Type:
Educational Exhibit
Keywords:
Abdomen, CNS, Thorax, CT, MR, PET-CT, Complications, Toxicity
Authors:
S. LON COLVÉE, M. Guerrero Martin, �. Rueda De Eusebio, S. Gómez Peña, M. D. C. Polidura Arruga, N. Gomez Ruiz, P. Hernandez Mateo, I. Martin Lores
DOI:
10.26044/ecr2023/C-14856
Background
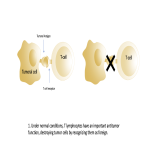
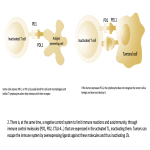
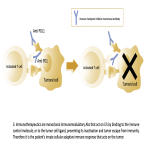
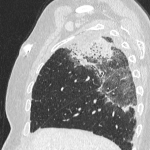
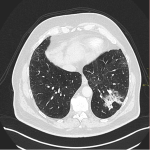
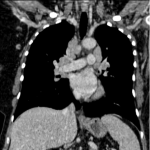
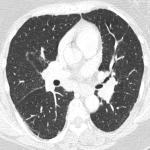
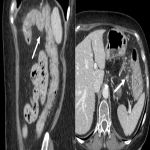
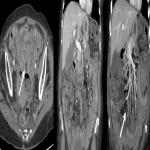
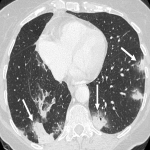
Immunotherapy potentiates the host's own adaptive immune system to stimulate an antitumor response. Tumor cells have created multiple mechanisms to evade destruction by the immune system such as disrupting effective antigen presentation, reducing efficacy of T-cell function and up-regulating pathways that promote tolerance and T-cell anergy. Immunotherapy attempts to disrupt the aforementioned mechanisms, and are grouped into different subtypes by their mechanisms of action. Checkpoint proteins are receptors expressed on the surface of T lymphocytes and help to down-regulate the immune system, when they are bound to certain ligands expressed on the antigen presenting cells of normal and cancerous tissue, T-cell-mediated death is evaded. Using this mechanism, tumors have evaded destruction by the immune system. A type of immunomodulatory monoclonal antibody, known as immune checkpoint inhibitors (ICI) are monoclonal antibodies which prevent the checkpoint protein and ligands from binding and thereby permitting T-cell-mediated death of tumor cells.
Immunotherapy has a very different mode of action compared to conventional cytotoxic chemotherapy. This contrast has resulted in distinct tumor responses requiring unique response criteria, and adverse event profile which will be the topic of this review.
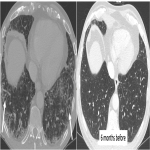
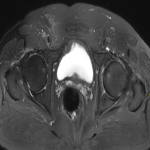
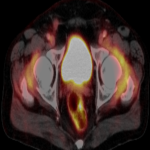
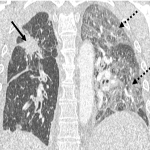
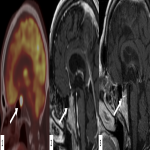
We selected a group of patients that underwent immunotherapy treatment in our institution from 2018 to 2022. We correlated the clinical features they referred to during emergency episodes or routine follow-up with CT, PET-CT and/or MRI imaging findings. Adverse effects were classified according to organ-specificity findings, the timing in their development after starting the treatment and the type of immunotherapeutic agents.