomy.
Fig. 2 Fig. 5
In our study,
43 tumors (69,3%) were centered in the pancreatic head,
3 in the uncinate process (4,8%),
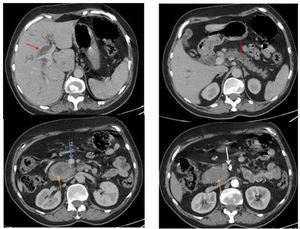
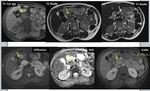
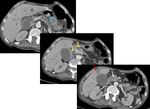
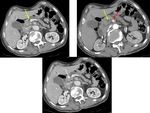
Fig. 6: A resecable pancreatic adenocarcinoma in a 56 year-old-women involving the head and the uncinate process (orange arrow) with dilatation of pancreatic, intrahepatic and extrahepatic ducts (red arrows). The tumor is staged T2 > 4cm, without vascular involvment of SMA (white arrow) and SMV (blue arrow). There is a nodal involvment (N1) and no liver metastasis (M0).
9 tumors in the body (14,5%)
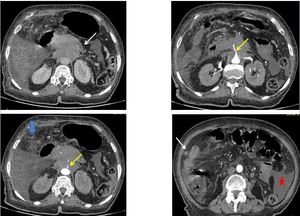
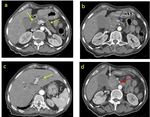
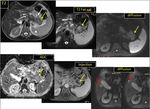
Fig. 14: An unresecable pancreatic ductal adenocarcinoma in a 78-year-old women involving the body and a part of the head, with dilatation of upstream pancreatic duct (white arrow), vascular involvement :narrowing of celiac axis and SMA (yellow arrows). There is also a metastatic peritoneal thickening (blue arrow) and ascites (red star)
and 7 tumors in the tail (11,3%)
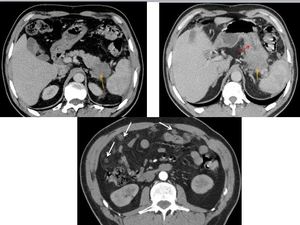
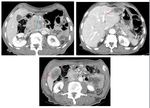
Fig. 15: An unresecable tail pancreatic adenocarcinoma (orange arrows) in a 62 years old man with involvment of the stomach (red arrow). There are many peritoneal nodules (a distant metastatic disease). It was staged T3N1M1.
The size of the tumor is also important,
as it contributes to the T stage and could be important for determining response to the therapy on subsequent studies.
Direct invasion of adjacent structures (n=14) (duodenum,
stomach,
adrenal gland,
kidney,
and colon) did not make disease for a patient unresectable,
if this extension could be safely resected.
2.
Perineural invasion
It refers to retrograde extension of disease along the neural fascicles of the neurovascular bundles.
It is indicative of a very poor prognosis.
Perineural invasion is extremely common with pancreatic carcinomas of the head and uncinate process,
being reported in up to 53-100% of cases,
and often results in positive resection margins at surgery.
Perineural invasion of head tumors can be seen on MDCT as direct contiguous extension of tumor soft tissue extending posterior to the portal vein to along the medial upper margin of the uncinate process or along the GDA to the common hepatic artery (CHA),
respectively.
Perineural invasion of Uncinate process tumors can be seen as direct contiguous tumor soft tissue extending along the posteroinferior pancreaticoduodenal artery (PIPDA) up to and along the SMA.
Note is made that tumor can also extend along this pathway to involve the mesenteric root.
If stage IV is identified in the liver,
a critical analysis of the peripancreatic vessel involvement is not necessary.
The limitation of imaging staging is that perineural invasion is not routinely assessed.
3.
Vascular involvement
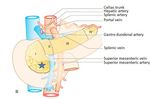
Evaluation of involment of the celiac artery,
SMA,
CHA,
SMV,
and portal vein are essential for accurate staging and determining subsequent therapy.
It is the most important component of determining the resectability of a borderline or locally advanced tumors.
Note that isolated tumor involvement of the pancreaticoduodenal artery does not constitute borderline resectability or unresectability,
as this vessel is routinely resected as part of a Whipple procedure.
The degree of vascular contact is best evaluated perpendicular to the long axis of the vessel,
so,
for example,
the SMA and SMV should be assessed on axial images,
while a coronal or sagittal reformatted image might better demonstrate involvement of the portal vein and CHA.
