Multiple sclerosis (MS) is an inflammatory disease of the central nervous system (CNS) characterized by infiltration of immune cells and subsequent loss of myelin, oligodendrocytes, and axons. Conventional magnetic resonance imaging (MRI) is used routinely for diagnosis and monitoring disease activity.
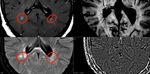
There are many challenges in determining the time (DIT) and space dissemination (DIS) in MS. They were addressed at the latest McDonald criteria, which characterized DIT as at least one new T2 and/or gadolinium-enhancing lesion on follow-up Magnetic Resonance (MRI) compared to a baseline MRI scan, irrespective of the timing or the simultaneous presence of asymptomatic gadolinium-enhancing and non-enhancing lesions at any time. As for DIS, it requires â‹ť1 T2 MRI lesion in at least two typical MS areas (periventricular, infratentorial, spinal cord or cortical/juxtacortical). When available, advanced MRI sequences such as DIR (2 TIs for fat and water) or T2SWI (T2 susceptibility weighted images), should be applied to visualize those cortical lesions.
The T2SWI sequence exploits intrinsic differences in susceptibility to generate contrasts by using magnitude and phase images from a gradient echo sequence (GRE), where the phase image reflects local susceptibility-induced changes of the resonance frequency. In brain tissue, susceptibility is mostly determined by diamagnetic tissue water, the presence of paramagnetic iron, the oxygenation level of blood in capillaries and veins, and the diamagnetic constituents of myelin. In MS, SWI have demonstrated a hypointense rim around lesions and nodular hypointensities in the WM. These findings seem to be relatively specific to patients with clinically isolated syndrome (CIS) or MS over other neurological disorders. Consequentely, the SWI MRI hypointensities may indicate a significant contributor to tissue damage, disease severity, and/or progression. The non-haem iron deposition seems to be a major contributor, but demyelination and free radicals related to inflammatory processes might also have an important role.
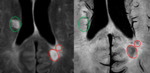
Iron is absent from early active, gadolinium-enhancing lesions as suggested by imaging studies of MS patients and by MRI and histological analysis of MS brain tissue. At this stage, macrophages contain myelin fragments and are M2 polarized, which is associated with low iron content. The Breakdown of the blood–brain barrier (BBB) is detected using gadolinium-diethylene-triamine-penta-acetic-acid. However, experimental imaging studies in MS patients with PET imaging or detect infiltrating monocytes with ultra-small iron oxide particle enhancement visualize patterns of inflammation distinct from Gd-enhancement. Thus, inflammation can occur in the context of an intact BBB and additional imaging modalities are required to obtain a more complete picture of the inflammatory activity in MS patients.
In a recent study that combined ultra-highfield phase MR imaging with histopathological analysis, Bagnato et al correlated MRI phase signal with presence of iron within histological sections. It confirmed that phase imaging detects iron in brain tissue with high sensitivity and identified the cellular localizations of iron depositions. In MS WM lesions, iron was present within microglia/macrophages at the lesion perimeter. In normal appearing WM, iron was found in oligodendrocytes, confirming prior reports that oligodendrocytes are the major iron-containing cells in the adult CNS. In addition, iron precipitates were present in hemosiderin aggregates within and outside of white matter MS lesions suggestive of remote microhemorrhages, but also may be advented form myelin/oligodendrocyte debris, concentrated iron in the macrophages (that phagocytize the destructed myelin/oligodendrocyte). The accumulation of iron in some but not all lesions suggests a potentially disease-relevant process, however its pathophysiological significance remains unknown.
The amount of iron deposition could reflect the extent of tissue damage, so iron could be used as a biomarker to predict clinical outcome. This is a reasonable hypothesis given recent findings, which show iron deposition encircling dilated veins in MS. Some studies appear to indicate that chronic lesions may vanish on T2WI in some instances. This may explain why the number of lesions on T2WI has not been very specific to the severity of the disease.
GRE phase imaging at ultra-highfield MRI is highly sensitive for iron. A number of GRE studies with MS patients and autoptic MS tissue, have demonstrated that iron accumulates in white matter and cortical lesions. Several patterns of phase signal within lesions have been identified, including nodular lesions that were uniformly phase hypointense, lesions with a hypointense rim at the margin and lesions containing veins.
Mehta et al found that phase hypointense lesions were more prevalent in patients with high inflammatory activity (relapse and/or Gd-enhancement within the last 9 months) than in patients with secondary progressive disease. These results confirmed others that have showed that the lesion ages at which phase hypointense signal (and tissue susceptibility) becomes most prominent. The authors found that phase hypointensity increased rapidly within the first 0.5 years and peaked at 0.5–3 years.
In summary, brain iron homeostasis is perturbed in MS, resulting in various pathologic outcomes. First, iron accumulates after initial acute demyelination in activated microglia at the rim of white matter lesions, where it might contribute to long-lasting neurotoxic inflammation. Moreover, with increasing disease duration, iron is slowly depleted from normal appearing white matter, which potentially compromises oligodendrocyte functions including myelin maintenance and contributes to axonal damage outside of white matter lesions. Iron is also depleted in inactive cortical lesions, which may affect neuronal function. Finally, iron accumulates at an increased rate in the deep nuclei of MS patients. Although the pathogenic significance of this has not been fully elucidated, iron deposition reflects has been shown to correlate well with various measures of disability in MS and is associated with neurotoxicity in other neurodegenerative diseases.

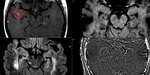
![Fig. 1: Iron deposit in MS plaques: Iron deposition in white matter MS plaques. The overview image 1- shows an oil-red O-stained section with an actively demyelinating lesion [1] and a demyelinated lesion [2]. The actively demyelinating lesion (1) contains an abundance of large, myelin-laden macrophages as indicated by the presence of oil-red O positive material within CD68+ macrophages. These macrophages do not contain iron as indicated by Perls’ staining [d], or the iron storage protein ferritin [e]. In the demyelinated lesion [2], at least two populations of macrophages were observed: in the lesion center [2], small round CD68+ macrophages contain condensed myelin. Occasional macrophages contain iron [j] and correspondingly and ferritin [k]. In contrast, macrophages at the lesion rim (3) have a ramified appearance and do not contain oil-red O positive material. These cells contain large amounts of iron [p] and iron-storing ferritin [q].
Acute lesion: Open BBB - Gadolinium enhancement. Active demyelination.
Chronic active lesions: Closed BBB - No enhancement. High tissue susceptibility. No/slow demyelination.
Chronic silent lesion: Closed BBB. No enhancement. No susceptibility.No demyelination. No inflammatory cells.
References: Adapted from Mehta V et al. Iron Is a Sensitive Biomarker for Inflammation in Multiple Sclerosis Lesions.doi:10.1371/journal.pone.0057573.g001](https://epos.myesr.org/posterimage/esr/ecr2020/156056/media/859389?maxheight=150&maxwidth=150)