Scheuermann disease
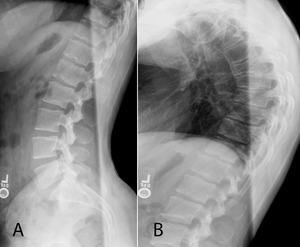
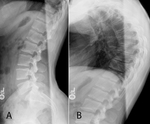
Fig. 1: Scheuermann disease. 15-year-old male with back pain and worsening posture. Lateral radiographs of the lumbar (A) and thoracic spine (B) demonstrate multi-level Schmörl nodes and anterior vertebral body wedging.
References: .
Scheuermann disease is the most common form of thoracic kyphosis in adolescence [5].
It is considered an osteochondrosis affecting the vertebral epiphyseal growth plates.
When the ring apophysis becomes avascular,
there is inhibited endochondral ossification with resulting endplate perforations and intravertebral disc herniations.
Etiology is unknown,
but it is thought to have a familial predisposition with a dominant autosomal pattern of inheritance.
Repetitive axial loading activity also poses as a biomechanical stress factor in the immature spine [6,7].
Compressive forces cause wedging deformity of the vertebral bodies,
defined by anterior wedging of three or more consecutive vertebrae by 5º,
resulting in a thoracic kyphosis greater than 45° with the apex more commonly seen between T7 and T9 [8–10].
In the atypical pattern of disease,
the apex of the kyphosis is lowered at the thoracolumbar junction,
situated between T10 and T12 [11].
Other associated imaging findings in the vertebrae also include endplate undulations from multiple Schmörl nodes and loss of the intervening disc height,
especially anteriorly (Fig. 1) [7].
The reported incidence ranges from 1% to 8% of the general population,
although its true incidence is probably understated since it is often either missed or attributed to poor posture [2,12].
If affects boys and girls equally and age of onset is between 10 and 12 years [13].
Atypical Scheuermann kyphosis (lumbar type) has a greater incidence reported in athletically active adolescent males [14].
Pain and increasing back deformity are the most common presenting symptoms.
It should be differentiated from postural kyphosis with a lack of correction on hyperextension [10].
The condition is expected to have a benign course with few symptoms persisting beyond maturity,
and treatment is mainly conservative. Bracing may be recommended for those with remaining growth and a curve between 50° and 70°,
especially if painful [10,13].
Those with severe kyphosis above 70° are surgical candidates [9].
Surgical correction and fixation is only considered in patients who have a severe kyphotic curvature above 70°,
who have refractory pain or an unacceptable cosmetic deformity [9,11,13,15].
Legg-Calvé-Perthes disease
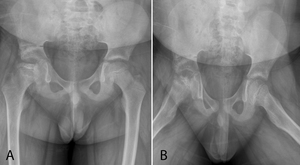
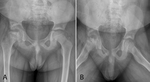
Fig. 2: Legg-Calvé-Perthes disease. 5-year-old boy with hip pain and limp. AP and frog-leg lateral radiographs of the bilateral hips demonstrating flattening, sclerosis and fragmentation of the right femoral head.
References: .
Legg-Calvé-Perthes disease is an avascular necrosis of the developing femoral head.
Etiology remains unclear,
with various factors being proposed.
The pathogenesis is based on the osteonecrosis of the capital epiphysis of the femur,
which leads to structural abnormality characterized by flattening and collapse.
The necrosis of the capital femoral head may be related to repeated microtrauma,
especially on a more susceptible skeleton; and due to a hypercoagulable state such as due to exposure to passive smoke [16].
Legg-Calvé-Perthes disease usually affects children aged 4 to 8 years [17].
This disorder is more frequent in boys than in girls,
with a ratio of approximately 5:1 [18].
The clinical presentation includes hip pain,
limping,
and limited hip motion.
The symptoms are usually unilateral; but can be bilateral in approximately 15% of the cases,
but almost always asynchronous [18].
Other causes of avascular necrosis should be excluded in the differentials,
such as sickle cell anemia,
corticosteroid treatment or Gaucher’s disease [18].
