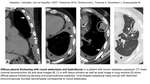
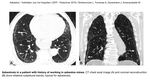
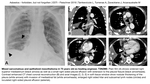
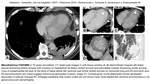
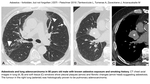
Benign asbestos pleural effusion (BAPE) can be the earliest presentation of asbestos-related disease,
but latency period is variable.
The fluid is exudative in origin,
often hemorrhagic with lymphocyte dominancy and does not contain asbestos bodies.
BAPE can also coexist with other benign asbestos related diseases and is usually not related with mesothelioma formation.
Effusion can resolve without treatment,
but diffuse pleural thickening (DPT) can develop as a consequence,
especially in recurrent cases (Fig. 1).
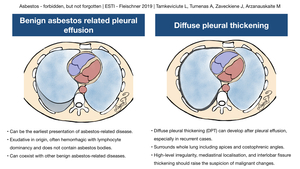
Fig. 2: Benign asbestos pleural effusion and diffuse pleural thickening
References: MD Laima Tamkeviciute
DPT does not calcify and surrounds whole lung including apices and costophrenic angles.
The localisation and shape of pleural thickening can help differentiate DPT from mesothelioma.
High-level irregularity,
mediastinal localisation,
and interlobar fissure thickening should raise the suspicion of malignant changes.
Positron emission tomography combined with computed tomography (PET-CT) is a noninvasive diagnostic method that can allow to assess the metabolic activity within the pleura and thus be used to differentiate DPT from mesothelioma.
BAPE has to be differentiated from other causes including malignant (lung cancer,
mesothelioma,
metastases,
etc.) and non-malignant (tuberculous pleuritis,
bacterial pleuritis,
collagen diseases,
and heart failure).
Microscopy,
staining,
bacterial cultures of the pleural fluid and pleural biopsy are useful to exclude alternative diagnoses.
Biochemical fluid analysis can also be of value: elevated hyaluronic acid,
adenosine deaminase,
and carcinoembryonic antigen levels suggest other diagnoses.
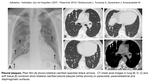
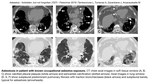
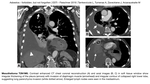
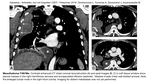
Pleural plaques (Fig. 3) are localised areas of calcified or non-calcified pleural thickening usually occurring on parietal pleura,
but can be found on visceral layer too.
Pleural plaques are most frequently bilateral,
but can be unilateral in rare cases.
They can be seen in paracostal,
mediastinal and diaphragmatic surfaces with sparing of apices and costophrenic angles.
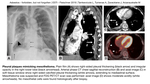
The size differs from small measuring 1-2mm to very large (Fig. 4). If a plaque is located in visceral pleura,
fibrotic parenchymal lung bands arising from the plaque can develop.
Extensive pleural plaques can result in reduced lung function.
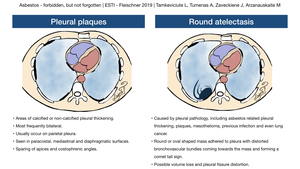
Fig. 5: Pleural plaques and round atelectasis
References: MD Laima Tamkeviciute
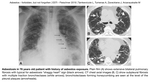
Round atelectasis (Fig. 1 C-D) can virtually be caused by any pleural pathology,
including infection and asbestos related pleural disease.
Radiologically it appears as a round or oval shaped mass adhered to pleura with distorted bronchovascular bundles coming towards the mass and forming comet tail sign.
Volume loss and pleural fissure distortion can also be seen.
Pleural thickening is the most common cause,
but mesothelioma or lung cancer can also be the reason of the atelectasis.
Hence,
follow-up or further investigation with PET/CT,
MRI or biopsy is needed.
Asbestosis is a dose dependent fibrotic lung disease and develops after prolonged exposure to high doses of asbestos.
In cases of lower dose exposure,
low grade fibrosis may develop and it may not be visible on CT.
The diagnosis of asbestosis is based on exposure history,
clinical and radiological findings.
Biopsy may be necessary in difficult cases when clinical or radiologic features are atypical or non-diagnostic.
Patients with asbestosis usually complain of breathlessness and dry cough.
On physical examination end-inspiratory crackles in the lower zones can be found.
Lung function is restricted with reduced lung volume,
forced vital capacity and diminished carbon monoxide diffusing capacity.
Asbestosis patients may even have hypoxemia of the arterial blood.
Histologically fibrosis and asbestos bodies are required for the diagnosis.
Fibrosis starts in the alveolar walls,
in peribronchiolar distribution.
Fibrosis of the walls of the respiratory bronchioles and alveolar ducts may also be present.
- Grade I asbestosis is limited to the first layer of alveoli.
- When asbestosis progresses,
fibrosis extends more to the periphery of the acinus,
although in grade II asbestosis some unaffected alveoli between two bronchioles is seen.
- Grade III asbestosis is diagnosed when all alveoli between two adjacent bronchioles are fibrosed.
In advanced disease peribronchiolar pattern may no longer be evident.
Different patterns are possible in such cases including usual interstitial pneumonia and non-specific interstitial pneumonia.
Sometimes the pattern may not match any other form of pulmonary fibrosis.
