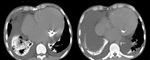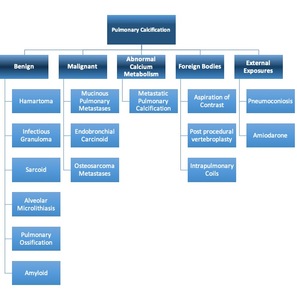
Table 1: Causes of pulmonary calcification
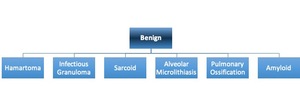
Table 2: Benign causes of pulmonary calcification
Starting with the benign causes of pulmonary calcification,
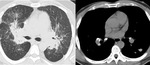
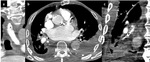
one of the most commonly encountered and incidentally found pulmonary nodules are granulomas (Figure 1).
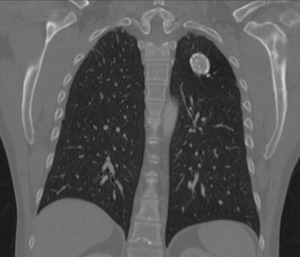
Fig. 1: Left upper lobe calcified granuloma
Granulomas are not a specific disease entity unto themselves,
rather,
an end point of an infective or inflammatory process which forms focal aggregation of macrophages due to infection or inflammation.
These are most commonly encountered as small,
well-defined and calcified nodules requiring no further investigation or follow-up as they are often clinically of no significance. [1]
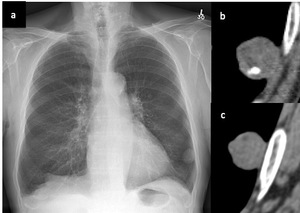
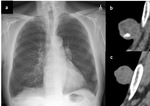
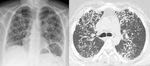
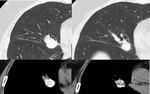
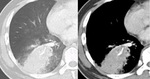
Fig. 2: Left lower lobe subpleural shown here on chest x-ray (a) and on CT imaging to contain calcification (b) and fat (c) in keeping with a hamartoma.
These appearances are typical for a hamartoma.
The calcification can be punctate,
“popcorn” or absent.
Prior imaging can be very useful to confirm interval stability and the longstanding chronic nature and therefore often do not require ongoing investigation.
Fat attenuation needs to be demonstrated to confirm the diagnosis.
Thin sections of 1-1.5 mm are also used to study differential density to assess for fat density because otherwise partial volume effects can preclude accurate assessment. [2]
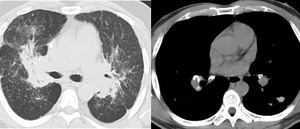
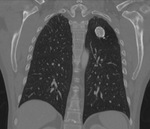
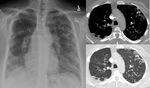
Fig. 3: Lung and mediastinal windowed images of a patient with bilateral perilymphatic and spatial nodularity and bihilar and mediastinal adenopathy with calcification, in keeping with sarcoidosis.
Sarcoidosis is a non-caseating granulomatous disease affecting multiple systems including the lung and mediastinum,
characteristically producing the Garland triad of adenopathy involving right paratracheal,
right hilar and left hilar nodes seen in chest x-ray,
often containing calcification.
On CT multiple diffuse perilymphatic lung nodules can be seen with corresponding nodular septal thickening.
Biochemical correlation with serum angiotensin converting enzyme,
hypercalcaemia and hypercalciuria can aid in diagnosis,
with staging and follow up imaging via chest x-rays or high resolution CT.
Treatment consists of cortisteroids and is only required in those with active disease with a majority of cases resolving within in 1-2 years. [3]
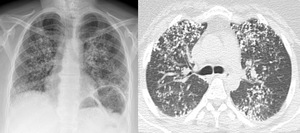
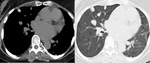
Fig. 4: Chest x-ray demonstrating upper lobe predominant nodularity and CT chest slice showing alveolar calcification as seen in pulmonary alveolar microlithiasis.