A plain radiograph,
including a frog-leg lateral view,
is usually the first chosen imaging method.
Appearance depends on the disease progression,
and early on there may be no significant changes.
Four radiographic stages have been described by Waldenstrom: initial stage,
fragmentation stage,
reossification stage,
and the healed stage [17].
The earliest radiographic finding is sclerosis of the affected femoral head.
Varying degrees of fragmentation may be seen with flattening (Fig. 2) [18].
A subchondral lucency (crescent sign) in the anterior aspect of the femoral head,
better depicted on the frog-leg lateral view,
is suggestive of imminent articular collapse [19].
MRI will allow a prompt diagnosis due to its high sensitivity and specificity.
It can also accurately stage the disease process,
provide prognostic information and detect possible complications [18].
T1-weighted images reveal loss of high bone marrow signal in the ischemic bone.
T2-weighted/STIR sequences can show variable signal intensity,
including areas of increased signal thought to represent edema.
Sometimes,
a curvilinear subchondral T2-weighted signal hyperintensity and T1-weighted signal hypointensity may be observed in the anterosuperior aspect of the femoral head,
correlating with a subchondral fracture.
In suspected early and radiographically occult disease,
MRI with intravenous administration of gadolinium-based contrast media may assess proximal femoral epiphyseal perfusion.
Hypoperfusion may be one of the earliest indicators of Perthes disease [18].
After healing,
Perthes disease may result in deformity of the femoral head such as coxa plana (nonspherical femoral head),
coxa magna (enlarged),
coxa breva (short) and coxa vara (higher greater trochanter) [20].
The more frequently observed is coxa magna [21].
Treatment may be operative or nonoperative.
This a self-healing disease,
with vessel recanalization and neovascularization,
and nonoperative measures may be used,
such as rest and use of braces.
Occasionally,
osteotomy must be done to achieve containment of the femoral head by the acetabulum [17].
Panner disease
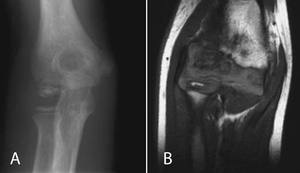
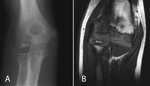
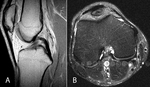
Fig. 3: Panner disease. 7-year-old girl with right elbow pain. (A) AP radiograph demonstrating sclerosis, fragmentation and poorly defined cortical margins of the humeral capitellum. (B) Coronal T1 weighted MR image demonstrating a small ossific fragment along the inferior portion of the capitellum, and low signal in the articular surface.
References: .
Panner disease is a benign condition characterized by osteochondrosis of the humeral capitellum.
The capitellar epiphysis has a tenuous vascular supply as it relies on one or two isolated vessels that enter posteriorly and traverse the capitellum,
with no metaphyseal collateral flow.
Therefore,
development of disease is thought to be related to repetitive microtrauma that can disrupt the blood flow to the capitellum’s ossification center [22,23].
This results in ischemia,
followed by disordered endochondral ossification affecting the entire capitellum.
This is one feature that distinguishes Panner disease from OCD,
as the latter is associated with localized necrosis and is typically located at the anterior and lateral aspect of the ossifying capitellar epiphysis [22].
These two disorders may in fact represent a spectrum of disease that differs in the age of presentation [24].
While Panner disease is more common in boys 7 to 12 years of age,
OCD mostly affects male athletes between 12 and 16 years of age and occurs at a time when the capitellar epiphysis is almost completely ossified [22].
Both are believed to be a consequence of abnormally high valgus compressive forces along the radiocapitellar joint [25,26].
Patients present with lateral elbow pain,
swelling,
and stiffness [22].
Imaging findings on conventional radiography are demineralization of the capitellum with ill-defined cortical margins,
followed by sclerosis and fragmentation of the capitellar epiphysis (Fig. 3) [22,26,27].
On MRI,
there is low signal intensity on T1-weighted images with variable signal intensity on T2-weighted images [22].
Contrarily to OCD,
the overlying cartilage in Panner disease should be intact,
with no loose body formation [22].
