Study population
One hundred and thirty-six consecutive patients with an acute ischemic stroke of less than 8h from symptoms onset were enrolled between February 2015 and May 2016.
All patients underwent non-contrast CT (NCCT),
CT perfusion (CTP),
and multiphase CT angiography (mCTA) on admission.
Only individuals with occlusion of the intracranial internal carotid artery (TICA) or M1 segment of the middle cerebral artery (MCA) proven by CTA and treated with endovascular thrombectomy were evaluated.
After excluding those without evaluable mCTA or CTP,
90 patients were finally included in the study.
Neuroimaging Protocol
All images were obtained at a Siemens SOMATOM Definition AS 128 slices CT.
Standard NCCT was performed from foramen magnum to vertex.
NCCT scans were assessed to rule out hemorrhage and large ischemic core (defined by an Alberta Stroke Program Early CT Score (ASPECTS) bellow 6).
Perfusion CT protocol was as following: 45 seconds acquisition on Cine imaging mode,
at 80 kVp and 250 mA using a 32 sections-slab,
after the injection of 50 mL of IV contrast at 4 ml/s flow.
Multiphase CTA was performed using the following parameters: collimator of 128 × 0.6 mm,
120 kVp,
and 250 mAs,
covering the first phase from the aortic arch until the vertex and the second and third consecutive phases from the foramen magnum to the vertex.
Acquisition was triggered using a bolus tracking (100 HU) in the aortic arch after 60 ml of intravenous contrast injection,
followed by 40 mL of saline with a flow of 4-5 ml/min.
The second and third phases started 4 s after the previous phase.
Intracranial occlusion was determined in the first phase of CTA.
All patients received endovascular reperfusion treatment performed by expert neurointerventionists.
Proximal arterial occlusion was confirmed by DSA before treatment and recanalization was determined by the modified thrombolysis in cerebral ischemia (TICI) score.
A final TICI score of 2b–3 was defined as complete recanalization.
Early clinical evaluation was determined by the difference between baseline and 24h NIHSS.
A 24h NIHSS reduction of more than 4 points or 24h NIHSS<2 was considered as early clinical improvement.
A 24- to 36-hour control CT was performed to detect hemorrhagic transformation of the previous ischemic tissue.
If the hemorrhage was associated with clinical deterioration (NIHSS score increase > 4),
symptomatic intracranial hemorrhage (SICH) was defined.
Infarct volume was measured in the control CT scan by the ABC/2 rule.
Functional evaluation was determined at 3 months by the modified Rankin score (mRS).
Neuroimaging evaluation
CC status and CTP data were not considered for clinical decision making in the acute stroke phase but measured off-line by a stroke neurologist and/or a neuroradiologist blinded to clinical data.
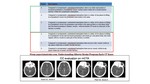
Measurement of the CC status was visually determined by comparing backfilling arteries beyond the occluded artery to similar arteries in the opposite unaffected hemisphere,
according to the Calgary mCTA score.
For statistical analysis,
patients were dichotomized into good and poor CC status: poor CC: 0–2 and good CC: 3–5.
CC evaluation was assessed by an expert stroke neurologist.
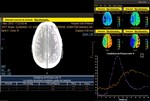
Using Philllips Intelispace ™ workstation tool,
CTP analysis was performed on all individuals.
In the cerebral axial plane,
using the first section above the lateral ventricles in the caudo-cranial direction and demarcating the theoretical superficial territory of the MCA,
a region of interest (cortical ROI,
cROI) was freehand drawn,
following a parasagital line parallel to the cerebral falx,
from the superior frontal gyrus and extending posteriorly.
The external delimitation of the ROI was made following carefully the cerebral hemispheric surface,
excluding the skull.
A second ROI for the study of the deep leptomeningeal circulation was set at the level of the basal ganglia (subcortical ROI,
sROI).
A free-hand drawn area of the theoretical territory of the lenticulostriate circulation,
using the first section where the head of caudate,
lentiform and thalamus nucleus were included in a cranio-caudal direction.
Insular ribbon was included and thalamus nucleus excluded.
A specular image was automatically generated in the healthy hemisphere at both cortical and subcortical levels (Figure).
The usual CTP parameters (time to peak (TTP),
mean transit time (MTT),
cerebral blood volume (CBV),
cerebral blood flow (CBF)) were obtained from both ischemic and healthy hemispheric ROIs.
We studied absolute values from the ischemic hemisphere on cROI (ABS-cROI) and sROI (ABS-sROI).
A second value,
called relative ROI (REL-sROI and REL-cROI),
consisted in a ratio resulting from the division of the values of the ischemic hemisphere by the value of the healthy hemisphere.
CTP evaluation was performed by a neuroradiologist blinded to clinical and mCTA data.
Statistical analysis
Categorical variables are presented as absolute values and percentages,
and continuous variables as mean and standard deviation (SD) if normally distributed or median and interquartile range if not normally distributed.
Statistical significance for intergroup differences was assessed by Fisher’s exact test for categorical variables and by Student t or Mann–Whitney U test for continuous variables.
Wilconxon Signed Rang test was used to evaluate correlation between non-normally distributed continuous variables.
Multivariable logistic regression analyses were performed for each group to determine factors that could be considered as independent predictors of low infarct volume and favorable clinical outcome.
Variables showing P<0.1 in univariate analysis were included in the multivariate model.
A two-sided P value<0.05 was considered significant for all tests.
All statistical analyses were carried out using IBM SPSS 17.0 software.