Purpose
Approximately 20% of patients with pancreatic ductal adenocarcinoma (PDAC) have resectable or borderline resectable disease, 40% of patients presents with locally advanced disease and 40% with metastatic disease.[1] Neoadjuvant chemotherapy (NAC) is increasingly used in treatment of potential resectable PDAC, but only 40% of the patients undergo resection after neoadjuvant treatment. [2] Response rates for palliative chemotherapy range up to 30%.[3]
Current restaging after chemotherapy using conventional contrast-enhanced CT (CECT) is insufficient for reliable response assessment with Response Evaluation Criteria in Solid Tumors (RECIST). Change...
Methods and materials
We present initial data from a prospective study in 12 patients (7 men and 5 women, 47-74 years) with biopsy proven PDAC, treated with chemotherapy between January 2018 and December 2019 (neoadjuvant n=9, palliative n=3). CTP was performed at baseline and follow-up after 3 months.
All patients were scanned on a 320-row detector CT scanner with 160 mm coverage. The amount of contrast agent (400 mg I/ml) is based on body weight (1,3 mg I/kg) with a fixed duration of the injection time of 5...
Results
Using RECIST-criteria patients were classified as 4 responders and 8 non-responders.
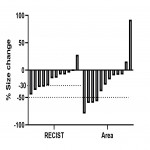
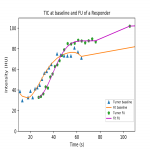
Mean blood flow at baseline was 32,7 ml/100g/min (±6,5) in responders and 32,4 ml/100g/min (±12,8) in non-responders (p=0,972). At follow-up BF increased with 25% (±37%) in responders and 0,5% (±33%) in non-responders (p=0,269). Maximum enhancement increased for responders with 45,5% (±36%) and decreased for non-responders with 2,5% (±22%) (p=0,017).
Using area of tumor ROI, patients were classified as 4 responders and 8 non-responders, with however a different distribution compared to the RECIST criteria. Responders...
Conclusion
The major finding in this study was that percentual change in blood flow was higher in patients who responded to chemotherapy.
A possible explanation for increased blood flow is that treatment slows down extracellular matrix accumulation in the tumor, allowing improved perfusion.[6]
After neoadjuvant chemotherapy current staging tools (RECIST) often underestimate treatment response. More accurate restaging can improve selecting patients for R0-resection (resection margin free of tumor) and prevent futile surgery. Earlier response assessment for palliative chemotherapy could help in preventing unnecessary toxicity for non-responders,...
Personal information and conflict of interest
T. H. Perik:
Nothing to disclose
E. Van Geenen:
Nothing to disclose
E. J. Smit:
Nothing to disclose
L. A. A. Brosens:
Nothing to disclose
M. W. J. Stommel:
Nothing to disclose
E. C. Gootjes:
Nothing to disclose
K. J. H. M. Van Laarhoven:
Nothing to disclose
H. Huisman:
Nothing to disclose
J. J. Hermans:
Nothing to disclose
References
[1] C. G. Willett, B. G. Czito, J. C. Bendell, and D. P. Ryan, “Locally advanced pancreatic cancer,” J. Clin. Oncol., vol. 23, no. 20, pp. 4538–4544, 2005, doi: 10.1200/JCO.2005.23.911.
[2] F. Petrelli et al., “FOLFIRINOX-based neoadjuvant therapy in borderline resectable or unresectable pancreatic cancer: A meta-analytical review of published studies,” Pancreas, vol. 44, no. 4, pp. 515–521, 2015, doi: 10.1097/MPA.0000000000000314.
[3] T. Conroy et al., “FOLFIRINOX versus Gemcitabine for Metastatic Pancreatic Cancer,” N. Engl. J. Med., vol. 364, no. 19, pp. 1817–1825, 2011, doi:...


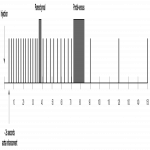


![Fig 6: Percentual change in blood flow (BF) after chemotherapy in responders [R] and non-responders [NR]. Categorized by the RECIST-criteria (p=0,972) and Area (p=0,024).](https://epos.myesr.org/posterimage/esr/ecr2021/158239/media/901820?maxheight=150&maxwidth=150)
![Fig 7: Percentual change in maximum enhancement after chemotherapy in responders [R] and non-responders [NR].](https://epos.myesr.org/posterimage/esr/ecr2021/158239/media/901821?maxheight=150&maxwidth=150)