Panner disease is self-limited and amenable to conservative treatment,
having an excellent prognosis [22].
Kienböck disease
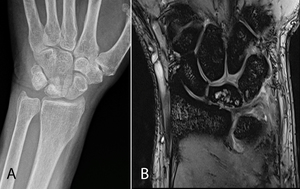
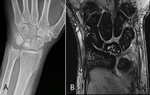
Fig. 5: Kienböck disease. 30-year-old male with grip weakness and pain. (A) PA radiograph and (B) Coronal fat-saturated T2 weighted MR image of the hand demonstrating sclerosis and mild flattening with foci of high T2 signal involving the lunate bone.
References: .
Kienböck disease is defined by avascular necrosis of the lunate bone [28].
The etiology is not well established but there are several predisposing risk factors,
namely a vulnerable blood supply,
a fixed position of the lunate and the presence of a short ulna [29].
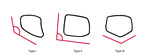
The lunate shape (Fig. 4),
also appears to be associated with the risk for lunate necrosis,
which is higher in the square or rectangular shape [30].
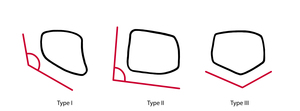
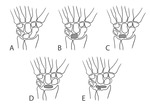
Fig. 4: Antuña Zapico classification based on lunate bone morphology. There are three types, based on the angle between the lateral scaphoid and proximal radial sides of the lunate. In type I, the angle is greater than 130°. In type II, the angle is inferior to 130°. In type III, there are two distinct surfaces which articulate with the radius and with the triangular fibrocartilage complex. Types I and II present a higher risk of Kienböck disease.
References: Filipa Duarte Figueiredo
Its incidence is higher in males aged 20-40 years,
and it is related to axial load stress to the dorsiflexed wrist as occurs in manual workers [30].
It is uncommon in the immature skeleton.
The disease is usually unilateral,
the reported incidence of bilateral disease being around 3-7% [31].
Symptoms are unspecific,
however the most common are dorsal wrist pain and lunate tenderness [28].
Imaging findings include sclerosis and collapse of lunate bone (Fig. 5).
Kienböck disease is slowly progressive and may result in joint destruction if left untreated.
It often requires surgery,
and the procedures vary according to the stage of the disease.
[32]
Staging based on radiographic appearance was developed by Stahl and later modified by Lichtman (Fig. 6).
There are four stages of disease.
In stage I,
the lunate appears normal on both conventional radiography (CR) and computed tomography (CT) while on MRI there’s low signal intensity on T1-WI and typically low signal on T2-WI (although a high signal intensity on T2-WI may occur in the presence of edema).
In stage II,
the lunate appears sclerotic and with increased density on CR and CT without changes in its morphology,
while MRI shows low signal intensity on T1-WI and variable signal on T2-WI with areas of low signal intensity on T2 representing sclerosis.
Stage III is determined by lunate articular surface collapse,
and may be further subdivided into IIIa,
IIIb and IIIc.
In stage IIIa,
there is no scaphoid rotation; in stage IIIb,
there is fixed scaphoid rotatory subluxation,
and in stage IIIc,
a coronal fracture of the lunate is present in addition to scaphoid rotation.
Stage IV is when there are stage III findings with secondary radiocarpal or midcarpal osteoarthritis [28–30].
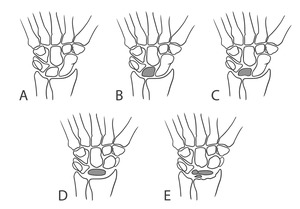
Fig. 6: Lichtman classification of Kienböck disease. (A) Stage I, without density or shape change. (B) Stage II, increased lunate density without change in lunate size or shape. (C) Stage IIIa, lunate articular surface collapse without scaphoid rotation. (D) Stage IIIb, lunate articular surface collapse with fixed scaphoid rotatory subluxation. (E) Stage IV, secondary osteoarthritic changes.
