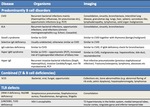
The imaging findings in primary immunodeficiency vary depending on the type of primary immunodeficiency.
The thoracic imaging findings can be broadly characterised into early and late findings.
The early findings encompass respiratory tract infections and small airways disorders whilst late manifestations include chronic lung changes as well as malignancy.
Infection
Patients with humoral immunodeficiencies often present with recurrent infections before a diagnosis of immunodeficiency is made.
The imaging patterns of infections in immunodeficiency are non-specific,
however the CT findings can narrow the differential diagnosis (typical vs.
atypical infection) depending on the background of pre-existing lung damage (bronchiectasis,
ILD),
(Fig 1-6).
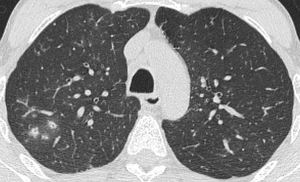
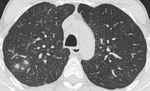
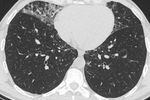
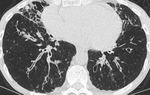
Fig. 1: This CT demonstrates focal ground glass opacities. This patient had a diagnosis of XLA and had recurrent infections. Sputum samples were positive for Haemophilus influenzae.
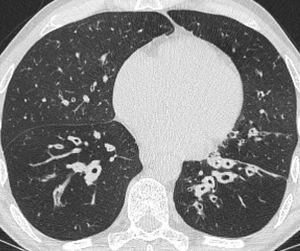
Fig. 2: In the same patient with XLA there is lower lobe bronchiectasis and bronchial wall thickening.
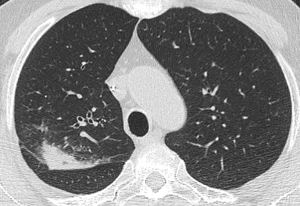
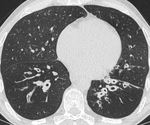
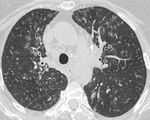
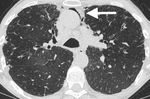
Fig. 3: Axial CT image showing segmental right upper lobe consolidation in a patient with CVID.
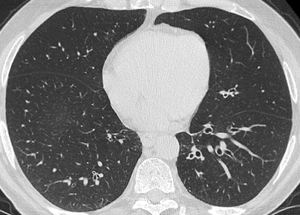
Fig. 4: In the same patient with CVID there is lower lobe bronchiectasis.
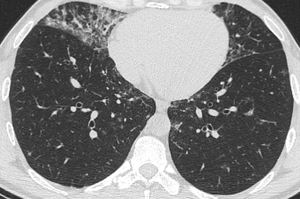
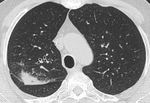
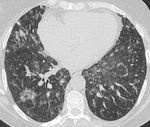
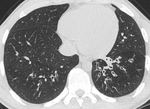
Fig. 5: Axial CT image in a patient with CVID showing multiple micronodules with patchy ground glass opacities, predominantly in the middle lobe and lingula suggestive of an atypical infection. Sputum samples confirmed Haemophilius influenzae and Pseudomonas aeruginosa.
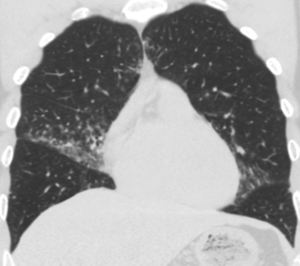
Fig. 6: Coronal reconstruction in the same patient with CVID depicting the mid zone distribution of the nodules and ground glass opacification.
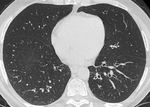
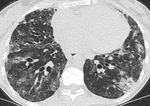
The most frequently identified pathogens are encapsulated bacteria particularly both Haemophilus influenza and Streptococcus pneumonia which for example are commonly linked to agammaglobulinaemia,
CVID,
hyper-IgM syndromes,
complement factor deficiencies and SCID/CID.
These patients are predisposed to opportunistic infections,
which include fungi (Pneumocystis jiroveci and Aspergillus),
Cytomegalovirus and Mycobacteria (Figure 7-9).
In both chronic granulomatous disease (CGD) and SCID/CID Aspergillus infections are common.
Specifically Aspergillus fumigatus and Aspergillus nidulans have been associated with CGD.
Infection with Pneumocystis jiroveci has been associated with SCID/CID,
idiopathic CD4 lymphopenia and hyper-IgM syndrome and highlights the vital role of CD4 T cells.
