1-Pancreatic laboratory tests and clinical approach in specific contexts:
1.1-Acute pancreatitis (AcP)
:
1.1.1.Symptoms:
- Epigastric and periumbilical pain; may radiate to the back, chest, flanks, and lower abdomen.
- Nausea, vomiting, and abdominal distention.
- Distress and anxiety.
- Fever, tachycardia, and hypotension.
1.1.2.Physical Examination:
- Abdominal tenderness.
- Jaundice (infrequent): edema of the head of the pancreas or biliary stones/sludge.
- Basilar rales, atelectasis, pleural effusion (left-sided).
- Erythematous skin nodules.
- Diminished bowel sounds.
- Enlarged pancreas palpable.
- Cullen’s sign: blue discoloration around umbilicus in hemoperitoneum.
- Turner’s sign: blue or green-brown discoloration of the flanks in severe necrotizing pancreatitis.
1.1.3.Laboratory:
- Lipase: high specificity and sensibility. Peaks at 24 hours; elevated for 8–14 days. Enzyme of choice for diagnosis of AcP. False negatives (FN): chronic pancreatitis, hypertriglyceridemia. False positives: renal insufficiency.
- Amylase: less specific. Rise within 3-6 hours; elevated for 5 days.
- Leukocytosis.
- Hyperglycemia (B-cell injury)
- Alkaline phosphatase, aminotransferases, hyperbilirubinemia: ALT >150 U/L suggests gallstone pancreatitis and a more fulminant disease course.
- Hypertriglyceridemia: etiology.
- Ca 2+: hypocalcemia -> saponification of fats in the retroperitoneum; hypercalcemia -> etiology.
1.1.4.Markers of severity:
- C-reactive protein: high levels 24-48 hours after presentation correlate with organ failure. ≥ 10 mg/dL indicates severe pancreatitis.
- Hematocrit>44%: severe disease
- Blood urea nitrogen>22 mg/dL: mortality risk factor
1.2- Chronic pancreatitis (CP)
1.2.1.Symptoms:
- Abdominal pain
- Steatorrhea
- Weight loss
- Bloating
1.2.2.Physical exam:
- Abdominal tenderness.
- Pancreas can be palpated if pseudocysts are present.
- Splenomegaly (splenic vein thrombosis).
- Erythema ab igne (rare but typical).
- Signs of nicotine abuse, alcohol abuse, or malnutrition.
1.2.3.Laboratory:
- Subclinical inflammation -> markers of inflammation will not be elevated.
- Elevated pancreatic enzymes in patients with abdominal pain have low specificity for CP.
- Markers of malnutrition in pancreatic exocrine insufficiency (PEI): prealbumin, hemoglobulin, vitamin D, E, Magnesium e Zinc
1.2.4.Complications:
- PEI: pancreatic function testing. Fecal elastase-1: <200 ug/g - confirms the diagnosis of any sort of CP.
- Endocrine insufficiency: hyperglycemia
- Pseudoaneurysm rupture: Hemoglobin/hematocrit
- Biliary obstruction: alkaline phosphatase/bilirubin
1.3-Pancreatic carcinoma
1.3.1.Tumour markers:
CA19-9:
- Elevated in 75-85% of pancreatic carcinomas.
- In the absence of biliary obstruction, liver disease, or benign pancreatic disease, CA 19-9 >100 U/mL is highly specific for malignancy.
- 5-10% lack the enzyme to produce CA 19-9 so monitoring disease with this marker will not be possible.
- Clinical applications are described in
.
CEA
- 40-45% of patients with pancreatic carcinoma have elevated levels.
- Benign and malignant conditions lead to elevated levels: CEA is not sensitive or specific for pancreatic cancer.
1.4–Imaging:
1.4.1-US in AcP: identify gallstones or biliary ductal dilatation. In the absence of another etiology, the presence of cholelithiasis or sludge in the gallbladder on US in a patient with AcP is diagnostic of biliary pancreatitis.
1.4.2-CT with contrast: evaluation of equivocal presentations of AcP with identification of necrosis or other etiologies of abdominal pain.