Pulmonary alveolar microlithiasis is a rare entity thought to be cause by a genetic mutation causing inactivation of a sodium-dependant phosphate cotransporter found mainly in alveolar cells leading to accumulation of phosphate in the alveolus and calcium phosphate microliths formation.
The calcification can become confluent andsignificant morbidity with no known treatment. [4]
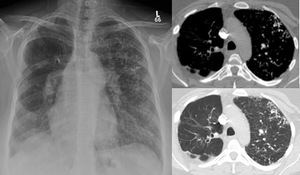
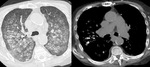
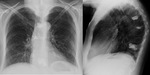
Fig. 5: Chest radiograph shows right lung allograft and the left native lung has multiple multifocal nodules. CT soft tissue and lung windows showing calcification diffuse native left lung calcified nodules.
Pulmonary ossification is a rare finding and It can be idiopathic or secondary to chronic lung,
cardiac or systemic disorders. [5] The above figure depicts idiopathic pulmonary ossification within the native left lung which demonstrates the presence of mature bone in alveolar and/or interstitial spaces,
which can be localised or diffusely disseminated throughout the lung parenchyma.
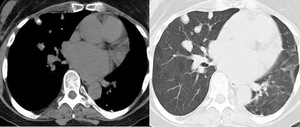
Fig. 6: Calcified and non calcified pulmonary nodules on CT chest in keeping with amyloidosis, confirmed on histology.
Amyloid lung disease is a rare disease entity within the lungs most commonly seen as its nodular form with multifocal well defined perilymphatic nodules and lower lobe predominant masses which can calcify with diffuse airway infiltration and narrowing.
Nodular amyloid lung nodules can mimic a wide range of diseases and therefore tissue diagnosis is essential. [6]
Table 3: Malignant causes of pulmonary calcification.
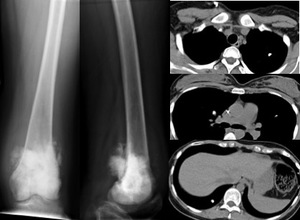
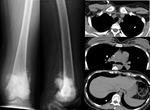
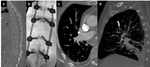
Fig. 7: AP and lateral radiographs of the left femur demonstrating an ill defined sclerotic bony lesion in the distal femur in keeping with an osteosarcoma. The Ct chest images show multiple ossified osteosarcoma pulmonary metastases.
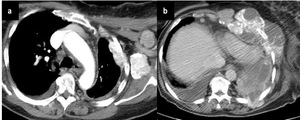
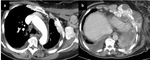
Fig. 8: Two CT chest slices. Figure 8a shows avidly enhancing and partially calcified left pleural metastasis and left axillary nodal metastasis. Figure 8b shows a left basal multiloculated malignant effusion with chest wall invasion and avidly enhancing thickened, nodular pleura.
A number of malignancies are recognised to cause calcified pulmonary metastases including mucinous and serous carcinomas,
sarcomas and thryoid cancer.
Figures 7 and 8 above show metastases from osteosarcoma and mucinous ovarian carcinoma.
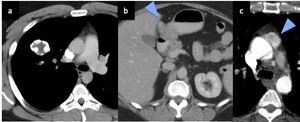
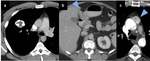
Fig. 9: Carney’s triad: (a) shows a right lung pulmonary chondroma with peripheral calcification, (b) shows an arrow indicating towards a pyloric gastrointestinal stromal tumour and (c) shows an arrow pointing towards an extra adrenal mediastinal paraganglioma.
Figure 9 shows Carney’s triad,
a rare tumour syndrome consisting of an extra adrenal (mediastinal in this case) paraganglioma, right lung pulmonary chondroma with peripheral calcification and a pyloric gastrointestinal stromal tumour.