Imaging signs of vascular invasion are:
- encasement (>180˚ circumferential contact) of a vessel by tumor,
- tumor causing vessel deformity (tear-drop configuration) or narrowing (regardless of degree of contact),
vessel irregularity,

Fig. 14: An unresecable pancreatic ductal adenocarcinoma in a 78-year-old women involving the body and a part of the head, with dilatation of upstream pancreatic duct (white arrow), vascular involvement :narrowing of celiac axis and SMA (yellow arrows). There is also a metastatic peritoneal thickening (blue arrow) and ascites (red star)
- direct invasion into a vessel,
and thrombosis.
Abutment (≤180˚ circumferential contact) of a vessel with tumor is not considered a sensitive sign of vessel invasion.
Fig. 3
Other findings that are not directly related to vascular invasion but otherwise affect surgical planning are :
- extension of the tumor along the CHA to the origins of the right and left hepatic arteries,
- extension of tumor along the SMA to the first branch,
and
- extension of tumor along the SMV to the most proximal draining vein.
In our study,
we found T1 (n=1),
T2 (n=5),
T3 (n=10) and T4 (n=46).
Fig. 4
4.
Nodal disease
Abnormal appearing region lymph nodes (>1 cm in short axis diameter,
rounded shape,
heterogeneity,
central necrosis,
absence of fatty hilum) that are in the surgical bed are considered nodal metastasis and are generally not a contraindication to surgery; however,
if confirmed at surgery,
adjuvant chemotherapy is indicated.
For cancers in the pancreatic head/neck,
this includes lymph nodes along the celiac axis and in the peripancreatic and periportal regions.
For cancers in the body/tail this includes lymph nodes along the CHA,
celiac axis,
splenic artery and splenic hilum.
In our study,
lymph node involvement (N1) was found in 53 cases.
5.
Distant metastatic disease
Lymph nodes outside of the surgical bed (infrarenal or retroperitoneal lymph nodes in a paraaortic location or lymph nodes on the left of the SMA within the jejunal mesentery) are considered distant metastatic disease and are a contraindication for surgery.
Distant metastatic disease most commonly occured in the liver (n=15),
peritoneum (n=16),
lungs (n=24) and bones (n=8).
MRI performs better than does MDCT in detecting small liver metastases..
These findings indicate that liver MRI is mandatory in patients with tumors deemed resectable based on MDCT.
Furthermore,
liver MRI should be performed before and after neoadjuvant therapy for borderline resectable tumors.
Peritoneal thickening/nodularity and/or ascites not otherwise explained have be considered suspicious for metastatic disease.
None of the imaging modalities are sensitive for the detection or early peritoneal disease.
At present,
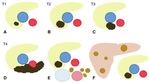
the most widely accepted classification is the National Comprehensive Cancer Network (NCCN) classification.
It is a three-stage classification system for resectability that started in 2006,
to improve patient selection and optimally match the treatment strategy to the spread of the disease.
Fig. 18
1.
Resecable tumors: no evidence of metastatic disease,
no definite involvement (abutment or encasement) of the SMV or portal vein,
and clear fat planes around the celiac artery,
hepatic artery and SMA.
They are still usually treated with primary resection surgery.
2.
Borderline resectable: locally advanced with involvement of the mesenterico-portal veins or local arteries.
They are technically resectable,
albeit often at the expense of vascular reconstruction,
but carry a high risk of positive resection margins and,
therefore,
a poorer prognosis.
Neoadjuvant chemotherapy or chemotherapy and radiation therapy were first developed for borderline resectable PDA. Fig. 9 Fig. 10
3.
Unresectable tumors: metastatic disease (liver or peritoneum),
or local spread that precludes complete resection (occlusion of a major peripancreatic vein by tumor,
encasement of a major peripancreatic artery,
contiguous invasion of adjacent organs that preclude resection such as the stomach and colon).
Fig. 11
In our study,
28 tumors were unresecable (45,2%),
22 were borderline resectable (35,5%) and 12 cases (19,3%) were resectable.
Of the 62 cases with surgical confirmation,
56) were derived from pancreatic epithelial components,
including the most common pancreatic mass,
primary pancreatic ductal adenocarcinoma.
One patient had adenosquamous carcinoma,
one patient had metastatic gastrinoma and 2 patients had primary endocrine cell tumor Fig. 5.