- Early phase: necrosis may not be reliably demonstrated <5-7 days. Even if collections or necrosis are present, no interventions are pursued in this phase.
- Late phase: >7 days; Imaging for assessing severity and complications, guiding treatments, and monitoring treatment response.
1.4.3.Complications:
- Infection: abdominal CT if suspected.
- Pseudoaneurysm: CT with IV contrast (arterial and venous phase).
- >4 weeks: Pseudocyst- well-defined wall; doesn’t contain solid material. Drained by simple percutaneous or endoscopic techniques. Walled-off necrosis- well-defined wall; contains necrotic debris that may lead to secondary infection. Requires surgical debridement, endoscopic necrosectomy, cyst-gastrostomy stents, or percutaneous irrigation and drainage.
2 -Pseudopancreatic lesions (
)
2.1.1-Pancreatic
Variations of pancreatic contour: healthy tissue with unusual contour, mimicking pancreatic neoplasms. Key: attenuation identical to pancreas on all phases.
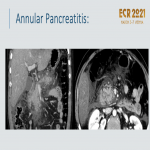
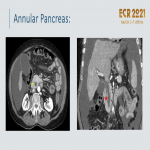
Annular pancreas:
- Embryological migration failure producing a band of pancreatic tissue surrounding the duodenum, in continuity with the pancreas.
- 74%: D2 and 21%: D1 and D3
- Asymptomatic or associated with duodenal stenosis, postbulbar ulcerations, pancreatitis
, or biliary obstruction
.
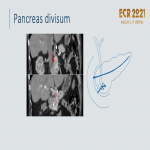
Pancreas divisum
- The most common congenital variant of the pancreas (10%).
- Failure of dorsal and ventral pancreatic anlage to fuse
.
- Asymptomatic (more frequent) or recurrent episodes of pancreatitis.
- May cause enlargement of the pancreatic head and be mistaken for a mass.
Variations of the course (descending, sigmoid, vertical, loop-shaped) or configuration of the pancreatic duct (bifid with dominant duct of Wirsung, dominant duct of Santorini without divisum, absent duct of Santorini, ansa pancreatic)
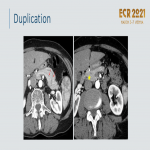
Duplication anomalies
.
Ectopic pancreas and accessory lobe.
2.1.2.Spleen
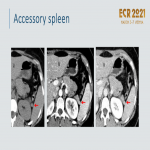
Accessory spleen: congenital foci of splenic tissue separated from the main spleen. Well-marginated, homogeneously enhancing round masses < 2 cm (Figure 8).
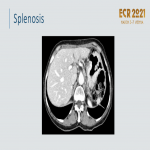
Splenosis: seeding of splenic cells after splenectomy or splenic rupture
. Typically adjacent to small-bowel serosa, greater omentum, parietal peritoneum, and diaphragm.
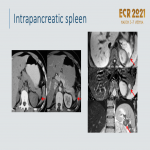
Intrapancreatic accessory spleen: nuclear scintigraphy with 99mTc-labeled heat-damaged RBCs->most specific imaging method. Inferior anatomic resolution -> increasing likelihood of misdiagnosis. Similar to the spleen on pre and contrast-enhanced images
-
inhomogeneous enhancement in early phases may be a diagnostic clue.
2.1.3.Duodenum
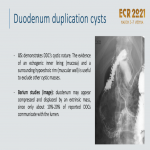
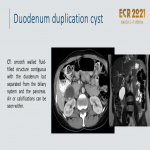
Duodenal Duplication Cyst:
- Noncommunicating spherical cysts adjacent to the wall of D1/D2
.
- Asymptomatic or pain, bowel obstruction, hemorrhage, jaundice, and pancreatitis.
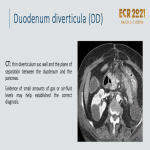
Duodenum Diverticula:
- In D2/D3, within 2 cm of the ampulla of Vater. Can be confused with cystic pancreatic neoplasms
.
- Intraluminal diverticula may show the classic sign “windsock” at barium examination.
