Evaluation of the heart in a non-cardiac gated study is often limited due to motion artefact; however,
there are numerous important cardiac conditions that are crucial to be able to identify to guide appropriate further investigation and treatment.
Examples include but are not limited to the following: coronary artery disease,
myocardial infarction,
right ventricular thrombus,
cardiac metastasis,
displaced sternotomy wire,
and pericardial effusion.
Pericardium:
– Calcification and pericarditis
– Effusion
– Malignancy,
such as nodularity in mesothelioma
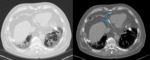
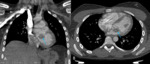
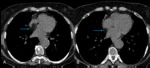
Pericardial calcification.
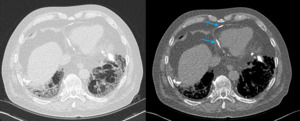
Fig. 2: Image 1, A: Axial CT thorax in lung windows of a patient with known asbestos exposure. Chronic fibrotic lung changes noted were in keeping with asbestosis (further images not shown); pericardial calcification also demonstrated. B: Axial CT thorax in soft tissue window at the same level. The pericardial calcification is much more conspicuous.
References: Deapartment of Radiology, Liverpool Heart and Chest Hospital, UK,2019
This patient had a known history of asbestos exposure; they underwent a CT thorax to assess the severity of their lung disease.
Pericardial calcification was identified incidentally and was in keeping with calcified asbestos plaques; there was no known history of pericarditis.
Pericardial calcification (PC) is rare,
its presence may suggest pericardial scarring,
but it can be seen in the absence of constrictive physiology and therefore can be picked up incidentally.
1
Tuberculous pericarditis is reported as the primary cause of PC,
other causes of pericardial calcification include sequelae of the following 2:
· Viral pericarditis
· Trauma
· Cardiac surgery
· Radiation
· Connective tissue disorders
· Malignancy
Patients with PC can be entirely asymptomatic.
Signs and symptoms arise from the development of constrictive pericarditis which is typically insidious in onset and can even develop decades after an episode of trauma or pericarditis.
3
Presenting features include the stigmata of right heart failure and low cardiac output:
· Hepatomegaly
· Ascites
· Peripheral oedema
· Exertional dyspnoea
· Orthopnoea
· Fatigue
· Raised JVP
· Hepatojugular reflux
· Kussmauls sign
· Pulsus paradoxus
It is reported that approximately 30% of patients with PC may have signs and symptoms of constrictive pericarditis,
and up to 50% of patients with constrictive pericarditis demonstrate PC on plain chest radiographs 1,4.
Therefore,
the absence of PC does not exclude constrictive pericarditis.
4
It is therefore important that pericardial calcification is reported if found incidentally.
The patient may have an appropriate history or pericarditis or could be a truly incidental finding.
Either way its presence may have clinical significance for the patient and should be mentioned when seen.
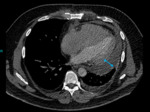
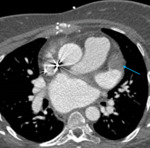
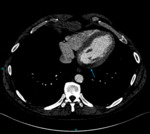
Pericardial Effusion.
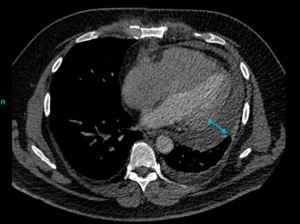
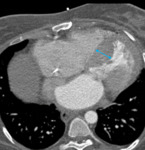
Fig. 3: Image 2: Axial CT chest in soft tissue window, the arrow indicates the significant pericardial effusion.
References: Deapartment of Radiology, Liverpool Heart and Chest Hospital, UK,2019
This 53 year patient was post op following an ascending root replacement and was slow to progress post op.
The CT chest was performed to rule out a possible infection or collection.
The significant pericardial effusion was not beyond the realms of an expected post-operative complication,
but was felt to be more significant than expected for the time period post op.
Pericardial effusion is a common finding in clinical practice,
be it incidental or as a manifestation of a systemic disease process.
The spectrum of effusions range from mild and asymptomatic to severe causing cardiac tamponade and cardiovascular compromise.
The onset may be insidious or rapid.
Management is dependent on haemodynamic impact,
size,
the presence of pericarditis and probable causative aetiology.
5
On CT,
generally,
pericardial effusions are of low density in the range of 0–20 Hounsfield units (HU).
