-
Definition of TPNOCA and MINOCA
Radiologists should understand the conceptual difference between acute myocardial injury and myocardial infarction (AMI). Acute myocardial injury indicates rise and fall of cardiac troponin in serum within two weeks. Persistent or prolonged elevation of cardiac troponin (i.e., more than two weeks) can occur in chronic ongoing myocardial injury in various diseases such as dilated cardiomyopathy, various heart failure, and hypertrophic cardiomyopathy. As the elevation of cardiac troponin is chronic myocardial injury in these conditions, these should not be included in the category of TPNOCA. In contrast to acute myocardial injury, AMI is defined as presence of acute myocardial injury plus at least one of the following findings (symptoms of acute myocardial ischemia, ischemic ECG change, new development of pathologic Q waves, new loss of viable myocardium or wall motion abnormality). Definition of TPNOCA requires presence of acute myocardial injury, regardless of underlying etiology, thus including AMI, pulmonary embolism, myocarditis, or Takotsubo cardiomyopathy plus nonobstructive coronary arteries on coronary angiography, whereas MINOCA only indicates acute myocardial injury caused by myocardial ischemia (i.e., AMI caused by oxygen supply-demand mismatching) and nonobstructive coronary arteries on coronary angiography. The prevalence of MINOCA ranges from 5% to 25% of all AMI. TPNOCA should be subcategorized by epicardial coronary causes [i.e ., myocardial infarction with nonobstructive coronary arteries (MINOCA)] (Fig. 1-2), microvascular causes (i.e., myocarditis, Takotsubo cardiomyopathy), and extracardiac causes.
., myocardial infarction with nonobstructive coronary arteries (MINOCA)] (Fig. 1-2), microvascular causes (i.e., myocarditis, Takotsubo cardiomyopathy), and extracardiac causes.
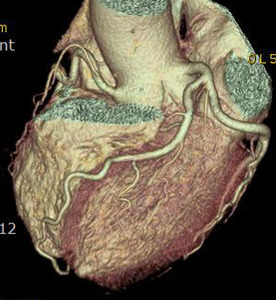
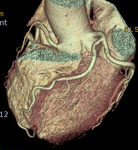
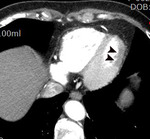
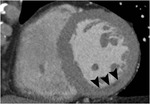
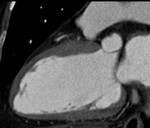
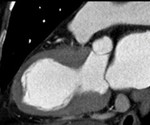
Fig. 1: A case of MINOCA in a 62-year-old female with indeterminate etiology of MINOCA. The patient presented into the ED with acute chest pain, but her initial ECG and cardiac troponin was normal. A. there is no evidence of obstructive coronary artery disease on a volume rendered image. B. Subendocardial perfusion defect along the apical inferolateral wall is noted (arrowheads), suggesting AMI in the left circumflex coronary artery territory.
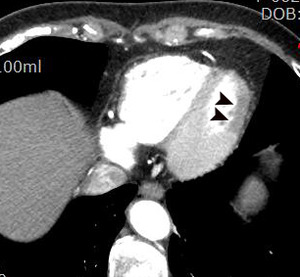
Fig. 2: Same patient with Fig.1. Subendocardial perfusion defect along the apical inferolateral wall is noted (arrowheads), suggesting AMI in the left circumflex coronary artery territory.
-
Various causes of TPNOCA
1. Epicardial coronary TPNOCA  (MINOCA)
(MINOCA)
In contrast to classic AMI, MINOCA is caused by oxygen supply and demand mismatching (type 2 AMI). Possible mechanisms of MINOCA include spontaneous thrombolysis, epicardial coronary artery spasm (CS) (Fig. 3-7), coronary embolism, and spontaneous coronary artery dissection (SCAD). Notably a shared mechanism of MINOCA is myocardial ischemia (decreased perfusion) caused by an event within an epicardial coronary artery.
