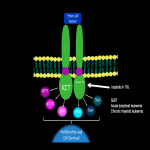
There are two main types of treatment, tyrosine kinase inhibitors (TKI) that are named with the IB suffix (i.e. erlotinib and imatinib) and monoclonal antibodies (MAB) that end with the AB suffix (i.e. bevacizumab and trastuzumab).
TKI have higher toxicity than monoclonal antibodies, given the ubiquitous location of tyrosine kinases in the body.
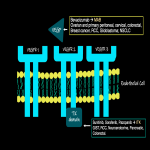
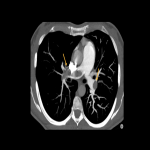
There have been 10 mechanisms described that allow cancer to survive (Fig1), each of them having a potential role as a therapeutic target. We will review the 3 main mechanisms, considering they share some AE and imaging findings, being:
- Induction of angiogenesis
- Maintain proliferative signal.
- Evasion of the immune response.
- Induction of Angiogenesis
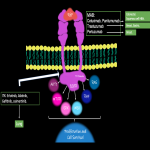
Non-tumor cells temporarily activate angiogenesis as part of healing processes; contrasting from tumor cells which constantly activate angiogenesis through signaling pathways, such as the Vascular Endothelial Growth Factor Receptor (VEGFR) pathway (Fig 2).
- Targeted Therapy (Fig 2): Inhibition of VEGF can diminish the renewal of endothelial cells which leads to endothelial damage.
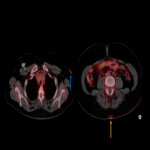
a. Arterial Hypertension (AHT): It is the most frequent AE, with a frequency up to 30%. It occurs because VEGF has an important role regulating vessels, by inducing arterial relaxation, lowers blood pressure. Also, VEFG induces phosphorylation of nitric oxide synthetase, increasing the production of nitric oxide, which acts directly on the endothelium. Therefore, when VEGF is blocked we can expect vasoconstriction, and increased blood pressure.
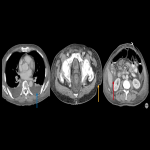
Although this effects are impossible to visualize using current imaging methods, we can detect some of its associated complications.
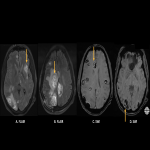
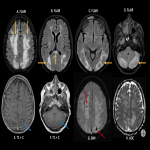
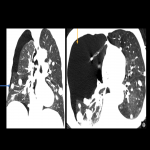
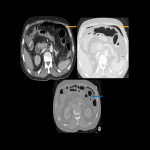
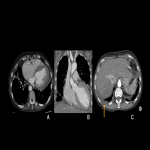
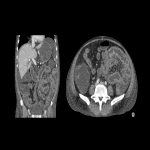
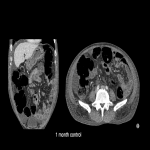
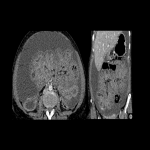
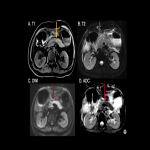
The most common imaging findings are intratumoral hemorrhage, which may be frequently found in brain metastases (Fig 3), and Posterior Reversible Encephalopathy (Fig 4).
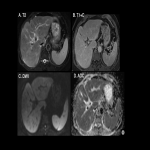
B. Hepatic steatosis: very common AE of antiangiogenic therapies (Fig 5).
C. Thrombosis: occurs due to endothelial damage exposing subendothelial collagen and activating tissue factor.
The most common manifestation is Pulmonary Embolism (Fig 6), but it can also occur in the brain, aorta, mesenteric arteries, pelvis, and extremities.
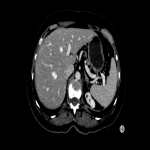
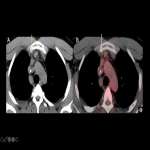
d. Pneumothorax: More frequent in patients with pulmonary metastases (Fig 7). Its important to recognize it as an adverse effect related to treatment because warrants discontinuation of therapy and prompt pneumothorax correction.
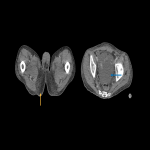
e. Cavitations in pulmonary metastases (Fig 8): Nodules can increase in size as result of targeted therapy, and should not be confused with progression. In some cases, cavitation is associated with better response to treatment.
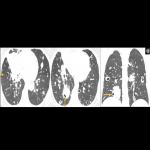
f. Gastrointestinal pneumatosis (Fig 9): Due to the reduction of the intestinal villi capillaries, causing micro perforation and allowing the infiltration of gas in the intestinal wall. It can occur up to 13 months after beginning of treatment.
70% are asymptomatic.
Unless there is perforation, expectant management is indicated.
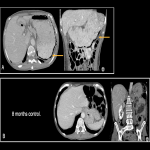
g. Intestinal perforation (Fig 10): In about 4% of patients. Changes in microvascularization cause ischemia and impaired healing, especially after surgery, pre-existing ulcers, diverticulitis, or necrosis of tumor deposits in the serosa. Symptoms are nonspecific and may even be absent.
h. Fistulas (Fig 11): Frequency up to 2%. Can result as a complication from perforation or at other sites, e.g. intestine, bladder, vagina, uterus, gallbladder or skin.
- Maintain proliferative signal:
Cancer cells can proliferate in a chronic and non-selective way (Fig 12); sustained by the production of growth factor ligands that signal normal cells to express growth factors. Compared to normal cells, cancer cells have more surface receptors, making them sensitive to growth factor ligand.