When the effusion is more proteinaceous,
such as in bacterial infections,
or haemorrhagic,
its density may raise to 50 HU and more.
Inflamed pericardium may also show contrast enhancement. 5 It has been reported that large idiopathic chronic effusion (>3 months) have a 30–35% risk of progression to cardiac tamponade.6
Subacute (4–6 weeks duration) large effusions not responsive to conventional therapy and with echocardiographic signs of right chambers collapse may have an increased risk of progression according to some authors who recommend preventive drainage.7
On CT chest,
interventricular septal flattening,
interventricular septal bowing to the left or atrial collapse may reflect haemodynamic impact.
These findings need to be reported and correlated with echocardiographic or dedicated cardiovascular magnetic resonance findings.
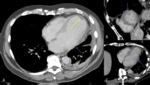
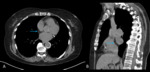
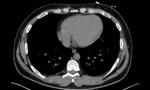
Pericardial involvement in mesothelioma.
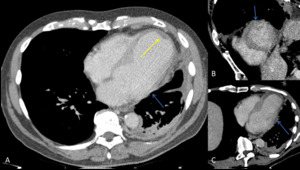
Fig. 4: Image 3: Axial CT thorax in soft tissue windows of a patient with confirmed mesothelioma. A; Pericardial nodules and thickening of the visceral layer of the pericardium. B&C: loss of subepicardial fat layer at the level of basal lateral LV wall on short axis view and horizontal long axis reconstruction.
References: Deapartment of Radiology, Liverpool Heart and Chest Hospital, UK,2019
According to the TNM 8 staging of mesothelioma,
the presence of transmural pericardial involvement in pleural mesothelioma automatically alters the patient’s tumour stage to a T4,
meanwhile non-transmural invasion classifies the tumour as T3.
It is therefore essential that the pericardium is scrutinised in patients undergoing staging for mesothelioma.
The scans performed for this role are unlikely to be ECG gated and so an example of nodular transmural pericardial involvement in known left pleural mesothelioma is included in this review.
Note the absence of epicardial fat at the level of the mid anterior wall (B) and basal inferolateral wall (C),
blue arrows and the incidental aneurysmal apical part of the left ventricle with an apical thrombus (yellow arrow).
Primary malignant pericardial mesothelioma (PMPM) is an extremely rare malignant tumour that accounts for approximately 0.8% of all mesotheliomas and 2-3% of all pericardial tumours 8,9 The majority of cases are diagnosed post mortem.10
Incidental significant pericardial findings are best assessed by a dedicated ECG-gated cardiac MRI protocol,
where haemodynamic impact and tissue plane invasion can be assessed,
and thorough tissue characterisation can be performed.
Atria
– PFO
– Myxoma
– Thrombus
– Lipomatous hypertrophy of the interatrial septum.
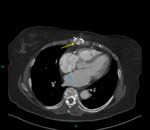
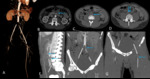
Patent foramen ovale.
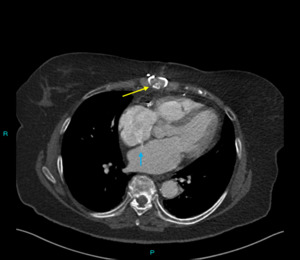
Fig. 6: Image 4. Axial Ct thorax in soft tissue window performed following slow healing of a sternotomy wound (blue arrow). There is a clear communication between the atria via atrial septal defect, and inflammatory changes around the sternotomy (yellow arrow).
References: Department of Radiology, Liverpool Heart and Chest Hopsital, UK,2019 .
This 80-year-old patient had undergone a CABG several months before.
She has a slow healing sternotomy wound,
the CT revealed delayed bone healing and evidence of infected would.
The CT scan also demonstrated a patent foramen ovale that had not been reported previously.
A patent foramen ovale (PFO) is the persistent embryonic defect in the atrial septal wall.
There is potential for right to left intra cardiac shunting across the defect and therefore paradoxical embolism into the intracranial circulation and subsequent ischaemic stroke.
The incidence of PFO in the general population is estimated at 23.7% from post mortem studies.
11
Recent literature reports an association of preoperatively diagnosed PFO with increased perioperative morbidity,
in particular ischaemic stroke within 30 days of surgery,
in patients undergoing non-cardiac surgery (3.2 % vs 0.5 %).12 It is therefore an important incidental finding to make the clinicians aware of,
the patient may require repair if there is a significant left to right shunt or concern regarding risk of stroke.
