Keywords:
Molecular imaging, Nuclear medicine, Oncology, PET, PET-CT, Diagnostic procedure, Staging, Cancer
Authors:
A. Gaber, R. Núñez, E. Delpassand; HOUSTON, TX/US
DOI:
10.26044/ecr2021/C-10556
Methods and materials
110 subjects with histopathology confirmed prostatic adenocarcinoma were enrolled in this prospective study. Gleason score ranged from 6(3+3) to 10(5+5). 8 subjects had a Gleason score of 3+3(7.3%), 22 subjects had a Gleason score of 3+4(20%), 25 subjects had a Gleason score of 4+3(22.7%), 26 subjects had a Gleason score of 4+4(23.6%), 18 subjects had a Gleason score of 4+5(16.4%), 8 subjects had a Gleason score of 5+4(7.2), 2 subjects had a Gleason score of 5+5(1.8%) and 1 subject with undefined Gleason score (0.9%). The median age of all enrolled subjects was 68.1years (range: 52 to 88years).
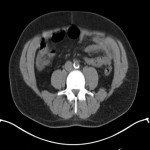
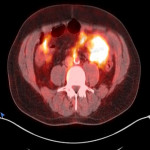
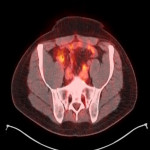
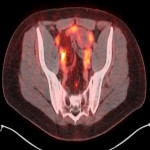
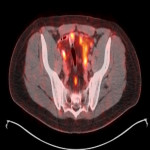
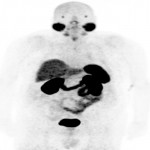
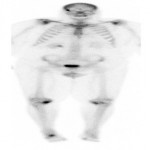
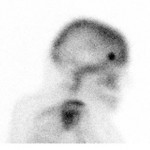
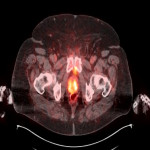
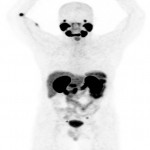
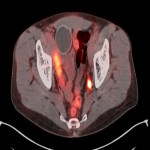
PSA ranging from 0.06ng/ml to 414.5ng/ml (average 13.2ng/ml). All subjects received 3 to 7mCi of 68Ga-PSMA-11 (average 4.9mCi). PET/ CT imaging was performed 60±15 minutes post-injection. For subjects with a PSA less than 2ng/ml, an additional late scan at 180 minutes post-injection was also done. PET/CT images were read by an experienced nuclear medicine physician aware of clinical data. Evaluation of the results of the 68Ga-PSMA-11 PET/CT scan was compared to other available conventional anatomical and functional imaging modalities.
This study received FDA authorization and approval from the Biomedical Research Alliance of New York Institutional Review Board (BRANY IRB).