References: Filipa Duarte Figueiredo
Osteochondritis dissecans
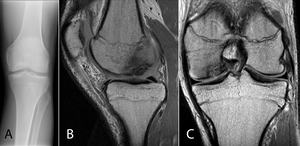
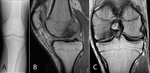
Fig. 7: Osteochondritis dissecans. 18-year-old male soccer player with locking sensation and pain in knee. (A) AP radiograph of the left knee demonstrating a lucent subchondral defect involving the medial aspect of the medial femoral condyle. Sagittal (B) and Coronal (C) PD MR images demonstrating low signal intensity within an ovoid subchondral defect involving the medial femoral condyle.
References: .
The term osteochondritis dissecans (OCD) was introduced by Franz König [33].
As previously stated,
this designation can be misleading as this pathology is not primarily related to inflammation [33].
There are various factors contributing to the etiology,
such as repetitive microtrauma or overuse,
ischemia and genetic predisposition [33,34].
OCD is regarded as an acquired lesion of subchondral bone with different degrees of bone resorption,
collapse and sequestrum formation unrelated to an acute osteochondral fracture [35].
The knee is the most common involved joint,
occurring in the lateral aspect of the medial femoral condyle in 69% of the cases,
followed by the lateral femoral condyle (15%); patella (5%) and femoral trochlea (1%).
It less frequently affects the elbow or ankle [36,37].
There is a male predominance with a ratio of 5:3.
Bilateral involvement is described in up to 25% of cases [38].
Presentation is often vague and includes poorly localized pain exacerbated by activity [36].
In the knee,
pain is particularly worsened when climbing stairs [38].
If the process involves the capitellum,
as occurs with repetitive valgus stress on the elbow in baseball pitchers,
the symptoms are pain,
tenderness and swelling on the elbow’s lateral aspect [37].
For the diagnosis of OCD,
conventional radiography is useful to exclude other bone lesions and to evaluate skeletal maturity [34].
It may show an irregular crescentic subchondral radiolucency (Fig. 7) [33,36].
A tunnel view allows better visualization of the posterior aspect of the femoral condyles [34].
MRI has become increasingly more important in the diagnostic evaluation of OCD,
as it can identify the location of the suspected lesion,
accurately estimate its size and evaluate the status of cartilage and subchondral bone,
being able to ascertain whether there is instability or detachment of the fragment [33,38].
MRI has also been found to be advantageous as a noninvasive means to document lesion progression or healing [39,40].
Most lesions are stable and heal favorably with nonoperative treatment such as immobilization.
Surgical treatment is indicated if the nonoperative treatment fails,
as well as in unstable lesions and in patients approaching physeal closure [38].
The most common surgical procedures are chondroplasty or debridement [41].
Osgood-Schlatter disease
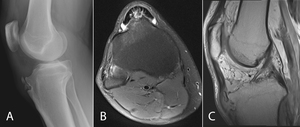
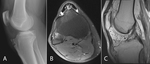
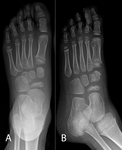
Fig. 8: Osgood-Schlatter disease. 18-year-old male with right knee pain. (A) Lateral radiograph demonstrating fragmentation of the tibial tubercle. (B) Axial fat-saturated T2 weighted MR image demonstrating high signal at the base of the tibial tuberosity. (C) Sagittal PD MR image demonstrating fluid within the infrapatellar bursa.
References: .
Osgood-Schlatter disease is a traction apophysitis of the tibial tubercle due to repetitive strain at the insertion of the patellar tendon with chronic avulsion of the tibial tubercle [42,43].
This disease is more common in boys than in girls,
and is related to physical activity that requires jumping,
squatting or kicking [44].
It occurs between the ages of 12 and 15 years in boys and 8 to 12 years in girls [42,45].
The disease occurs bilaterally in 25-50% of the cases [43].
It usually presents as a painful bump over the tibial tuberosity,
corresponding to secondary reactive heterotopic ossification [46].
Plain radiographs in the lateral view show calcification and thickening of the patellar tendon,
irregular ossification of the tibial tubercle with separation of the apophysis from the tibial tuberosity,
and anterior soft tissue edema (Fig. 8) [46].
