Neuroimaging strategies in cognitive impairment and dementia
MRI is the primary imaging modality in neurodegenerative diseases to rule out other pathologies and to assess focal brain atrophy and white matter disease.
A brain MRI protocol including structural MRI and high spatial resolution anatomic images is recommended(Alzheimer Centre in Amsterdam)Fig. 2:
- Saggital T1 MPRAGE(magnetization prepared rapid acquisition gradient echo).
- Axial FLAIR(fluid-attenuated inversion recovery).
- Axial T2.
- Axial T2* or Susceptibility Weighted Images(SWI).
- Diffusion Weighted Images(DWI).
Multi-detector CT is used if there are MRI contraindications.
Nuclear medicine(FDG-PET or Amyloid-PET)is a secondary modality in case of unusual clinical presentation, inconclusive MRI or when molecular diagnosis is required(e.g.assessment of amyloid status).
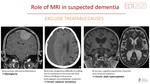
Role of MRI in suspected dementia
1. Exclude treatable causesFig. 3
2. Assessment of brain atrophy(Imaging BIOMARKERS)
- To differentiate normal brain aging from neurodegenerative diseases
- To identify specific atrophy patterns of neurodegenerative diseases
- To identify imaging biomarkers that can help to an early diagnosis and/or predict conversion
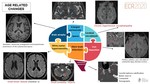
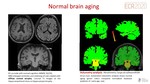
Normal brain aging
With age, normal structural changes are found within the brain secondary to cellular aging, genetic control, environmental facts or lifestyle.
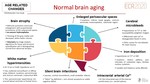
The most relevant structural radiological findings identified on MRI in cerebral aging are(Fig. 4,Fig. 5):
i. Brain atrophy
ii. White matter hyperintensities
iii. Enlarged perivascular spaces
iv. Silent brain infarctions
v. Cerebral microbleeds
vi. Iron deposition
vii. Intracranial arterial Ca2+
These findings are identified in both normal and pathological aging(Fig. 6, Fig. 7).
Fig. 8 resumes several keypoints to help us.
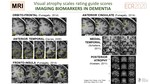
Visual atrophy rating scales proposed for evaluation of cognitive impairement
Several visual rating scales have been developed to facilitate reporting of brain atrophy, providing highly reproducible information especially in the medial temporal lobe and posterior cortical atrophy.
They have been established as MRI imaging biomarkers in dementia and are useful tools in the clinical setting of dementia.
Each scale quantify on a subjective manner a specific region particularly susceptible to change in dementia, providing a score depending on the severity of the atrophic changes(Fig. 9):
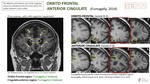
- ORBITO-FRONTALFig. 10
- ANTERIOR CINGULATEFig. 10
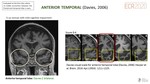
- ANTERIOR TEMPORALFig. 12
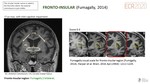
- FRONTO-INSULAFig. 11
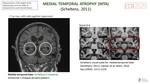
- MEDIAL TEMPORALFig. 13
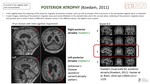
- POSTERIOR ATROPHYFig. 14
Specific MRI atrophy patterns of neurodegenerative diseases
Dementia syndromes include a wide spectrum of diseases where clinical presentation and imaging findings are sometimes overlapping.
We review the most common atrophy patterns in the main entities presenting with dementia:
- Alzheimer’s disease
- Dementia with Lewy Bodies
- Frontotemporal lobar degeneration
- Vascular dementia
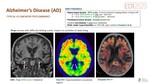
1. ALZHEIMER’S DISEASE (AD)
Is the most common cause of dementia(55% of all cases).
Clinically is characterized by a progressive deterioration that starts with an isolated loss of memory.
An abnormal accumulation of amylid-β in neurons and an abnormal phoshporylation of “tau” protein leads to the development of neurofibrillary tangles and neuronal death.
Imaging findings in typical AD(Fig. 15)
MRI:
- Hippocampal atrophy(MTA scale, Scheltens) with decreasing height of hippocampus and dilated choroid fissure is one of the earliest and most prominent imaging feature of AD although it is not pathognomic.
- In MTA score ≥2 on ONE side–abnormal
- In >75 yo: MTA score ≥2 on BOTH sides–abnormal
- Parietal/precuneus atrophy(Koedam score ≥2)
- Cerebral microbleeds in lobar/mixed distribution: overlap in AD, VaD and Amiloid angiopathy.
- White matter hyperintensities are quite common(Fazekas 1 or 2).
FDG PET or ASL: hypometabolism (PET)/hypoperfusion (ASL) in posterior cingulate cortex and temporoparietal regions. Occipital lobe normally spared.
AMYLOID PET: abnormal amyloid accumulation in cortical distribution (may be present years before onset of symptoms).
TAU PET: abnormal tau accumulation, usually closer related to symptom onset.
