Keywords:
Abdomen, Contrast agents, Pancreas, CT-Quantitative, Experimental investigations, Treatment effects, Cancer
Authors:
T. H. Perik, E. Van Geenen, E. J. Smit, L. A. A. Brosens, M. W. J. Stommel, E. C. Gootjes, K. J. H. M. Van Laarhoven, H. Huisman, J. J. Hermans; Nijmegen/NL
DOI:
10.26044/ecr2021/C-14092
Purpose
Approximately 20% of patients with pancreatic ductal adenocarcinoma (PDAC) have resectable or borderline resectable disease, 40% of patients presents with locally advanced disease and 40% with metastatic disease.[1] Neoadjuvant chemotherapy (NAC) is increasingly used in treatment of potential resectable PDAC, but only 40% of the patients undergo resection after neoadjuvant treatment. [2] Response rates for palliative chemotherapy range up to 30%.[3]
Current restaging after chemotherapy using conventional contrast-enhanced CT (CECT) is insufficient for reliable response assessment with Response Evaluation Criteria in Solid Tumors (RECIST). Change in tumor size has a poor correlation with histopathological grading of response, with RECIST underestimating the response.[4-5] We evaluated CT perfusion (CTP) as a functional biomarker for chemotherapy response in patients with pancreatic ductal adenocarcinoma (PDAC).



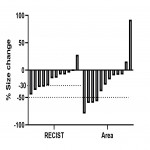

![Fig 6: Percentual change in blood flow (BF) after chemotherapy in responders [R] and non-responders [NR]. Categorized by the RECIST-criteria (p=0,972) and Area (p=0,024).](https://epos.myesr.org/posterimage/esr/ecr2021/158239/media/901820?maxheight=150&maxwidth=150)
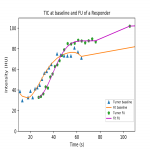
![Fig 7: Percentual change in maximum enhancement after chemotherapy in responders [R] and non-responders [NR].](https://epos.myesr.org/posterimage/esr/ecr2021/158239/media/901821?maxheight=150&maxwidth=150)