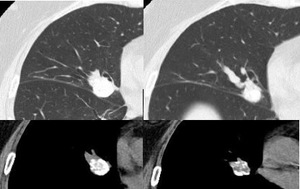
Fig. 10: Lung and soft tissue windows of two slices through a peripherally calcified right middle lobe carcinoid tumour.
Bronchial carcinoid tumours are neuroendocrine tumours are well circumscribed,
often calcified round or ovoid lesions most commonly endobronchially occurring or closely related to the larger segmental airways. Central tumour can lead to bronchial obstruction and associated pneumonia,
atelectasis and bronchiectasis with clinical features including cough and wheezing,
whereas more peripheral tumours can be asymptomatic and incidentally discovered. [7]
Table 4: Abnormal calcium metabolism
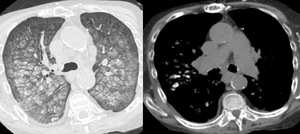
Fig. 11: Centrilobular nodules and ground glass of calcific attenuation in keeping with metastatic pulmonary calcification.
The deposition of calcium in lung parenchymal in causes of serum hypercalcaemia can cause dense calcific centrilobular nodules and ground glass opacification,
often upper lobe predominant (thought to be due to the higher ventilation to perfusion ratio in the apices therefore less carbon dioxide partial pressures and more alkaline environment) with perifissural and subpleural sparing as seen in figure 11.
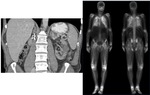
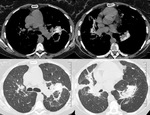
Causes of severe serum hypercalcaemia include chronic renal failure (figure 12 below),
hyperparathyroidism,
extensive bone metastases,
hypervitaminosis D and Milk- Alkali syndrome. [8]
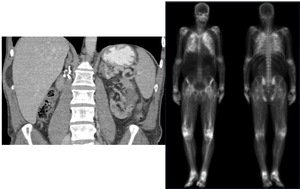
Fig. 12: Coronal CT upper abdomen showing surgical clips in the right nephrectomy bed with and atrophic left kidney and bone scan demonstrating diffuse lung uptake in metastatic pulmonary calcification.
Table 5: Foreign bodies causing pulmonary calcification or high density
Foreign bodies are another common cause of high attenuation within the lungs and careful clinical history is often key to illuminating the causative factor.
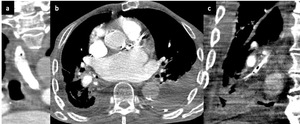
Fig. 13: CT Chest demonstrating a contrast filled oesophagus (a). Oral contrast has been aspirated and fills the the right lower lobe bronchi (b) and coronal reformat of the right lower lobe bronchi shows contrast with locules of gas, with distal atelectasis and consolidation (c).
Figure 13 shows oral contrast within the oesophagus and also oral contrast aspiration,
filling the right lower lobe bronchi with locules of gas,
with distal atelectasis and consolidation,
and small reactive pleural effusions.
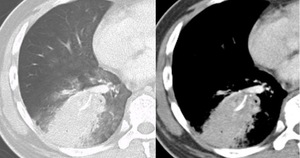
Fig. 14: Lung and mediastinal window CT images of the right lower lobe a high attenuation focus within the right lower lobe bronchus with adjacent dense consolidation. This turned out to be a obstructing chicken bone with obstructive sequelae.
Figure 14 shows aspiration of a high density foreign body within the right lower lobe bronchus,
namely a chicken bone in this case,
with associated airways obstruction and downstream collapse and consolidation.
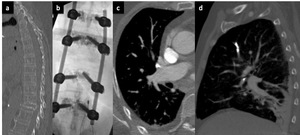
Fig. 15: (a) Sagittal thoracic spine on bone windows shows a lower thoracic vertebral collapse with fluoroscopy of spinal fixation (b) and very dense material within the upper lobe arteries on CT (c) and (d).