Pathways:
- Epidermal Growth Factor Receptor (EGFR) (Fig 13): Key survival mechanism in lung cancer, found mutated in up to 25% of patients. Targeted Therapy (Fig 13): MABs: Cetuximab, Trastuzumab. TKIs: Erlotinib, Afatinib.
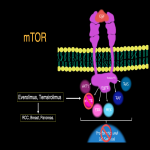
- mTOR (Fig 14). Targeted Therapy (Fig 14): Everolimus and Temsirolimus.
- KIT receptor (Fig 15). Mutation in 55% of GISTs. Targeted Therapy (Fig 15): Imatinib.
TIKs for EGFR and mTOR pathway: The most frequent toxicities are dermatological, colitis and pneumonitis. (We will review dermatological toxicities and colitis in the next mechanism of action, since they share imaging findings)
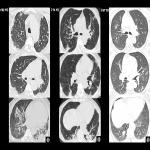
a. Pneumonitis (Fig 16): More frequent in patients with mTOR inhibitors (30%) compared to anti EGFR (5%). Dyspnea, with or without cough, in up to 50%.
Mechanism for pulmonary toxicity is not clear, but it is suggested that repair of epithelial cells requires pneumocytes mediated by activation of EGFR which is inhibited in these therapies, inducing these AEs and corresponding imaging findings.
Anti mTOR: 1. GGO, 2. GGO and reticular opacities, 3. GGO and consolidations, 4. GGO, reticular opacities and consolidations.
Anti EGFR: 1. GGO, 2. Organizing pneumonia pattern with multifocal consolidations, 3. Acute eosinophilic pneumonia, 4. Extensive GGO with consolidations and traction bronchiectasis that may represent acute interstitial pneumonia or diffuse alveolar damage.
MABs for EGFR pathway:
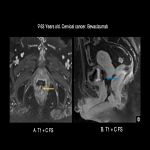
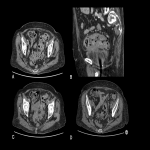
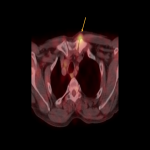
b. Cardiotoxicity (Fig 17): Usually reversible, unlike cardiotoxicity secondary to anthracyclines, thus the importance of early recognition. Can be expected in up to 14% of cases, with imaging findings like cardiomegaly, pleural effusion, interlobular septal thickening.
AntiKIT pathway:
c. Fluid retention (Fig. 18, 19): Imaging findings are ascites, pleural effusion, pericardial effusion, pulmonary edema and anasarca. They can reach congestive heart failure in up to 16% of patients, however, we should differentiate from tumor progression.
- Immune Destruction Evasion:
Cancer cells have antigens that stimulate cellular immunity (T cell mediated) and are usually controlled by natural killer cells. However, tumor cells can evade immune recognition by activating inhibitory pathways called "immune checkpoints" (Fig 20).
- Immunotherapy: It stimulates the immune system to act against cancer cells. These therapies are monoclonal antibodies (Fig 20).
This toxicity is known as immune-mediated adverse effects and its due to autoimmune reactions, therefore, they can affect any organ of the body.
a. Skin toxicity: One the most common AE, in up to 95% of patients (Fig 21).
b. Colitis: Estimated frequency of 46% with a mortality rate of 5%. Not associated with fever or blood test abnormality. Two imaging patterns of colitis:
- Segmental associated with diverticulosis: Compromising a segment of pre-existing diverticulosis. Segmental thickening of the walls, and pericolonic fat stranding (Fig 22).
- Diffuse: Dilation of mesenteric vessels, fluid-filled colon, diffuse or segmental mild thickening of the intestinal wall (Fig 23).
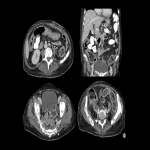
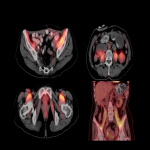
b. Enteritis: Diffuse intestinal wall thickening, with mesenteric fat stranding and ascites (Fig 24-26).
c. Hepatitis: Usually asymptomatic but may present with fever, fatigue, jaundice, and change in stool color. Images are nonspecific and can show mild hepatomegaly, periportal and gallbladder edema, diffuse hypoattenuation or heterogeneous enhancement of liver parenchyma (Fig 27).
d. Pancreatitis: Less than 1%. Can be asymptomatic or with typical symptoms and elevated lipase levels. Imaging findings include focal or diffuse pancreatic enlargement, with hypo enhancing areas and peripancreatic fat stranding (Fig 28).
e. Pneumonitis: Bilateral consolidations and GGO in a peripheral distribution that mimics cryptogenic organized pneumonia or with peripheral basal subpleural GGO and interlobular septal thickening that mimics nonspecific interstitial pneumonia.
f. Sarcoid-like granulomatous reaction: New or enlarged nodes and coexisting pulmonary changes ranging from irregular nodular and parenchymal opacities to GGO or bilateral peripheral interstitial opacities (Fig 29).
g. Endocrine organs: Hypophysitis (1-6%), Thyroiditis (2%), with or without associated hypophysitis, Adrenalitis (3-6%).
h. Musculoskeletal: Patients can refer arthralgia and myalgia. Imaging findings include inflammatory arthritis up to 12% and myositis (Fig 30, 31).
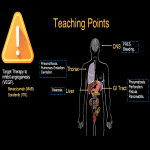
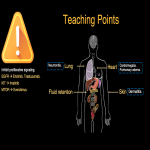
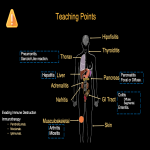
Teaching points are summarized in Fig 32, 33, 34.