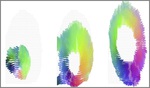1- Left ventricular modeling of the myocardium:
Ranjbar S.
et al.
(2013) recently developed the first novel left ventricular myocardial model mathematically based on echocardiography,
by MATLAB software and LSDYNA software in normal subjects,
which dynamic orientation contraction (through the cardiac cycle) of every individual myocardial fiber could be created by adding together the sequential steps of the multiple fragmented sectors of that fiber.
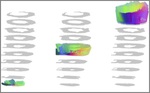
The left ventricular myocardial modeling of the heart shows that in normal cases myocardial fibers initiate from the posterior-basal region of the heart,
continues through the left ventricular free wall,
reaches the septum,
loops around the apex,
ascends,
and ends at the superior-anterior edge of left ventricle (Figures 6,
7,
8,
9,
10 and 11).
2-Achievability of the assessment of the mitral valve leaflets by mathematical equations of inelasticity
The mechanics of the mitral valve leaflet as a nonlinear,
inelastic and anisotropic soft tissue results from an integrated response of many mathematical/physical indexes' that illustrate the tissue.
In the past decade,
finite element modeling of complete heart valves has greatly aided evaluation of heart valve surgery,
design of bio-prosthetic valve replacements,
and general understanding of healthy and abnormal cardiac function.
Such a model must be based on an accurate description of the mechanical behavior of the valve material.
It is essential to calculate velocity/displacement and strain rate/strain at a component level,
that is to work at the cellular level.
In this study we developed the first three-dimensional displacement vectors field in the characterization of mitral valve leaflets in continuum equations of inelasticity framework based on echocardiography.
Echocardiography imaging of the mitral valve motion during the cardiac cycle of planes TTE in the short axis,
long axis and four chamber views,
and multi-planes TEE in the lower esophageal views at 0,10,15,30,
45-60,
75,
80,
90,
110-125 and 135 degrees were prospectively acquired for 200 healthy subjects of 65 time-frames from diastole to systole (early diastole,
mid diastole,
atrial systole,
end of diastole,
end of systole).
In each plane and for each frame during diastole to systole,
mitral valve annulus and leaflets,
and bases,
middles and tips of AML and PML,
were manually identified using MATLAB software.
3D MV annulus geometry,
leaflets measurements,
AML and PML positions were automatically computed for each frame,
and used as input to displacement vectors modeling of the mitral valve leaflets.
By solving mathematical equations of inelastic properties of leaflets,
each plane was replaced with 60 vectors of displacement and ultimately the mitral valve leaflets were realized by 1125 vectors of displacements where show also translations,
rotations and pure strains of bases,
middles and tips of AML and PML simultaneously per cardiac cycle.
Preliminary results concerning a different aspect of MVL biomechanics,
such as leaflets dynamics,
displacements/velocities and strain rates/strains of points on leaflets,
were in good agreement with in echocardiographic observations
(Figure 12 ).
3- Fluid dynamic inside the left ventricle by using 1,
2 in above:
The standard way to model the motion of blood inside the left ventricle would be to treat the left ventricle as an elastic membrane obeying Newton’s laws of motion with forces calculated in part from the elasticity of the membrane and in part by evaluating the fluid stress tensor on the surface of the membrane.
Then the fluid equations would have to be supplemented by the constraint that the velocity of the fluid on either side of the membrane must agree with the instantaneously known velocity of the elastic membrane itself.
There is a difficulty with this standard approach to the problem.
Challenge is the practical one of evaluating the fluid stress tensor on either side of the boundary.
This seems difficult (or at least messy) to do numerically,
unless the computational grid is aligned with the boundary.
On the other hand,
in a moving boundary problem,
it is both expensive and complicated to re-compute the grid at every time step in order to achieve alignment.
This means that the sum of the elastic force and the fluid force on any part of the boundary has to be zero.
Once we know this,
it becomes unnecessary to evaluate the fluid stress tensor at the boundary at all! We can find the force of any part of the boundary on the fluid by evaluating the elastic force on that part of the boundary.
(Note the use of Newton’s third law: the force of boundary on fluid is minus the force of fluid on boundary) All we need is a method for transferring the elastic force from the boundary to the fluid.
On a Cartesian grid,
this may be done by spreading each element of the boundary force out over nearby grid points.
The particular way that this is done in the boundary method involves a carefully constructed approximation to the Dirac delta function.
This force-spreading operation defines a field of force on the Cartesian lattice that is used for the fluid computation.
Then the fluid velocity is updated under the influence of that force field.
The Navier-Stokes solver that updates the fluid velocity does not know about the any consideration of the heart left ventricle geometry; it just works with a force field that happens to be zero everywhere except in the immediate of the vorticity region.
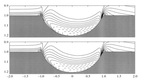
This approach can be used for instructional purposes and diagnosis of heart ailments.
The local fluid velocity at a point of the elastic boundary is evaluated by interpolation from the Cartesian grid.
The same approximate delta function that was used to spread force can also be used to get an interpolation operator that is the adjoint (or transpose) of the force-spreading operator.
In summary,
the boundary method avoids many of the difficulties and pitfalls of the standard approach to fluid-structure interaction.
By representing an elastic boundary in terms of the forces applied by the immersed elastic boundary to the fluid,
the boundary method avoids any assumption of boundary geometry in the fluid computation; makes it unnecessary to evaluate the fluid stress tensor at the elastic boundary; and makes it possible to simulate elastic boundaries,
like the chambers of the human heart (Figure 13).