Prevalence
Atrial Septal Defect (ASD) is the most common congenital heart defect to present in adulthood (1).
ASD accounts for 5% to 10% of all cases of congenital heart disease (2).
ASD can be caused by a defect of ostium secundum (80%),
ostium primum (10%),
unroofed coronary sinus (<1%) and sinus venosus (SVASD),
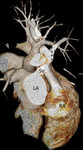
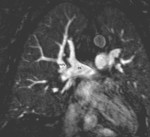
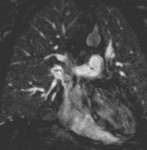
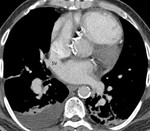
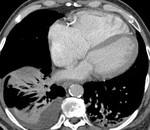
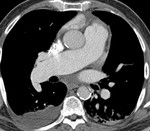
which is reported to represent approximately 2-10% of all cases of ASD (2) (Figure 2).
Embryology
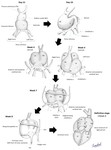
ASD can be classified in two types:
-
A) Direct communicatiom between the right and left atria: ostium primum and ostium secundum.
-
B) Does not involve the interatrial septum but physiologically behaves like interatrial septal defects: sinus venosus and unroofed coronary sinus (1).
Most of the atrial septal defects derive from a development alteration of the interatrial septum,
which is the structure that divides the primary atrium into the right and left atrial chambers.
On the other hand,
the exact embriology development of sinus venosus defect is controversial.
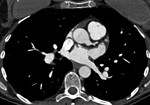
It is thought to result from the lack of septation between the pulmonary veins and the superior cava vein or right atrium (1) (Figure 3).
Anatomy
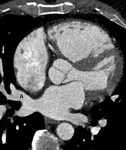
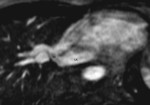
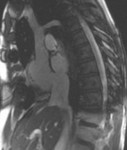
The are two types of SVASD: the superior and the inferior SVASD.
The superior form constitutes the majority and also the most well recognized type while the inferior SVASD,
also known as “Right atrial type SVD”,
is rare with only case reports and small case series published (as shown in Figure 21).
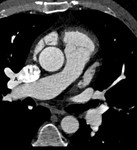
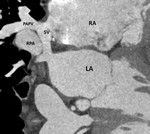
The anatomic alteration is localized: in case of superior SVASD,
there is a defect communicating the right atrium-SVC junction with the left atrium; in the case of inferior SVASD,
a communication exists between the right atrium-IVC junction with the left atrium (1,3,4).
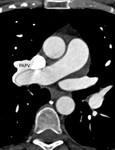
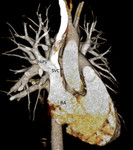
In the 80-95% of patients an associated anomalous right pulmonary venous drainage is evident (3-5); a partial anomalous pulmonary venous return (PAPVR) of the right upper lobe into the SVC in patients with superior SVASD,
and a PAPVR of the right lower lobe pulmonary vein into the pericardial segment of the IVC or right atrium in patients with inferior SVASD.
The pathognomonic feature of SVASD is the overriding of the intact rim of the fossa ovalis by the cava vein and the presence of a shunt at the atria level (4) (Figure 2).
Clinical behaviour
Although ASD represents the most common congenital heart defect presenting in adulthood,
it can be clinically silent for decades (1).
SVASDs produce a left-to-right shunt.
Long-standing shunting leads to dilatation of the right-sided chambers and enlargement of the pulmonary arteries.
Eisenmenger pathology may manifest due to the chronic overload of the right-sided chambers.
This right side overload leads to permanent pulmonary hypertension.
A pulmonary-to-systemic blood flow ratio (Qp:Qs ratio) of greater than 2:1 will likely be symptomatic and require surgical intervention by age 40.
When symptomatic,
clinical manifestations of SVASD are variable and nonspecific,
including dyspnea,
paradoxical embolism,
fatigue,
recurrent pulmonary infections,
arrhythmias,
palpitations and right chamber dilatation (1,6).
Imaging
-Transthoracic and transesophageal echocardiography:
Although transesophageal echography (TEE) has demonstrated good detection rates in the past (4,5),
a review by Kafka et al.
of 37 patients with right ventricle dilatation without a known cause by transtorathic ecography (TTE) or (TEE) revealed that nineteen of the patients were ultimately diagnosed with SVASD using MRI.
Additionally,
eighteen of them had anomalous pulmonary venous connections,
all of them right-sided (4).