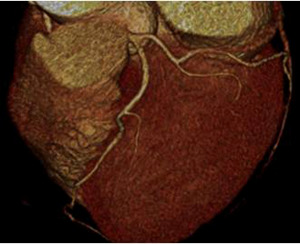
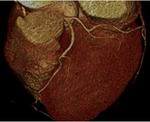
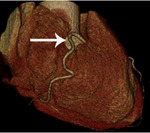
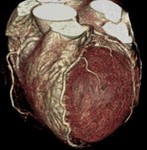
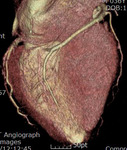
Fig. 3: Fig.3. A case of MINOCA caused by spasm-induced ACS in a 56-year-old male patient. A. Normal coronary arteries are noted on volume rendering images (A and B). Note that proximal middle right coronary artery is normal (arrow) while Troponin I increased (7.92) in this patient.
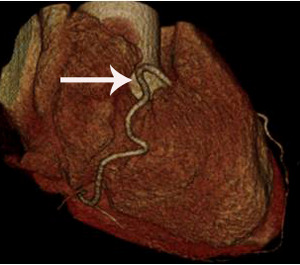
Fig. 4: Fig.4. Same patient with Fig.3.Normal coronary arteries are noted on volume rendering image. Note that proximal middle right coronary artery is normal (arrow) while Troponin I increased (7.92) in this patient.
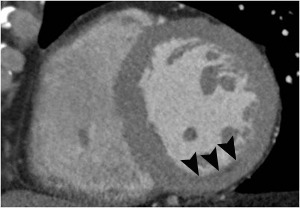
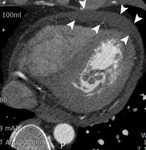
Fig. 5: Fig.5. Same patient with Fig.3. Subtle myocardial perfusion defect (arrowheads) in the subendocardial portion of the inferior wall of mid-portion of the left ventricle, suggesting AMI.
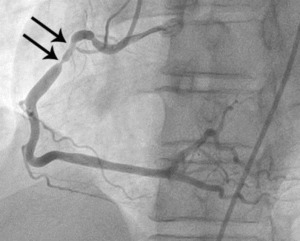
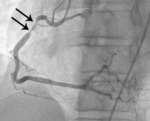
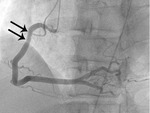
Fig. 6: Fig.5. Same patient with Fig.3. On subsequent ergonovine provocation test, proximal right coronary artery shows critical spasm (arrows) after administration of intravenous injection of ergonovine.
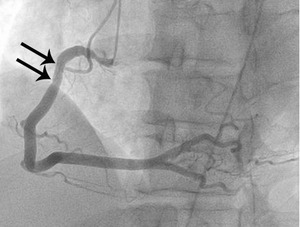
Fig. 7: Fig. 6. same with Fig.3. After administration of intraarterial nitroglycerin, the spasm (arrows) is completely relieved.
2. Microvascular causes of TPNOCA
2-1. Takotsubo cardiomyopathy (TCMP)
TCMP commonly occurs postmenopausal women with emotional or physical stress. The diagnosis of Takotsubo cardiomyopathy (TCMP) should include the following three (Fig. 8-10);
1) transient wall motion abnormality of left and/or right ventricle, extending beyond single coronary artery territory; the wall motion abnormality often improves within a few weeks.
2) nonobstructive coronary artery disease, although obstructive coronary artery disease does not necessarily exclude the diagnosis of TCMP.
