The hearts of six Dutch landrace hybrid pigs were obtained from the slaughterhouse.
Protocols at the slaughterhouse and laboratory were in accordance with EC regulations 1069/2009 regarding the use of slaughterhouse animal material for diagnosis and research,
supervised by the Dutch Government (Dutch Ministry of Agriculture,
Nature and Food Quality),
and approved by the associated legal authorities of animal welfare (Food and Consumer Product Safety Authority).
In this study,
perfusion setup according to Langendorff was used (PhysioHeartÒ,
LifeTec Group,
Eindhoven,
The Netherlands) (14,15).
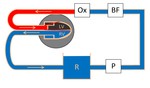
A centrifugal pump was used to pump porcine blood from the venous reservoir retrograde through the aorta to the heart (figure 1).
Blood was pumped through the coronary arteries,
preventing the aortic valve from opening.
Blood was oxygenated and kept at a temperature of approximately 38°C.
A mixture of glucose and insulin was added to keep the blood glucose level between 5 and 7 mmol/L.
All hearts were defibrillated at 10 to 30 Joules in order to acquire stable sinus.
An external pacemaker was used to stabilize the hearts,
if necessary.
Electrocardiography (ECG) leads were place on the platform,
which provided excellent conduction of the ECG signal to the CT (figure 2).
An inflatable cuff was placed around the proximal circumflex (Cx) artery,
after dissecting the proximal Cx from the surrounding tissue.
A pressure wire was placed inside the Cx artery,
allowing monitoring of the stenosis grade during the experiment.
The pressure wire allowed calculation of FFR by comparing pressure before and after the stenosis.
CT myocardial perfusion parameters were studied at six FFR-based stenosis grades: no stenosis,
FFR 0.8,
FFR 0.7,
FFR 0.6,
FFR 0.5 and total occlusion.
Heart rate,
arterial blood flow (ml/min) and arterial blood pressure (mmHg) were monitored during the experiment.
A third generation dual source CT system (SOMATOM Force,
Siemens Healthineers,
Forchheim,
Germany) was used to analyze perfusion of the porcine hearts.
Dynamic CT myocardial perfusion imaging was performed at all FFR-based stenosis grades.
Dynamic CT scans were performed in shuttle mode during end-systole,
providing a scan z-range of 102 mm,
covering the entire heart.
Other acquisition parameters included tube voltage 70 kV,
tube current time product 350 mAs per rotation,
and rotation time of 250 ms.
The inflow tube was looped through the field of view,
allowing for calculation of an arterial input function.
Myocardial enhancement returned to baseline by applying a 5-minute delay between each contrast-enhanced scan acquisition.
An injection of 15 mL (contrast to saline ratio 35%/65%) of ioxaglate (Hexabrix,
320 mg/mL,
Guerbet,
Paris,
France) at 3 mL/s was used for the dynamic CT scans.
Injection site was 200 cm prior to the aortic annulus,
allowing proper mixing of blood and contrast.
Dynamic scans were started five seconds before the injection of the contrast.
Dynamic CT data were reconstructed with 3.0 mm slice thickness and 1.5 mm increment in the short-axis plane.
Perfusion datasets were analysed using Volume Perfusion CT (VPCT) myocardium software (Siemens).
Myocardial segments were drawn at basal,
mid-ventricular and apical level using the American Heart Association (AHA) 17-segment model (16).
The apex was excluded from the analysis,
resulting in a total of 16 segments per scan.
The myocardial segments were divided into two groups based on the vessel territory: 1.
left anterior descending (LAD)/ right coronary artery (RCA) territory - non-ischaemic and 2.
segments perfused by the Cx - ischaemic.
The total occlusion scan was used to determine the Cx segments.
VPCT myocardium software calculates myocardial blood flow and volume for every separate voxel based on the arterial input function and the signal increase in the myocardium.
Mean values of myocardial blood flow (MBF) (mL/100mL/min) and myocardial blood volume (MBV) (mL/100mL) were calculated per myocardial segment.
Median values of MBF and MBV were compared between ‘non-ischaemic’ segments with normal perfusion and ‘ischaemic’ segments perfused by the Cx artery.
The comparison was performed for each stenosis grade (no stenosis,
FFR 0.80,
FFR 0.70,
FFR 0.60 and FFR 0.50).