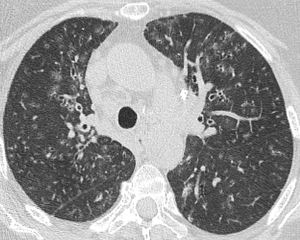
Fig. 7: Upper zone image from a CT chest of a patient with CVID and recurrent infections. There are ground glass opacities and micronodularity in a bronchocentric and centrilobular distribution with evidence of bronchial wall thickening. This patient had positive sputum samples for Aspergillus and Aspergillus nidulans.
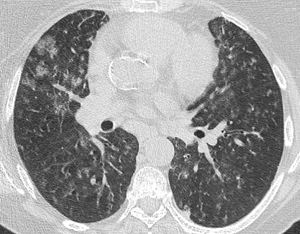
Fig. 8: Mid zone image from a CT chest of the same patient with CVID demonstrating the extent of the ground glass opacification.
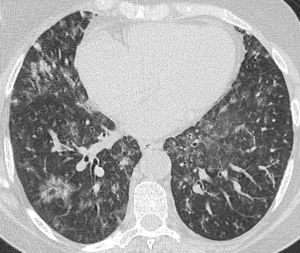
Fig. 9: Lower zone image from a CT chest of the same patient with CVID and recurrent infections, further revealing the extent of the ground glass opacification.
Understanding and identifying infections in primary immunodeficiency and in particular the pathogens more prevalent within each type of immunodeficiency,
can significantly impact management with regards to prophylactic antibiotics,
as well as immunisations that may be offered.
Small airway disorders (bronchiectasis,
bronchial wall thickening and air trapping)
Structural alterations secondary to recurrent infections in primary immunodeficiency can lead to bronchiectasis,
bronchial wall thickening and subsequent air trapping.
Bronchial wall thickening and bronchiectasis are the most common findings on a CT chest in longstanding humoral immunodeficiency.
Therefore in a patient with these findings immunodeficiency should be considered as a cause.
Bronchiectasis in humoral immunodeficiency is most commonly found in the lower lobes,
with a focal or diffuse distribution and cylindrical shape (Figure 10-12).
Bronchiectasis is not a common pulmonary finding in selective IgG deficiency due to compensating IgG antibodies unlike both CVID and XLA.
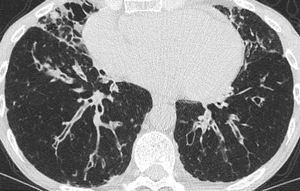
Fig. 10: Cylindrical lingual, lower and middle lobe predominant bronchiectasis with bronchial wall thickening in a patient with CVID.
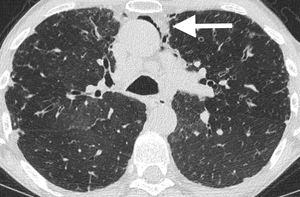
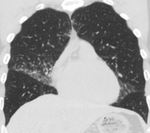
Fig. 11: Complication of recurrent infections with pneumomediastinum in the same patient with CVID.
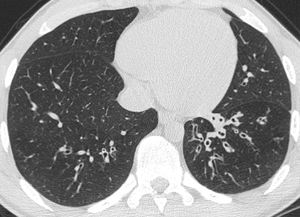
Fig. 12: Cylindrial bibasal bronchiectasis and bronchial wall thickening in a patient with Bruton’s XLA.
Chronic lung changes and inflammatory conditions.
ILD
Interstitial lung disease is a major complication in primary immunodeficiency.
Literature reports that chronic lung disease is one of the most prevalent complications in CVID,
which is thought to be due to immune dysfunction and delayed diagnosis.
Chronic lung disease and ILD are less common in XLA,
which is thought to be due to earlier diagnosis.
The most common types of ILD in patients with humoral immunodeficiency are organising pneumonia and lymphoid interstitial pneumonia (LIP).
Less commonly,
non-specific interstitial pneumonia (NSIP) and usual interstitial pneumonia (UIP) can also be present.
Organising pneumonia can be seen in primary immunodeficiency and is most commonly described in CVID (Figure 13).
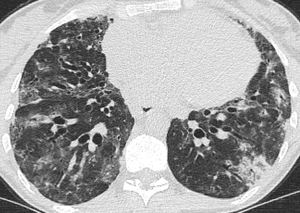
Fig. 13: Axial CT image of a patient with a granulomatous variant of CVID demonstrating perilobular consolidation and ground glass opacities suggestive of organizing pneumonia. There is associated fibrosis evident by volume loss and traction bronchiectasis.
Granulomatous and lymphocytic lung disease
Granulomatosis is often diagnosed before a humoral immunodeficiency is identified and can be seen in patients with CVID.
Imaging appearances can be similar to sarcoidosis with perilymphatic nodules,
pulmonary fibrosis and lymphadenopathy and distinction between the two on histology may not be possible (Figure 14-16).