Atrial myxoma.
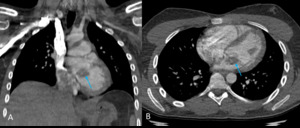
Fig. 7: Image 5: Venous phase coronal and axial slices of A CT thorax performed as part of a trauma series. The large atrial filling defect is event even on this non-cardiac gated image (blue arrows). Note central enhancement within the lesion.
References: Department of Radiology, Liverpool Heart and Chest Hopsital, UK,2019
This is a case of incidental left atrial myxoma in a young male patient that underwent a trauma series following a road traffic accident.
There was a large soft tissue density filling defect in the left atrium that was not reported at the time of the scan.
This patient presented several months later with acute abdominal and lower back pain,
acute loss of range of motion in both lower limbs with bilaterally weak femoral pulses,
pale cold feet with no palpable peripheral pulses.
Imaging at that time did not initially include chest imaging but did reveal diffuse abdominal and peripheral embolization.
Correlation was then made with previous imaging and the diagnosis of left atrial myxoma was confirmed on echocardiography.
See images figure 8.
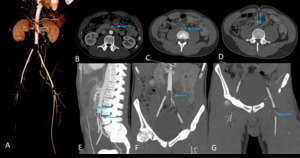
Fig. 8: Image 6: Same patient as in Image 5. CT abdomen in soft tissue windows illustrating the sequelae of multiple emboli from a left atrial myxoma. A, E-G: Digital reformats of the abdominal angiogram showing multiple central arterial filling defects. B-D: Axial slices of abdominal angiogram in soft tissue windows illustrating emboli within the SMA and a saddle embolus within the distal aorta extending proximal common iliac vessels bilaterally
References: Department of Radiology, Liverpool Heart and Chest Hopsital, UK,2019
If the incidental atrial myxoma had been detected in the patient’s initial trauma scan,
and appropriate follow up investigation/treatment had been initialised,
this could have been potentially avoided.
Atrial myxomas are rare benign primary cardiac neoplasms that most frequently occur in the left atrium.
They are almost always diagnosed incidentally.
The aetiology of cardiac myxomas is uncertain; however,
10% of cases are known to be familial.
13 Cardiac myxoma can be seen in the context of Carney complex.
Atrial thrombus.
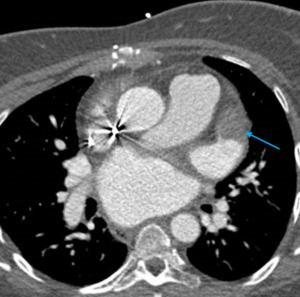
Fig. 9: Image 7: Axial soft tissue window CT thorax with non enhancing filling defect within left atrial appendage.
References: Department of Radiology, Liverpool Heart and Chest Hopsital, UK,2019
Thrombus is the most common cardiac filling defect with a predilection for the left atrial appendage (LAA) and left ventricular apex. When in sinus rhythm,
LAA thrombi are associated with mitral valve disease.
When in atrial fibrillation,
the incidence of LAA thrombi increases significantly.
14,15
The risk of intracardiac thrombus is also increased with the presence of foreign materials,
such as catheters,
pacemakers and prosthetic valve replacements.
It is reported that intracardiac thrombi are complicated by arterial embolism in up to 20% of cases,
and therefore early identification and appropriate therapy is essential.15
|
Myxoma
|
Thrombus
|
|
Enhances
|
Typically,
does not enhance
|
|
Usually attached to the interatrial septum at foramen ovale
|
Usually in LAA
|
|
Larger
|
Smaller
|
Table 1: Key features of atrial myxoma versus atrial thrombus.
Lipomatous hypertrophy of the intra atrial septum and cardiac lipoma
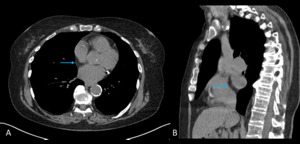
Fig. 10: Image 8: Axial and sagittal slices of CT thorax in soft tissue windows. The arrows indicate the lipomatous hypertrophy of the interatrial septum.
References: Department of Radiology, Liverpool Heart and Chest Hopsital, UK,2019
Benign cardiac tumours represent around 75 % of primary cardiac tumours.
16 The following make up the most common forms;
· Myxoma
· Lipoma
· Papillary fibroelastoma
· Angioma
· Fibromas
Lipomatous hypertrophy of the atrial septum (LHAS) was first described by Prior in 1964,
based on autopsy studies.