- Mostly asymptomatic.
2.1.4.Biliary
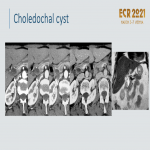
Choledochal cysts:
- Congenital anomalies.
- Cystic or fusiform dilatations of the biliary tree; may simulate a cystic mass.
- ERCP, MRCP, or CT can confirm the diagnosis non-invasively
.
2.2.Inflammatory/Infectious
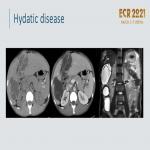
As an exocrine and endocrine gland lying at the crossroads of the enteric and biliary tracts, the pancreas is susceptible to unique inflammatory and infectious diseases that may mimic malignancy often with surgical resection such as chronic, autoimmune, and groove pancreatitis. Although exceptionally rare, pancreatic parasitic cysts have been described
.
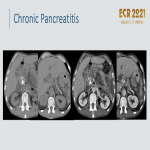
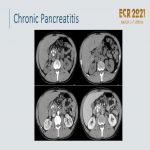
Chronic pancreatitis:
- Progressive, irreversible, fibrosing disease.
- Dilatation of the pancreatic duct, pancreatic calcifications
, and parenchymal atrophy. CP can present as focal enlargement of the gland due to a chronic inflammatory mass, often in the head, simulating adenocarcinoma
. Local lymphadenopathy and vessel involvement raise concern for malignancy
.
- Both can appear a hypodense masses with associated dilatation of CBD, main pancreatic duct strictures, infiltration of the adjacent fat, arterial encasement, and peripancreatic venous obstruction.
- Adenocarcinoma can be superimposed on patients with CP therefore specific findings of CP may not be sufficient to exclude malignancy without biopsy.
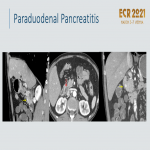
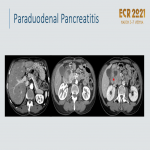
Paraduodenal pancreatitis:
Uncommon type of focal CP affecting the groove between the pancreas, duodenum, and CBD
. Hypodense lesion with delayed contrast enhancement due to fibrosis
. Pancreatic parenchyma is slightly compromised, and peripancreatic fluid is rare. Smooth tapering of the distal CBD, in contrast to abrupt obstruction seen in patients with pancreatic carcinoma. There can be imaging overlap between the two entities and biopsy is often indicated. Clinical and laboratory key features are described in
.
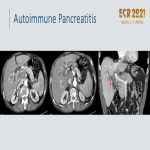
Autoimmune pancreatitis:
- Diffuse (“sausage pancreas”), focal, commonly in the head
, or multifocal.
- CT key findings and diagnostic criteria in
.
- Clinic: painless jaundice (CBD narrowing, raising suspicion of malignancy); weight loss. Absence of a history of acute pancreatitis or alcoholism.
- Laboratory: ANA, γ-globulin or IgG.
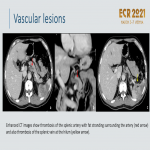
2.3–Vascular
- Pseudoaneurysms secondary to pancreatitis may mimic a mass, especially if the lumen is thrombosed
. Pseudoaneurysms complicate 10% of CP, most commonly the splenic artery.
2.4 - Miscellaneous
2.4.1.Lymphadenopathy:
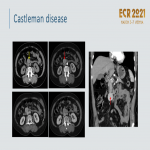
Lymphadenopathy in various disorders may mimic pancreatic lesions. These include lymphoma; metastatic disease; granulomatous disorders; angioproliferative disorders
; tuberculosis and peripancreatic lymph nodes.
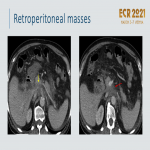
2.4.2 Retroperitoneal masses
Retroperitoneal processes such as hematoma, fibrosis, or neoplasms may mimic a pancreatic mass
.
Some mesenteric processes (retractile mesenteritis) or masses (carcinoid, desmoid, sclerosing mesenteritis) may pose diagnostic difficulty unless a clear plane of separation is identified.