The irregularity of the apophysis occurs in the early stages,
while fragmentation occurs later [42].
It is important to distinguish these changes from normal development in which there may exist fragmentation of the tibial tubercle representing the ossification center [44].
In this normal variant,
there is no surrounding inflammation [46].
MRI findings include bone marrow edema in the tibial tubercle and the surrounding soft tissues,
thickening and edema of the patellar tendon or infrapatellar bursitis [44,46].
Treatment is usually conservative unless the symptoms persist.
The presence of a residual ossicle may require surgical excision after skeletal maturity [42,47].
In the differential diagnosis,
one should take into consideration Sinding-Larsen-Johansson syndrome,
Hoffa’s syndrome,
tumors,
osteomyelitis,
fracture of the tibial tuberosity,
among other entities [47].
Sinding-Larsen-Johansson disease
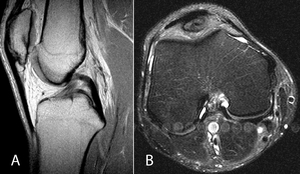
Fig. 9: Sinding-Larsen-Johansson disease. 11-year-old female with knee pain and swelling after gymnastics practice. (A) Sagittal PD MR image demonstrating a small ossific fragment along the inferior pole of the patella. (B) Axial fat-saturated T2 weighted MR image redemonstrating the small ossific fragment anterior to the distal femur.
References: .
Sinding-Larsen-Johansson disease is another common cause of anterior knee pain,
characterized by traction apophysitis that affects the patellar tendon at its origin in the lower pole of the patella [48].
The pathogenesis is similar to that of Osgood-Schlatter disease,
as both are related to traction injuries at the immature osteotendinous junctions [49,50].
However,
while the Osgood-Schlatter disease affects the distal portion of patellar tendon at its insertion into the tibial tuberosity,
Sinding-Larsen-Johansson affects its proximal insertion at the inferior pole of the patella.
Both diseases can occur simultaneously [51].
Sinding-Larsen-Johansson disease is seen more commonly in boys who play sports that involve running or jumping such as football,
gymnastics or running,
occurring typically between 10 and 14 years old [49].
It presents with swelling and pain localized to the lower pole of the patella.
Imaging findings in plain radiographs are soft tissue edema and bony fragments adjacent to the lower pole of the patella,
although the extent may be underestimated [52].
On MRI,
there is thickening and edema of the proximal portion of the patellar tendon,
fragmentation of the inferior pole of the patella,
and Hoffa’s fat pad edema (Fig. 9) [48].
Ultrasound may be useful in the early stages or for follow-up,
and findings include thickening of the patellar tendon,
fragmentation of the patella’s lower pole and cartilage swelling [52].
Köhler disease
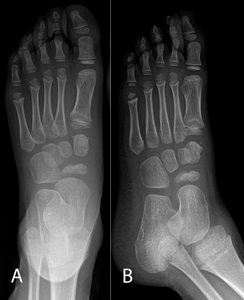
Fig. 10: Köhler disease. 6-year-old boy with left foot pain. AP and medial oblique views demonstrating flattening and sclerosis of the navicular bone.
References: .
Köhler disease refers to avascular necrosis of the tarsal navicular bone [53].
Pathophysiology may be related to mechanical stress associated with a delayed ossification.
As the navicular bone is the last tarsal bone to ossify,
it is susceptible to compression between the adjacent ossified talus and cuneiforms,
constricting the blood vessels and eventually leading to ischemia and avascular necrosis [53,54].
It is a rare disease that more commonly affects boys aged 4 to 9 years [55,56].
It may be bilateral in up to 25% of the cases [57].
It presents as a swollen painful foot,
and a characteristic limping as the patient avoids bearing weight on the medial side of affected foot [55].
Radiographic findings include sclerosis,
irregularity and bone fragmentation (Fig. 10).
The navicular may sometimes appear collapsed,
or it may be normal in shape with a uniform increase in density and minimal fragmentation.
Treatment is conservative,
with analgesics,
rest,
arch support,
and sometimes immobilization in a short leg walking cast for 4 to 6 months [54].