Imaging findings in atypical AD
Discrepant findings between clinical presentation and imaging findings may be explained by AD variants or other entities.
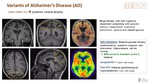
A) Posterior cortical atrophy variant: Early-Onset AD(Fig. 16)
Clinical manifestation as posterior cortical atrophy, with less memory loss compared to typical AD. Occurs at a young age(<65years).
Incidence: 5-10% of all cases.
Sporadic>Familial(mut PSEN1/PSEN2;Amyloid-β(A4)precursor protein).
MRI findings: bilateral parietal atrophy predominance, posterior cingulum and precuneus. Hippocampus can be normal.
B) Behavioral, Dysexecutive or a combination of both presentation of AD
Manifestates as behavioural or dysexecutive deficits.
MRI findings similar to typical AD.
Clinically might suggest FTLD, but image findings are more suggestive of AD. Differential diagnosis is made by clinical presentation.
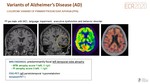
C) Logopenic variant of Primary Progressive Aphasia(PPA)(Fig. 17)
Clinical manifestation: difficulties in word retrieval with reduced speech rate.
MRI findings: predominantly focal left temporal lobe atrophy.
PET-TAU: more pronounced frontal involvement compared to AD.
2. DEMENTIA WITH LEWY BODIES (DLB)
Second most common neurodegenerative dementia(15-20% of all cases).
Belongs to the atypical Parkinson syndromes. Clinically present with cognitive fluctuations, visual hallucinations and parkinsonism.
It may mimic AD clinically, with overlaps pathologically.
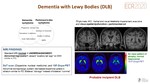
Imaging findings in DLB(Fig. 18)
Imaging findings in DLB and Parkinson disease(PD) are overlapping.
Standard MRI is normal or presents as generalized atrophy with no focal predilection, remaining underdiagnosed. Normal hippocampi.
Evaluation of nigrosome 1 of the substantia nigra with high-resolution SWI can provide additional information in cases of suspected DLB(normally used as an imaging marker of PD). Abnormal nigrosome 1 is seen (absent “swallow tail sign”).
FDG PET: hypometabolism in bilateral parietal regions including occipital lobe (unlike AD).
DaT scan and 18F-Dopa-PET: Abnormal domapinergic system (decreased dopamine uptake in striatum similar to PD: bilateral “dot sign” instead of bilateral “comma”).
3. FRONTOTEMPORAL LOBAR DEGENERATION (FTLD)
Heterogeneous group of neurodegenerative disorders characterized by selective involvement of the frontal and temporal lobes. FTLD results in variable clinical manifestations with behavioral and language variants, which in turn overlap with some related motor degenerative conditions.
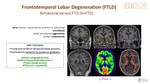
A. Behavioral variant FTD(bvFTD). Also known as “Pick disease”(Fig. 19).
Clinically presents as dysexecutive cognitive syndrome with changes in personality & social behavior. Memory usually preserved.
MRI findings: frontal and anterior temporal lobes atrophy. Characteristic anterior to posterior gradient.
FDG-PET: frontal and anterior temporal lobes hypometabolism.
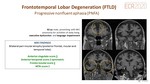
B. Progressive nonfluent aphasia(PNFA) Fig. 20
Progressive difficulties in speech production.
MRI findings: bilateral peri-insular atrophy(posterior frontal, insular and temporal lobe).
FDG-PET: asymmetric left frontal and temporal hypometabolism.
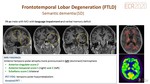
C. Semantic dementia (SD) variant of PPA Fig. 21
Patients present with loss of language comprehension. Speech remains fluent and grammatically faultless.
MRI findings: anterior temporo-polar atrophy more pronounced in left (dominant) hemisphere. There is also a right temporal variant of FTD which is similar to SD but on the right hemisphere.
FDG-PET: anterior temporo-polar hypometabolism.
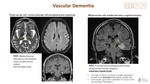
4. VASCULAR DEMENTIA (VaD)(Fig. 22,Fig. 23)
Term used for cognitive dysfunction associated with vascular brain damage.
Accounts for 20% of all cases of dementia.
Frequently patients with manifest AD have coexisting vascular disease which contributes to the demented state, as they have similar risk factors.
Both small or large vessels are involved, however, the majority of cases are microvascular angiopathies.
A. Small vessel disease.
Microvascular leukoencephalopathy(Fazekas score),lacunes and cerebral microbleeds.
Deep or infratentorial distribution of microbleeds are seen in VaD and AD, related to chronic hypertensive encephalopathy. Lobar and cortical distribution are typical of Amyloid angiopathy.
Strategic infarcts are those located in crucial areas for normal cognitive functioning of the brain.
B. Large vessel disease.
Territorial infarcts, usually secondary to carotid disease.
Radiological criteria for probable VaD is measured by the NINDS-AIREN criteria.