Background
Renal biopsy remains a gold standard procedure for the diagnosis of renal disease, which is performed in native and transplanted kidneys and is generally considered a safe procedure. (1)
The use of ultrasound guidance and automated biopsy gun provide a low risk of complications such as pain, bleeding, or a small hematoma. Major complications, including the need for nephrectomy or death, are extremely rare. (2)
Potential risk factors for bleeding complications are the female sex, elevated blood pressure, disturbed hemostasis and low hemoglobin level before intervention. The risk of bleeding appears to be lower for transplant than for native kidney biopsy. (2, 3)
Renal artery embolization is a minimally invasive procedure that is increasingly being used for treatment of a wide range of conditions. It was first introduced in the 1970s. The main indications include: prenephrectomy and preradiofrequency ablation infarction of renal tumors, management of renal angiomyolipomas and renal hemorrhage (life-threatening or chronic debilitating hematuria). It has shown good results in post-biopsy complications.(4)
The rate of major and minor complications has been reported around 10%, the most frequent being renal insufficiency and unintentional embolization of non-target areas. (5)
Study design
A retrospective observational study was performed with biopsied patients from the Hospital Civil of Guadalajara at the Radiology department. Preservation of renal function in patients who required invasive treatment was evaluated by performing renal gammagraphy, ultrasound test (US) and simple phase renal computed tomography (CT). Descriptive statistics, central tendency measures for frequencies and quantitative variables and percentages for qualitative variables were obtained.
Study Population
From January 1st, 2015 to January 1st, 2020, 430 renal biopsies were performed in patients with ages between 7 months and 79 years. 210 were renal graft and 220 were native kidney biopsies. There was a male predominance, 58% of the treated subjects were men and 42% were women. The most common diagnosis for performing the biopsy in renal graft was serum creatinine elevation (25%). The most common diagnosis for native kidney biopsy was proteinuria (56%).
Inclusion criteria
Hemoglobin test, platelet count, prothrombin time (PT) and partial thromboplastin time (PTT) in normal ranges were necessary for performing the biopsy.
Antithrombotic drugs were suspended 48 hours before the procedure,
systolic blood pressure could not exceed 150 mmHg.
Clinical data was also collected, including gender, age (years), history of hypertension, acute or chronic renal disease and proteinuria.
Body weight and height were collected in order to decide the size of the biopsy needle to be used.
Biopsy technique
Procedure was (US) ultrasonography-guided using an automatic tru-cut biopsy gun with a 16-18 Gauge (Ga) needle. (FIG. 1)
For each procedure 1 or 2 samples were obtained. All samples were checked by the same nephropathologist. Ten minutes manual compression was performed after each biopsy. A sonography test and a blood count were performed 4-8 hours later in order to discard complications.
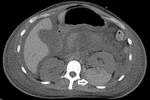
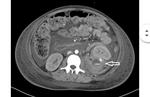
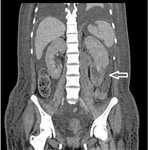
During sonography test four patients (1.1%) showed with renal hematoma and hemoglobin descent (2 or more grams compared to pre-procedure test). Contrast-enhanced CT was performed after US evaluation in these patients to have a better spacial resolution of imagenologic findings. (FIG. 2, 3, 4)
One was controlled by using IV etamsylate (250 mg every 8 hours), IV vitamin K (50 mg every 8 hours) and desmopresin (15 µg single dose).
The other three (0.69%) required endovascular approach performed within the first 12 hours post-procedure.
It drew attention that all four patients who presented with complications were young women with a pre-biopsy diagnosis of systemic lupus erythematosus (SLE). The four biopsies were performed with a 16 Gauge tru-cut needle.
Superselective embolization of the bleeding artery was performed preserving the rest of the circulation. Gel foam was used as the embolic agent in the three cases.
Embolization technique
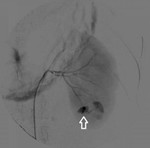
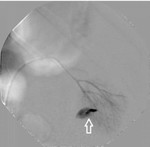
Procedure was performed under fluoroscopic guidance. Femoral approach was obtained using a 5 French arterial introducer, after that a 0.035 in x 150 cm hydrophilic curved end guidewire was advanced until reaching the main renal artery. A 5 French hydrophilic Cobra catheter was advanced until this position, performing an arteriography to localize the bleeding branches. (FIG. 5)
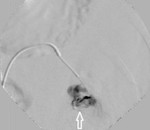
Once localized the site of bleeding, a superselective canalization of these branches was performed by advancing a 2.9 French microcatheter to the desired position.(FIG. 6)
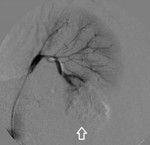
Contrast medium was injected to corroborate microcatheter’s position at the bleeding site. After that, non-ionic contrast medium was mixed with gel foam as embolic agent and injected through the microcatheter. (FIG. 7) Another arteriography was performed to ensure embolization of the desired branches was complete.
A final arteriography at the main renal artery is made in order to corroborate no embolic agent was mistakenly migrated into the rest of the renal circulation. (FIG. 8)
There were no immediate complications.