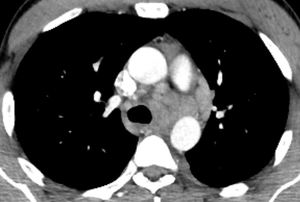
Fig. 14: Axial CT image on mediastinal windows, which demonstrates mediastinal and hilar lymphadenopathy in a patient with granulomatous CVID. EBUS histology showed non-necrotising granulomatous inflammation. The right lower lobe VATS wedge biopsy confirmed chronic granulomatous inflammation.
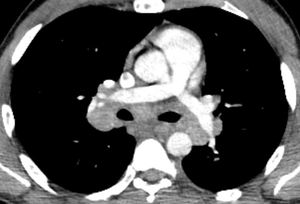
Fig. 15: Further axial CT image on mediastinal windows in the same patient demonstrating mediastinal and hilar lymphadenopathy.
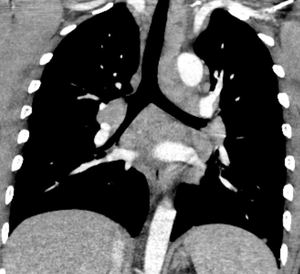
Fig. 16: Coronal reconstruction on mediastinal windows in the same patient showing the extent of the mediastinal and hilar lymphadenopathy.
Granulomatous and lymphocytic lung disease (GLILD) encompasses LIP,
follicular bronchiolitis and lymphoid hyperplasia.
Imaging findings most frequently include mid to lower zone reticulation,
nodules,
consolidation and lymphadenopathy (Figure 17-19).
GLILD is the most common type of interstitial lung disease in CVID.
It is not seen in XLA,
which is thought to be related to T cell dysfunction.
A diagnosis of GLILD suggests a worse prognosis and also an increased risk of lymphoproliferative disease.
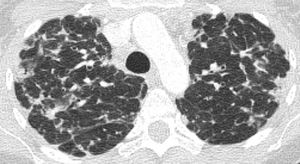
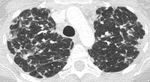
Fig. 17: Upper zone axial CT chest image in a patient with CVID, which shows extensive reticulation, parenchymal interstitial fibrosis and consolidation consistent with GLILD; proven on lung biopsy.
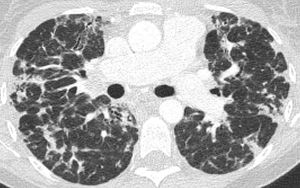
Fig. 18: Mid zone axial CT chest image in the same patient with CVID.
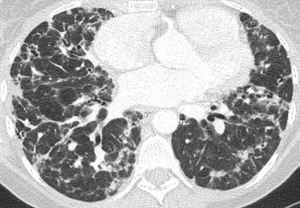
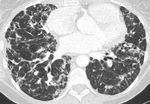
Fig. 19: Lower zone axial CT chest image in the same patient with CVID showing the extent of the interstitial fibrosis in GLILD.
Thoracic neoplasms
Patients with primary immunodeficiency are at increased risk of thoracic neoplasms such as nodular lymphoid hyperplasia,
lymphoma and thymoma.
Association between primary immunodeficiency and lymphoma has been described,
in particular with non-Hodgkin lymphoma.
The pathophysiology is thought to be influenced by immune factors including immune system impairment,
genetic factors and prolonged infections.
CT findings may include an anterior mediastinal mass,
enlarged intrathoracic lymph nodes and pleural or bony lesions.
Good‘s syndrome is a rare disorder characterised by increased susceptibility to severe infection and is the presence of a thymoma in patients with humoral immunodeficiency.
The presence of a thymoma in a patient with a history of recurrent infections or pulmonary structural alterations,
suggestive of recurrent infections/chronic inflammation,
should prompt suspicion of Good's syndrome.
Detection of mediastinal masses,
even when small is important as resection of small thymomas can improve prognosis (Figure 20).
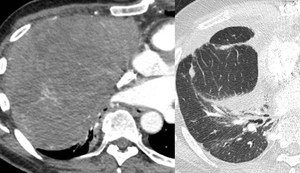
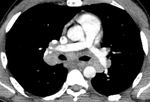
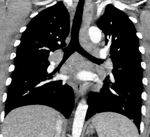
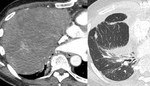
Fig. 20: This is an example of Good's syndrome in a patient with hypogammaglobulinaemia. The image on the left demonstrates a large right-sided soft tissue anterior mediastinal mass causing mediastinal shift consistent with a thymoma. Post resection imaging is demonstrated on the left.
Extrapulmonary manifestations
Primary immunodeficiency can affect multiple organs including both the gastrointestinal tract and the liver.
Gastrointestinal manifestations include an increased risk of gastric carcinoma.
Literature describes a range of liver involvement with infection,
nodular regenerative hyperplasia,
hepatomegaly,
splenomegaly,
cirrhosis,
and primary sclerosing cholangitis being a few of the many reported manifestations.