17 The aetiology of LHAS has not been fully understood.
The myocytes of the interatrial septum differentiate into adipocytes,
classically sparing the fossa ovalis,
which gives a characteristic hourglass configuration. LHAS is associated with obesity and occurs more frequently in elderly and females.
It is also seen more commonly in those with emphysema treated with steroid therapy,
in which there is a predisposition for mediastinal and intracardiac deposition of adipose tissue,
mediastino-abdominal lipomatosis and long-term parenteral nutrition.1LHAS are usually benign,
but >60% incidence of associated arrhythmias has been reported.19
LHAS vs Lipoma
LHAS and atrial lipoma can have very similar appearances,
however there are a few differences,
illustrated below in table 2:
|
LAHS
|
Lipoma
|
|
Not encapsulated
|
Encapsulated
|
|
Excessive epicardial fat deposition
|
Round
|
|
Infiltrates interatrial septum
|
Homogenous
|
|
Spares fossa ovalis
|
May not spare fossa ovalis
|
Table 2: Key features of LAHS versus Lipoma
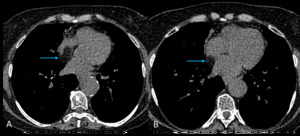
Fig. 11: Image9: Axial and sagittal slices of a non-contrast enhanced CT thorax in soft tissue windows. The arrows indicate the lipoma of the interatrial septum.
References: Department of Radiology, Liverpool Heart and Chest Hopsital, UK,2019
The majority of LHAS are asymptomatic and detected incidentally,
however there are several important complications that have been associated with these lesions: 19
Complications of LHAS:
· SVC syndrome
· Cardiac arrhythmias
· Pericardial effusion
· Heart failure
· Sudden cardiac death
It is therefore important that these lesions are picked up and reported,
and also not misinterpreted as other aetiologies.
Ventricles
– Wall infarction
– Lipomatous metaplasia
– Cardiomyopathies such as hypertrophic cardiomyopathy
– Metastases
– Ventricular wall aneurysms/pseudoaneurysms
Ventricular wall infarction.
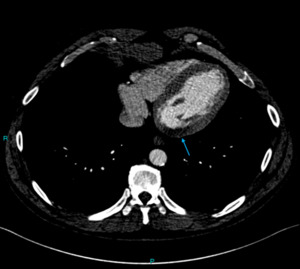
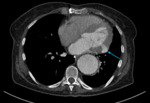
Fig. 13: Image 10: Subacute left ventricular wall infarction (blue arrow) in the territory of the left circumflex artery/right coronary artery. Soft tissue window axial CT chest.
References: Department of Radiology, Liverpool Heart and Chest Hopsital, UK,2019
This 57-year-old patient underwent a CT aorta for pre-operative planning prior to a CABG for unstable angina.
The scan demonstrated a perfusion defect in the basal inferolateral left ventricular wall indicating previous infarction in the territory of the left circumflex and right coronary arteries.
It is understood that revascularisation of transmurally scarred cardiac tissue is near futile and so the incidental finding in this patient’s case influenced the subsequent management options offered to the patient.
A silent myocardial infarction (MI) is one in which there is no acute clinical presentation; however,
the patient has increased risk of cardiovascular morbidity and mortality.
21 Diagnosis of a previous silent MI is therefore of significant clinical benefit to the patient,
especially if there was no previous diagnosis of ischaemic heart disease.
Cardiac infarctions are more clearly demonstrated on EGC gated CT and MRI,
however with multi detector CT there is improved visualisation of cardiac pathology on routine CT.
Findings suggestive of a MI on CT may well be demonstrated on non EGC gated arterial phase scans due to the perfusion defect.
Thinned and akinetic walls can be detected due to the resulting reduction in motion artefact is reduced.
This incidentally may allow visualisation of myocardial perfusion defects,
fat replacement and calcification.
Lipomatous metaplasia of the myocardium
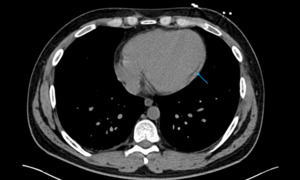
Fig. 14: Image 11: Soft tissue window axial CT abdomen (upper slice) demonstrates linear fatty metaplasia of the left lateral ventricular wall.
References: Department of Radiology, Liverpool Heart and Chest Hopsital, UK,2019
This is a case of a male in their thirties having a CT abdomen to assess for hydronephrosis.