Figure 15 shows lower thoracic vertebral collapse,
fluoroscopy of spinal fixation and vertebroplasty and very dense material which is denser than contrast within pulmonary arteries on CT,
which turned out to be cement emboli.
Figure 16 is a very similar case of cement emboli post vertebroplasty which can be appreciated on the chest x-ray with tiny parenchymal high density emboli and right hilar high densities.
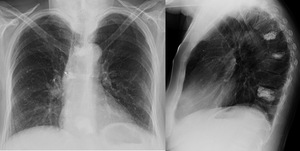
Fig. 16: Chest x-ray and lateral thoracic spine x-ray showing high density parenchymal foci and vertebroplasty.
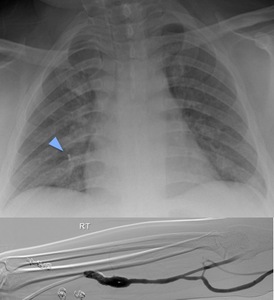
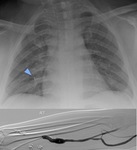
Fig. 17: Chest x-ray with arrow indicating a metallic coil and peripheral angiogram of the right arm with coil placement.
The CXR in figure 17 demonstrates a right midzone metallic density consistent with a coil but in a patient with no history of pulmonary vascular abnormalities or pulmonary coil embolisation.
Further investigation of previous imaging showed a peripheral angiogram of a right arteriovenous fistula with coiling of a right cephalic large venous collateral during which a coil was dislodged and was propagated to the lungs.
Table 6: External exposures causing high attenuation within the lungs.
Pneumconioses is an umbrella term of lung diseases caused by inhalation of dust particles often related to occupational lung disease.
The resultant tissue reaction which can be fibrotic or non fibrotic but due to the body’s attempt to clear the particles through the lymphatic system,
tend to produce high attenuation perilymphatic nodules but can be complicated by the associated hypersensitivity pneumonitis and the corresponding centrilobular pattern of nodularity.
Clinical history is therefore key for these diagnoses. [9]
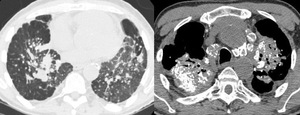
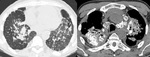
Fig. 18: Unenhanced CT Chest demonstrating calcified perhilar progressive massive fibrosis with associated calcification and characteristic eggshell calcification of the subcarinal lymph node.
Figure 18 demonstrates a case of silicosis with massive pulmonary fibrosis and characteristic eggshell calcification of the mediastinal nodes.
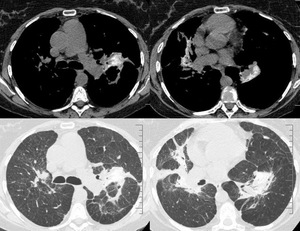
Fig. 19: Talcosis demonstrated on mediastinal and lung windows with fine high density nodularity and fibrosis.
Figure 19 demonstrates talcosis,
another type of pneumoconiosis with very fine multifocal nodulariy and dense consolidation with progressive massive fibrosis and emphysematous destruction of the lung parenchyma.
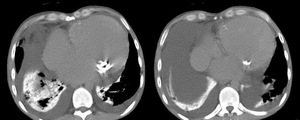
Fig. 20: CT chest slices demonstrating cardiomegaly, mitral annulus calcification and dense parenchyma of the right lower lobe in keeping with amiodarone deposition.
Lastly figure 19 shows a CT Chest with high attenuation material related to the pleura,
calcified mitral valve and annulus,
right lower lobe predominant dense parenchymal high attenuation and small bilateral small pleural effusions.
In the context of cardiomegaly,
these appearances represent pulmonary deposition of amiodarone which can also accumulate within the liver be seen to cause a diffusely high attenuation hepatic parenchyma.