3) new ECG change (ST-segment elevation and T wave inversion) simulating ischemia or mild increase in cardiac troponin
Stress induced cardiomyopathy (Takotsubo cardiomyopathy, TCMP) can be also subclassified into microvascular TPNOCA. Although exact pathophysiologic mechanism of TCMP is still unclear, in a study using myocardial contrast echocardiography, acute reversible coronary microvascular vasoconstriction may be a common pathophysiologic mechanism. In this study, extent of perfusion defect and wall motion abnormality significantly was reduced after intravenous adenosine infusion compared with baseline contrast echocardiography. In contrast, ST-elevated AMI patients demonstrated no change. Thus, the study concluded that microvascular dysfunction is a common final etiology although the exact mechanism of this entity remains unclear. If coronary CTA was performed by a retrospective ECG-gating, wall motion abnormality of the both ventricles can be analyzed. Thus, coronary CTA has a potential to diagnose TCMP as cardiac MR do. Characteristic CT features of TCMP are wall motion abnormality not following single coronary artery territory not accompanied by perfusion defect and obstructive coronary stenosis.
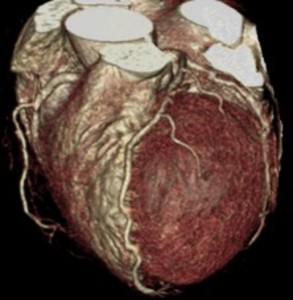
Fig. 8: F/63, Acute chest pain, Elevated cTnI (1.15 ng/mL), ECG change: Non-specific
CCTA with volume rendering image shows normal coronary arteries without significant stenosis
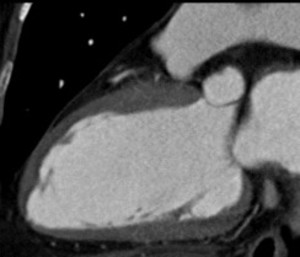
Fig. 9: Same patient with Fig.8. 2-chamber view of diasolic image.
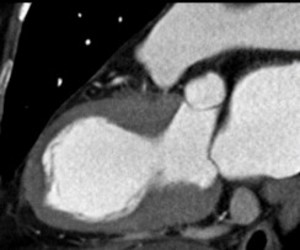
Fig. 10: Same patient with Fig.8. 2-chamber view of systolic image well demonstrates akinesia at LV mid-ventricular wall compared to normal contratile apical wall and hypercontractile basal wall, indicting Takotsubo cardiomyopathy
2-2. Myocarditis
Typical presentation of myocarditis includes antecedent viral illness, fever, pleuritic chest pain, cardiac failure, ST-change on ECG, or wall motion abnormality of the left and/or right ventricle, otherwise unexplained. With typical clinical presentations supported by increased level of c-reactive protein and ECG change, the diagnosis of myocarditis may be straightforward. However, diagnosis of myocarditis is often complicated due to significant overlap of clinical presentations with ACS. Thus, it should be kept in mind that myocarditis is exclusion diagnosis. It should be also stressed that infection of endothelial cells is demonstrated in a certain type of viral myocarditis (e.g., parvovirus B19), leading to microvascular spasm (CMS) and spread of inflammation into myocardial cells. Thus, some of myocarditis can be classified into microvascular cause of TPNOCA. Diagnosis of myocarditis is important because specific treatment can be initiated (i.e., ionotropic agents and/or mechanical circulatory supports and discontinuation of anticoagulation). According to recently revised 2018 Lake Louise criteria for the diagnosis of myocarditis, major criteria of myocarditis includes 1) evidence of non-ischemic myocardial injury on delayed enhanced MR, T1-mapping, or extracellular volume (ECV), and 2) myocardial edema on T2 mapping or T2W image (Fig. 11-14). In this new criteria, early hyperemia is excluded due to its nonspecific feature. Simultaneous presence of two major criteria on MR increases specificity of diagnosing myocarditis compared with those with only one major criteria. In the new criteria for myocarditis, supportive criteria for the diagnosis of myocarditis were also defined; pericardial effusion and wall motion abnormality. Mesocardial or epicardial delayed enhancement is a typical cardiac MR pattern. As routine cardiac CT without delayed scan can only evaluate the supportive criteria for the diagnosis of myocarditis, its potential role for the diagnosis of myocarditis should be limited. However, several reports indicated role of CT in the diagnosis of myocarditis on delayed scan (i.e., 10-15 minutes after administration of contrast media) which can be performed with low dose scan protocol (i.e., 80 kV scan). Even on MR, false negative diagnosis of myocarditis on delayed enhancement MR can occur in mild form of myocarditis. Thus, it should be noted that MR diagnosis of myocarditis has high specificity, but modest sensitivity. In a similar way, it is expected that cardiac CT by using delayed scan cannot identify mild form of acute myocarditis.