There is a band of lipomatous metaplasia in the lateral LV wall indicating a chronic infarct.
The band was isointense to the peripheral adipose (measurement not shown).
There are several appearances of the myocardium that may indicate a chronic MI:
· Fat attenuation within the myocardium
· Focal calcifications
· Focal wall thinning
· Wall aneurysms (may signify significant previous infarct).
Intraventricular thrombus (particularly at the apex in the context of aneurysmal wall)Lipomatous metaplasia is by far the most frequent finding in up to 84 % of chronic infarcts at postmortem.22 It is described as a region of attenuation of -10 HU,
which should also be visually isointense to fat.22 Presence of lipomatous metaplasia is an adverse prognostic factor.
24
Hypertrophic cardiomyopathy
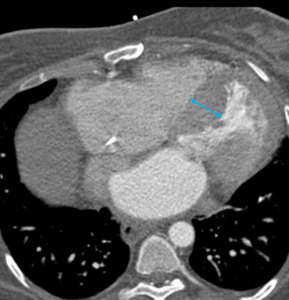
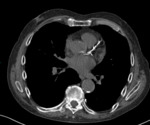
Fig. 15: Image12. Axial CT thorax in soft tissue windows, with septal hypertrophic cardiomyopathy (blue arrow). This patient has an asymmetrical septal hypertrophy of the left ventricle, they also had an incidental thrombus noted in the left atrial appendage (see image 7).
References: Department of Radiology, Liverpool Heart and Chest Hopsital, UK,2019
This patient has an asymmetrical septal hypertrophy of the left ventricle,
they also had an incidental thrombus noted in the left atrial appendage (see image 7).
Hypertrophic cardiomyopathy is a genetic disorder that occurs in the absence of any hypertrophic stimulus and is the most common cardiac cause of sudden death in athletes and previously healthy young adults.
25
There are several morphological forms that can be symmetric or asymmetric.
Asymmetric septal wall hypertrophy can lead to obstruction of the left ventricular outflow tract (LVOT) in up to a third of cases.
If severe enough,
this may affect the mitral valve due to systolic anterior wall motion (SAM).
26 The combination of anatomic narrowing of the LVOT during systole and SAM leads to dynamic subaortic obstruction.26
If severe enough,
it is possible on axial CT images to see narrowing of the left ventricular outflow tract,
in association with thickening of the mid ventricular septum.
Further evaluation with MRI should then be suggested. 26
Ventricular wall pseudoaneurysm.
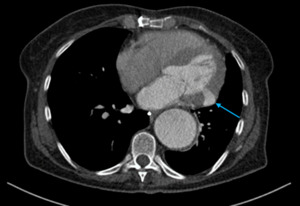
Fig. 16: Image 13: left ventricular wall pseudo aneurysm, soft tissue window axial CT chest.
References: Department of Radiology, Liverpool Heart and Chest Hopsital, UK,2019
This patient has undergone a previous frozen elephant trunk repair of the thoracic aorta with subsequent TVAR.
This scan was performed to assess for paravalvular leak.
There was an unexpected finding of left ventricular wall pseudo aneurysm.
A true aneurysm of the left ventricular wall is an area of dyskinetic,
thinned full thickness myocardium. A pseudoaneurysm is a result of rupture of the left ventricular free wall which is then contained by the overlying pericardium.
27 Differentiating between a true and pseudoaneurysm can be difficult; a pseudoanerysum will typically have a narrow neck,
whereas a true aneurysm will have a broad base.
27 Cardiac pseudoaneurysms are rare but carry considerable clinical significance.
It is estimated in the literature that 30-45 % of pseudoaneurysms will rupture,
which carries a high mortality rate.
28
The aetiology of cardiac pseudoaneurysm is mostly due to myocardial infarction,
but cardiac surgery,
blunt or penetrating trauma and infection are also important causative factors.
The most common cardiac pseudo aneurysm is of the left ventricular wall following a myocardial infarction; aortic valvular surgery and endocarditis are thought to precede pseudoaneurysms at the mitral-aortic intravalvular fibrosa.
29
Ventricular wall metastasis.
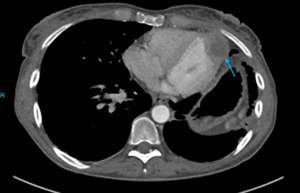
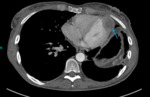
Fig. 17: Image 14: Left ventricular wall metastasis, soft tissue window axial CT chest.
