There is considerable overlap in imaging findings between acute and chronic, infectious and non-infections complications in patients with CVID.
1. Infection
Infection is the most important clinical problem in patients with CVID both at diagnosis and during follow-up.3
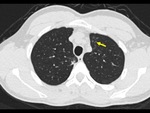
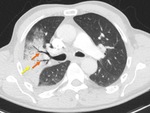
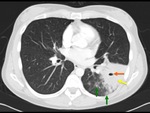
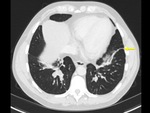
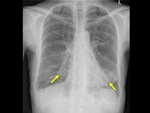
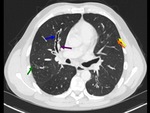
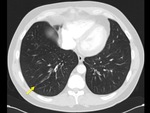
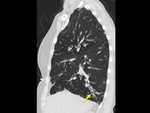
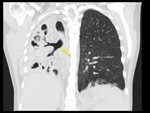
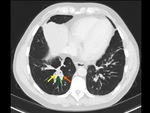
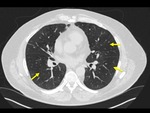
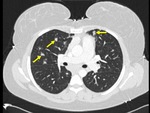
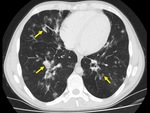
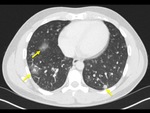
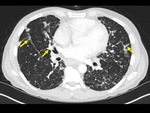
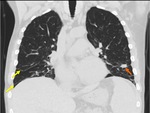
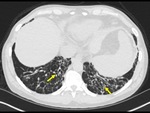
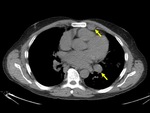
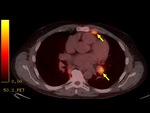
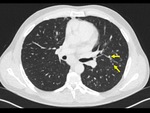
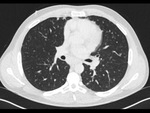
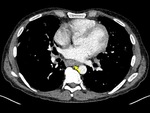
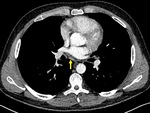
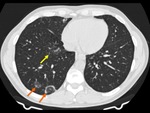
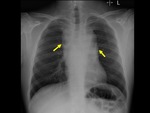
The imaging findings of lower respiratory tract infection in patients with CVID are similar to those in immunocompetent patients. Infection can appear as tree-in-bud nodularity when the bronchioles are affected, consolidation (either segmental or lobar) when the alveoli are affected and/or as pulmonary abscess. Linear parenchymal scarring can occur as a result of recurrent infection.7
Incidence of pneumonia decreases with immunoglobulin treatment.3
See figures 1-4.
2. Bronchial abnormalities/airways disease
Bronchiectasis is seen in up to 73% of patients with CVID.8
Recurrent infection and unregulated inflammation result in structural lung damage.
Typical CT findings of bronchiectasis include:
- Bronchoarterial ratio >1 (i.e. internal luminal diameter of bronchus exceeds diameter of adjacent artery
- Lack of tapering of bronchi
- Bronchi abutting mediastinal pleura/within 1 cm of pleural surface9
There are no pathognomonic imaging features in CVID-related bronchiectasis. Cylindrical bronchiectasis is the most common subtype. The middle, lower lobes and lingula are most frequently affected. There is often associated bronchial wall thickening and mucous plugging.8 Mucous plugging can result in atelectasis/collapse.
Bronchiectasis is associated with increased morbidity and mortality. Early diagnosis with CT is important as prophylactic antibiotics and increased immunoglobulin replacement may help prevent and treat bronchiectasis in patients with CVID.10
See figures 5-10.
Small airway obstruction can result in ventilation abnormalities, which manifest on CT as areas of low attenuation secondary to air trapping. This finding can be enhanced using expiratory-phase CT.7, 11
See figure 11.
3. Granulomatous disease
Granulomatous disease occurs in approximately 8% of patients with CVID.5 The lungs are the most common organ affected, followed by lymph nodes, liver, spleen and skin.12
In a 2017 Consensus Statement, 80% of respondents described solid/semi-solid or ground-glass opacities, thoracic lymphadenopathy and splenomegaly as “necessary” or “typical” for a diagnosis of granulomatous lymphocytic-interstitial lung disease (GL-ILD).13 However, multiple additional imaging findings have been described in GL-ILD including coarse reticulation, usually affecting the lower zones, architectural distortion and traction bronchiectasis.
Given the similarity of CT appearances between GL-ILD and sarcoid, GL-ILD can be considered “sarcoid-like”.14 Indeed, sarcoid as well as infection, organizing pneumonia, lymphoid interstitial pneumonia and lymphoma are included in the radiological differential diagnosis for GL-ILD.
Although CT thorax, pulmonary function tests and bronchoscopy (to exclude infection) should be performed as part of the diagnostic work-up for GL-ILD, lung biopsy is ultimately required to confirm the diagnosis and exclude differential diagnoses.13 Histology reveals granulomatous inflammation and lymphoproliferative changes.13 Treatment involves optimization of immunoglobulin therapy and steroids/immunosuppressants, as required.13
Unfortunately, granulomatosis portends a dismal prognosis.7
See figures 12-24.
Malignancy
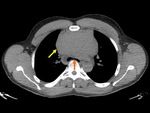
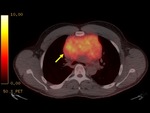
Patients with CVID are at 30 times increased risk of developing lymphoma. Caution must be taken in interpreting lymphadenopathy in patients with CVID. Lymph node biopsy should be performed if there is concern regarding the development of lymphoma.
Radiological signs suggesting lymphoma include anterior mediastinal mass and asymmetric, enlarged mediastinal or hilar lymph nodes that exert mass effect on adjacent structures.7
See figures 25-27.