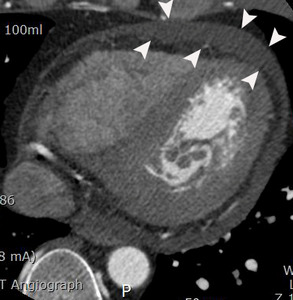
Fig. 11: A case of myopericarditis in a 33-year-old male patient. Moderate amount of pericardial effusion (arrowheads) is noted on an axial image of coronary CTA at the level of the left ventricle. However, there was no wall motion abnormality on cine view of coronary CTA (not seen here). Troponin I and c-reactive protein in the serum increased in this patient.
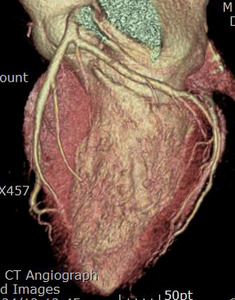
Fig. 12: Same patient with Fig.11. Normal coronary arteries are demonstrated on the coronary CTA.
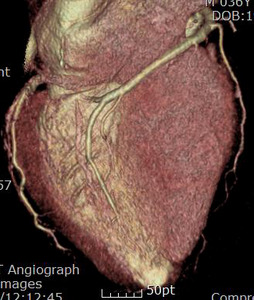
Fig. 13: Same patient with Fig.11. Normal coronary arteries are demonstrated on the coronary CTA.
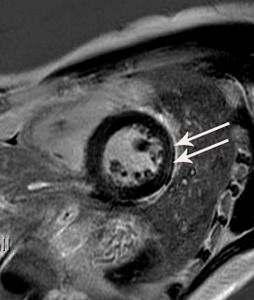
Fig. 14: Same patient with Fig.11. On cardiac MR performed on two weeks after cardiac CT, linear and patch delayed enhancement is noted at the lateral wall of basal left ventricle (arrowheads). The final diagnosis was myopericarditis.
3. Extracardiac causes of TPNOCA
Extracardiac causes of TPNOCA are aortic dissection extending into coronary artery (secondary coronary dissection) (Fig. 15) and pulmonary embolism. Cardiac troponin elevation in serum in patients with sub-massive or massive pulmonary embolism may be caused by the right ventricular stretching or ischemia.
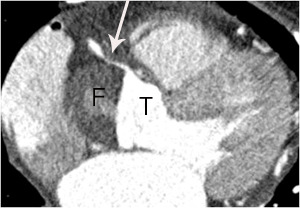
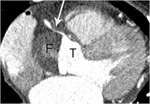
Fig. 15: A case of Stanford type A aortic dissection with secondary myocardial infarction due to right coronary artery extension in a 64-year-old female patient. A. Extension of aortic dissection in the aortic root into the proximal right coronary artery (arrow) is noted on an axial image at the level of left ventricle. F and T indicate false and true lumen.
Summary
Radiologists should try to identify 1) presence or absence of myocardial perfusion defect, 2) extracardiac causes of TPNOCA, and 3) CT feature of Takotsubo cardiomyopathy in patients presented with TPNOCA. The presence of typical myocardial perfusion defect indicates the diagnosis of MINOCA. If there is no myocardial perfusion defect, pulmonary embolism, aortic dissection, or CT features of Takotsubo cardiomyopathy, then cardiac MR can be an appropriate next downstream test to differentiate myocarditis from MINOCA not identified on cardiac CT.