References: Department of Radiology, Liverpool Heart and Chest Hopsital, UK,2019
This 56-year-old patient with non-small cell lung cancer was undergoing chemotherapy.
The CT chest was performed to assess the response to treatment.
The known lung lesion was comparable in size to previous; however,
there was a new ventricular wall metastasis that was not evident on the pre-treatment scans,
indicating there was significant disease progression despite treatment. The detection of this metastasis despite the stable appearances of the lung tumour influenced the patient’s prognosis and therefore ongoing treatment options.
Metastatic tumours that involve the heart arise various routes of spread:
1. Haematogenous - commonly melanoma,
lymphoma and sarcomas.
2.
Lymphatic - primary lung tumours.
3. Transvenous- tumour thrombus from infra diaphragmatic malignancies such as renal cell via the IVC into the right atrium,
or invasive lung cancers may propagate tumour thrombus in the SVC.
4.
Direct invasion- lung malignancies,
invasive thymoma,
lymphoma or malignant mesothelioma.
Although pericardial thickening and effusion may be non-specific,
in patients with a history of malignancy,
a malignant effusion should be considered.
The pericardium should be closely evaluated in patients with cardiac metastases. In patients with cardiac metastases and pericardial effusion causing tamponade is of significant concern and may present a life-threatening emergency.
Impending cardiac tamponade on CT may include findings of the following:30
· Collapse of the right heart chambers.
· Abnormal motion of the interventricular septum.
· Large pericardial effusion.
Coronary vessels and relevant extracardiac.
– Coronary calcification
– Sternotomy wire misplacement.
Incidental coronary calcification.
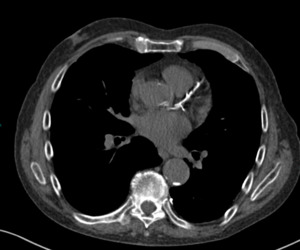
Fig. 19: Image15: Axial CT thorax in soft tissue windows with extensive calcification noted in the territory of the left main coronary artery.
References: Department of Radiology, Liverpool Heart and Chest Hopsital, UK,2019
Previous studies have identified that more than half (52-66%) of all CT chests performed have at least one incidental finding.
The most common of which is of coronary artery disease (CAD),
with a large proportion that goes unreported (51-80%).
301,32
Cardiovascular disease is a leading cause of death worldwide and accounts for up to 20% of all deaths in Europe annually.
33 The detection of CAD in patients undergoing non-cardiac surgery or cancer therapies would be beneficial to the patient as it may alter therapies or management offered.
It could also potentially reduce the morbidity and mortality associated with CAD if detected before the patient becomes symptomatic and should therefore be reported if present incidentally on CT imaging.
Sternotomy wire misplacement.
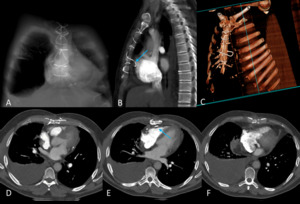
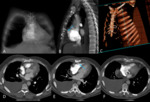
Fig. 20: Image 16. A Coronal Thorax MPR 50mm demonstrating displaced sternotomy wire. B; lateral Thorax MPR at 20mm demonstrating displaced sternotomy wire. C: Oblique 3D reconstruction of thoracic cage depicting sternotomy wires positioning. D-F: Displaced sternotomy wire in close proximity to the right ventricular free wall (blue arrow) with thickening of the pericardium and moderate right pleural effusion.
References: Department of Radiology, Liverpool Heart and Chest Hopsital, UK,2019
This CT pulmonary angiogram was performed at 13 weeks post CABG because the patient had ongoing SOB and pleural effusion (demonstrated on plain radiograph) who was D dimer positive.
The CT reconstructions clearly demonstrate the misplaced sternotomy wire that was closely associated with the pericardium.
While there is potential to mistake the wire for calcification (see Image 13 E) and therefore dismiss it,
it is important to correlate with previous images and reformat using the technology available to view in other planes.
The patient subsequently had an MRI study which showed a haemodynamically significant pericardial constriction with active pericarditis (images not shown).
Post-operative instability of the sternum is a well-known condition that occurs in 1–3% of patients following sternotomy.34 Complications that specifically refer to the wires make up to 90% of underlying causes,
mainly through displacement in the vertical line or mal-rotation,
while in about 20% of wire-complications they fracture.34,35