Keywords:
Breast, MR, Treatment effects, Cancer, Molecular, genomics and proteomics, Multidisciplinary cancer care
Authors:
I. Mendoza Arnau, M. D. Rabadán Caravaca, M. Culiañez Casas, S. Martínez Meca; Granada/ES
DOI:
10.26044/ecr2021/C-10320
Methods and materials
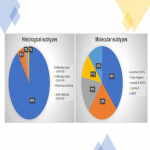
We have performed a descriptive and retrospective study including 103 women with breast cancer who were candidates for treatment with neoadjuvant chemotherapy in our hospital between October 2017 and August 2019.
Breast MRI was performed for staging and 15 days after systemic neoadjuvant treatment. All patients underwent surgery after neoadjuvant treatment and histologic examination of the surgical piece was performed.
The information about each procedure was communicated to every patient and they signed the informed consent.
Breast MRI was performed on a 1.5 T equipment (Signa, GE) by using a breast-specific antenna with the patient placed in the prone position. MRI protocol included FSE-T1WI, STIR, DWI and VIBRANT dynamic study without contrast and after the administration of intravenous Gadobutrol (Gadovist 1mmol/ml) 0.1mml/kg, 2ml/s, followed by 20ml of saline in an antecubital or dorsal metacarpal vein, with acquisition in six phases.
Exclusion criteria: patients with claustrophobia, pacemakers and metal implants, hidden breast tumors, lack of clinical response or signs of progression and those who did not complete neoadjuvant treatment.
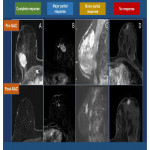
Radiological response evaluation: breast MRI was analyzed by 2 radiologists from the Breast Unit of our center. Morphological and dynamic uptake patterns were defined according to the BI-RADS classification. Radiological response was classified in four categories according to the Union for International Cancer Control (UICC) (Fig. 1):
- No response. No changes in tumor size.
- Partial minor response. Tumor size decreases less than 50% (major diameter)
- Partial major response. Tumor size decreases more than 50% (major diameter)
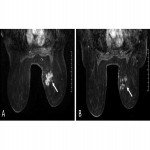
- Complete response. Absence of tumor and pathological enhancement (Fig. 2)
Additionally the following items were assessed:
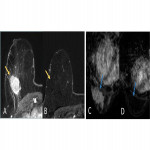
- Type of morphological response: concentric, fragmented and mixed (Fig. 3,4,5)
- Dynamic study. Intensity / time curve
- Complete radiological response: absence of enhancement.
- Late enhancement.
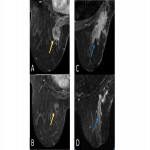
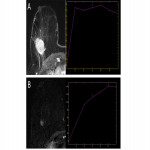
- Progressive (type 1 curve) (Fig. 6A)
- Plateau (type 2 curve) (Fig. 6B)
- Washout (curve type 3)
Pathologic response evaluation. Miller and Payne (MP) grading system:
- Grade 1: Minimal cellular changes, without significant reduction in invasive cellularity.
- Grade 2: Discrete reduction in invasive tumor cellularity, less than 30% of the tumor mass.
- Grade 3: Marked reduction in invasive tumor cellularity, between 30% and 90% of the tumor mass.
- Grade 4: Decrease of more than 90% of the tumor mass, with persistence of microscopic foci of invasive carcinoma.
- Grade 5: Absence of invasive tumor cells.
Persistence of in situ carcinoma in the absence of an invasive component was considered as a complete response.
Correlation between radiological and pathologic response was established:
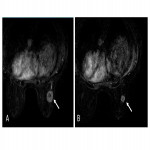
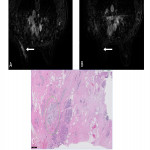
- Complete response was equivalent to Miller and Payne (MP) grade 5 (Fig. 7)
- Major partial response was equivalent to MP grade 4.
- Minor partial response was equivalent to MP grade 2 and 3.
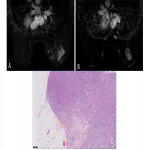
- No response was equivalent to MP grade 1 (Fig. 8)