The disease is self-limited,
and the prognosis is excellent.
Distinction should be made between this entity and spontaneous osteonecrosis of the navicular bone that may occur in the adult,
known as Mueller-Weiss syndrome or Brailsford disease [58].
Sever disease
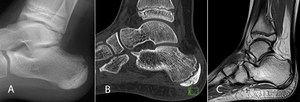
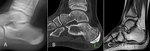
Fig. 11: Sever disease. 10-year-old boy with heel pain, worse with running and jumping. (A) Lateral radiograph and (B) Sagittal CT image demonstrating sclerosis of the calcaneal apophysis. (C) Sagittal PD MR image shows corresponding low signal intensity within the calcaneal apophysis.
References: .
Sever disease is the most frequent cause of heel pain in growing children who are physically active,
especially in high-impact sports such as football,
soccer or basketball [59].
It is a calcaneal apophysitis,
caused by repetitive traction of the Achilles tendon on its insertion site in the calcaneal tuberosity.
The disease is more common in boys and presents between the ages of 7 and 14 years [59,60].
Bilateral involvement is present in approximately 60% of cases [61].
Diagnosis is usually made on the basis of history and physical examination [62].
Radiographs are frequently obtained as part of the clinical evaluation in routine orthopedic practice,
but findings are non-specific (Fig. 11).
The increased density and fragmentation of the apophysis may be also seen as a variant of normal condition [63].
MRI may help establish the diagnosis,
being able to show edematous changes in the calcaneal apophysis [63].
Imaging exams are mostly useful to exclude other causes of heel pain,
such as fractures,
tumors or infection.
The disease is self-limiting,
but typically requires limiting exercise activity.
Anti-inflammatory drugs can also reduce symptoms.
A short-term cast may be used in severe cases [60].
Freiberg infraction
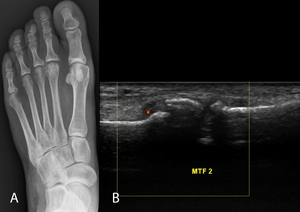
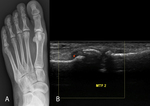
Fig. 12: Freiberg infraction. 18-year-old female with foot swelling and pain that awakens her from sleep. (A) AP radiograph depicts mild flattening and sclerosis of the second metatarsal head. (B) US image with color Doppler demonstrates fragmentation of the second metatarsal head.
References: .
Freiberg disease,
also known as Freiberg infraction is an osteochondrosis of the metatarsal head [64].
The term infraction refers to an incomplete fracture of bone without displacement of fragments [65].
However,
etiology is multifactorial and includes repetitive microtrauma and vascular compromise [66].
The most common location is the second metatarsal head (68%),
followed by the third (27%),
and rarely the fourth [66].
Bilateral involvement can be seen in up to 10% of cases [64].
This entity is more common in females with a ratio of 5:1 and has a peak incidence in adolescence (12-18 years) [65,67].
As the second metatarsal is the longest and the least mobile,
it may be subjected to an increased stress that ultimately compromises vascular supply to the subchondral bone and leads to osteonecrosis [64].
High-heeled shoes have been implicated as a causative factor [68,69].
Patients often present with swelling and pain around the affected area that is worse with bearing weight and walking [66].
Conventional radiographs may show sclerosis of the metatarsal head and widening of the joint space.
Complete collapse of the metatarsal head,
flattening and fragmentation can be seen in later stages (Fig. 12) [69].
Early MRI can depict bone marrow edema (low signal intensity on T1-WI and high signal intensity on T2-WI),
and as the disease progresses there may be flattening of the metatarsal head and bone sclerosis (low signal intensity on T2-WI) [68,69].
The most widely used classification of Freiberg infraction is that of Smillie,
in which five stages of metatarsal head degeneration were described (Table 2).
It can aid in the prognosis and treatment planning [66,67].
In early stages (I-III),
a conservative treatment may be adequate and will include analgesics,
proper footwear and activity modification.
In advanced stages (IV-V),
or in patients who do not respond to conservative measures,
a surgical approach is required [64,66].