Additionally,
although the detection rates by TTE may be less than 12% (5,6),
the American College of Radiology recommends TTE as the initial diagnostic imaging modality in adults with suspected or known cardiac congenital malformations.
The utility of TEE is limited by its invasive nature and by its inherent inability to image the cranial aspect of SVC,
where accessory right upper lobe pulmonary veins sometimes connect makes this modality less (7).
-Cardiac CT:
The complex anatomy of the heart may make differentiation of type,
size and extent of an ASD difficult.
The modern post-processing techniques of computed tomography angiography (CTA) are ideally suited for such complicated anatomy and pathology.
Compared with the centrally located secundum ASD,
the sinus venosus is located eccentrically,
either superior or inferiorly in the atrial septum at the sites of inflow of the superior vena cava or inferior vena cava,
respectively.
The superior sinus venosus ASD is far more common than the inferior type (2).
In cases of superior SVASDs,
cardiac CT images demonstrate a defect in the superior aspect of the interatrial septum at the level of the superior vena cava.
This defect may allow a communication between the left atrium and superior vena cava.
Chronically,
this defect may result in volume overload of the right heart chambers,
leading to enlargement of the right atrium,
the right ventricle and the pulmonary artery,
with development of pulmonary arterial hypertension.
On superior SVASDs,
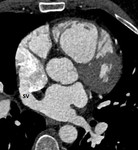
cardiac CTA imaging with a saline chaser can show concentrated contrast material pooling in the dependent left atrium,
a finding known as “fallen contrast” which is characteristic of a right-to-left shunt (1).
-Cardiac MR (CMR):
CMR is an excellent noninvasive imaging modality in the evaluation of SVASD.
CMR provides accurate information about the cardiac anatomy,
ventricular volumes and function,
and allows flow quantification measurements (7).
CMR may depict SVASD anatomy and associated anomalous pulmonary venous drainage,
provide quantitative information on the hemodynamic burden and may delineate the presence of additional cardiovascular abnormalities.
CMR may provide the surgeon with an excellent road-map for successful and uncomplicated surgical intervention.
CMR imaging techniques utilized for evaluation of SVASD may change in the future,
due to newer techniques and sequences.
Currently, a complete CMR protocol to diagnose a suspected SVASD may include: ECG-triggered,
breath hold gradient echo cine MR for assessment of SVASD and pulmonary venous anatomy and for quantitative evaluation of ventricular volumes and function; Gadolinium enhanced three dimensional magnetic resonance angiography with maximum intensity projection images; and flow velocity quantification in the ascending aorta and in the main pulmonary artery to calculate the pulmonary-to-sistemic flow ratio (Qp:Qs ratio) (7).
-Cardiac catheterization:
Cardiac catheterization has been used as a preoperative imaging modality,
however,
its invasive nature and exposure to ionizing radiation are significant drawbacks (7).
Treatment and prognosis
Surgical repair of SVASD is usually recommended,
as early as possible,
because the age-related decrease in left ventricular compliance increases the left-to-right shunt.
Compounded by the presence of anomalous pulmonary venous drainage,
right heart volume overload may occur more readily than with other types of ASD,
with resultant development of right heart failure and secondary pulmonary hypertension (8).
The basic principle of repair is redirection of the PAPVR through the interatrial communication into the left atrium.
The surgical procedure is more complex than other types of ASDs,
with an increased risk of stenosis of the SVC or pulmonary veins,
residual shunting and sinoatrial node dysfunction.
Different surgical techniques are uitilized,
depending on the anatomy of the defects and the preferences of the surgeon.
The usual surgical approach involves a median sternotomy and cardiopulmonary bypass with moderate systemic hypothermia (25-32ºC).
Repairs divert the PAPVR through the SVASD into the left atrium.
This diversion require a pericardial patch (8).
The prognosis for these patients is excellent.
There is a low complication rate and low postprocedural mortality.
The survival rate is similar to that expected in age and gender matched controlled populations.
Symptomatic improvement is achieved in the 77% of patients,
especially in older patients.
However,
postoperative sinus node dysfunction is more common than following secundum ASD repairs,
due to either anatomic anomaly of the sinus node or surgical trauma caused by proximity of the SVASD to the sinus node.
New-onset atrial fibrillation is a potential complication,
especially in older patients.
The mechanism is due to scar-dependent multiple reentries or increased size and fibrosis of the atrium,
secondary to increasing pulmonary venous pressure.