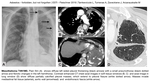
On plain film (Fig. 6),
fibrotic changes appearing as ground glass,
reticulations or honeycombing,
“shaggy” heart,
as well as blunted costophrenic angles and pleural plaques can be seen.
CT findings of asbestosis are variable.
Changes are usually located in the subpleural distribution and start at the lower lobes,
but can involve middle lobe and lingula or even upper lobes in advanced disease.
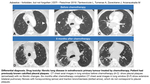
One of the earliest CT findings is the presence of sub-pleural curvilinear lines (Fig. 7).
Small subpleural nodules corresponding to peribronchiolar nodular fibrosis is another characteristic finding; these nodules together with small arteries can form “Y” shaped structures which do not reach pleura.
If pleural plaques are located in the visceral layer,
fibrotic lung changes might arise from them forming fibrotic bands radiating into the lung parenchyma.
Pleural based interlobular septal thickening is another feature of pulmonary fibrosis.
In advanced stages honeycombing and traction bronchiectasis can also develop (Fig. 8, Fig. 9).
Ground glass opacification might be the sign of fine fibrosis when changes are too small for CT resolution.
Asbestosis is usually related to pleural plaques,
and absence of them should raise the suspicion for an alternative cause of pulmonary fibrosis.
On the other hand features of asbestosis do not necessarily follow the distribution of pleural plaques.
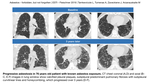
Asbestosis usually has slow course and does not progress quickly,
this can help to differentiate from other fibrotic lung diseases (Fig. 10).
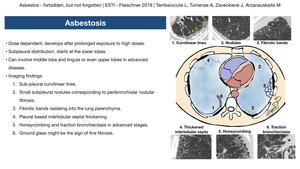
Fig. 11: Asbestosis
References: MD Laima Tamkeviciute
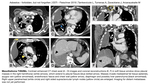
Mesothelioma is a malignant tumor originating from pleura which is highly related with asbestos exposure.
Chest wall radiation,
mineral erionite and cangerogenic simian virus 40 are another precursors.
The latency period after asbestos exposure is long and counts 30-40 years.
Although early detection can lead to more favourable outcomes,
overall prognosis is poor.
Mesotheliomas should be classified according to TNM 8 classification as a widely accepted standard.

Fig. 12: Mesothelioma TNM staging
References: International Association for the Study of Lung Cancer (IASLC) Staging Project
Mesothelioma can arise from either parietal or visceral layers.
Based on gross anatomy mesothelioma can be diffuse or localised.
Histologically it can be epithelioid (most often type),
sarcomatoid,
desmoplastic and biphasic.
Clinically mesothelioma manifests as chest pain and breathlessness.
The pain is usually dull and unilateral,
but sometimes can become neuropathic or pleuritic.
At early stages breathlessness is due to pleural effusion,
but in advanced disease it is caused by lung entrapment and reduced lung function.
Chest x-ray usually is the first imaging modality.
The earliest sign may be pleural effusion,
which can be loculated.
Later,
irregular pleural thickening with involvement of interlobar fissures can be seen (Fig. 13).
Pleural plaques can also be noted and should suggest asbestos exposure if the clinical history is unclear.
Although chest x-ray films can provide important information,
it is neither sensitive nor specific modality.
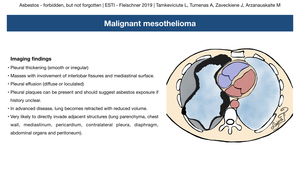
Fig. 14: Mesothelioma
References: MD Laima Tamkeviciute
According to British Thoracic Society guidelines,
CT thorax with contrast,
optimised for pleural evaluation,
should be the initial cross-sectional imaging modality in the evaluation of patients with suspected pleural mesothelioma.
In diffuse forms lobulated or smooth pleural thickening is the main feature,
followed by loculated pleural effusion.
In advanced disease lung becomes retracted with reduced volume.
CT is very important for staging,
because mesothelioma is very likely to invade adjacent structures directly.
Invasion of the lung parenchyma (Fig. 15),
mediastinum (Fig. 16,
Fig. 17,
Fig. 18),
chest wall (Fig. 19),
contralateral pleura,
diaphragm,
abdominal organs and peritoneum,
as well as spread to lymph nodes can be evaluated on contrast enhanced CT.
If T stage remains unclear and differentiation of the affected structures is important for management,
then dedicated MRI should be considered.
Hematogenic metastases are quite common,
but usually reported late or in autopsy.
FDG PET-CT should be considered if excluding distant metastases could change management,
but this modality should be avoided for patients who have had prior talc pleurodesis.
Due to potential false positive interpretations,
FDG PET-CT should be performed with a caution for population with a high prevalence of tuberculosis.
Thoracocentesis,
biopsy and biomarkers also play an essential role to differentiate mesothelioma from benign and other malignant pleural disease.
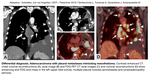
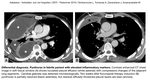
The most common differential diagnosis is pleural metastasis (Fig. 20), benign diffuse pleural thickening and empyema (in a relevant clinical setting)(Fig. 21).
Asbestos is also realated with incread fisk for lung cancer especially when combined with smoking history.
In those cases clinically and radiologically lung cancer manifests in its usual manner (Fig. 22).