Strategy for differentiation
Below are strategies for differential diagnosis.
- Analysis of vascularity in low attenuation area
- Perform Expiratory Imaging
- Analysis the Pulmonary artery
- Analysis the Airways
- Analysis the Heart
- Find additional clues
When you face to mosaic pattern lung attenuation,
to determine which portion of the lung parenchyma is abnormal is most important point and that is the first step to do.
Analysis of vascularity in the low attenuation area is the first thing to do.
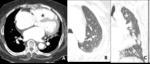
Small airway disease and pulmonary vascular disease shows decreased vascularity in the low attenuation area (Fig.
7).
Small airway obstruction or fibrosis leads to hypoxic vasoconstriction.
And attenuated peripheral pulmonary vascularity area of low attenuation lung means segmental or sub segmental arteries are narrowed or occluded.
But the other cause,
parenchymal disease (GGO) shows uniform vascularity of the lung.
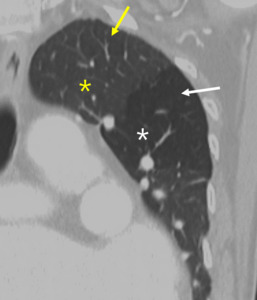
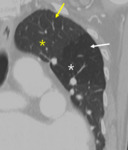
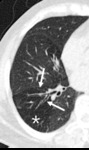
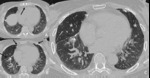
Fig. 7: Decreased size of the vasculature (arrow) is prominent in the hypoattenuated region (white star) of the left upper lobe. Compare the equivalent size of vasculatues in the normal attenuated lung (yellow star) of the apical lung.
To differential diagnosis small airway disease and pulmonary vascular disease.
Expiratory CT is best method to differential diagnosis of small air way disease.
Common indications for additional expiratory imaging are worsening symptom in patients with known bronchiolar disorder,
risk factor for constrictive bronchiolitis and incidental finding of mosaic attenuation without clear cause.
In case of small airway disease,
air cannot readily escape (air trapping).
Attenuation of involved segment remains relatively unchanged or much more pronounced difference in attenuation between normal and abnormal areas (Fig.
8).
Without small airway disease,
lung attenuation on expiratory CT shows relatively diffuse increase in attenuation and appears grayer.
Some pitfalls are existing.
Some degree of heterogeneity is expected in normal individuals.
Areas of lobular air trapping may appear 40-50% in normal patients.
Otherwise,
air trapping involving more than one segment usually indicated underlying pathologic causes.
Inadequately performed due to lack of patient effort or poor coaching could be happened.
To check the adequate effort,
you need to check posterior wall of trachea and main-stem bronchi should bow inward due to increased intrathoracic pressure.
Occasionally,
air trapping can be seen in pulmonary vascular disease or lung parenchymal disease.
CTEPH is due to reactive bronchoconstriction.
Hypersensitivity pneumonitis is due to mechanism of disease primarily involve respiratory bronchioles,
and DIP is due to coexistent RB.
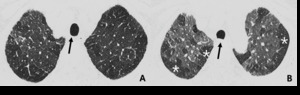
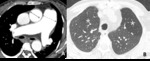
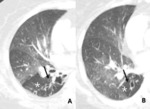
Fig. 8: 72-year-old man with constrictive bronchiolitis obliterans (A) on inspiratory CT scan, inconspicuous hypoattenuated areas are defined both upper lobes. (B) Expiratory CT scan at the same level, comparing anterior bowing of posterior wall of trachea (arrow) reveals multifocal areas of hypoattenuated region conspicuously (white star).
Analysis pulmonary arteries
The size and morphology of the pulmonary arteries are helpful clues to differentiate vascular causes of mosaic attenuation.
Two main cause of the pulmonary vascular disease is pulmonary artery hypertension (PAH) and Chronic thromboembolic pulmonary hypertension (CTEPH).
Common findings are dilatation of segmental pulmonary artery (segmental artery-to-bronchus ratio > 1 in 3 or more lobes),
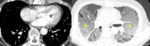
regional variation in caliber of subsegmental pulmonary arteries and abrupt vessel narrowing. The findings of PAH are abruptly tapering or corkscrew vessels,
central pulmonary artery enlargement (not specific) and pulmonary artery-to-aorta ratio > 1 (Fig.
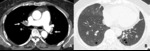
9). The findings of CTEPH are adherent thrombus,
abrupt occlusion of pulmonary arteries,
luminal irregularities with eccentric wall thickening,
abrupt caliber change (due to recanalization) and webs or bands (Fig.
10). In case of small air-way disease pulmonary artery size and morphology are usually normal.
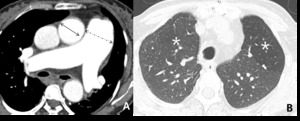
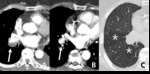
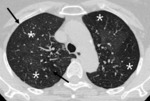
Fig. 9: 58-year-old woman with pulmonary arterial hypertension (PAH). (A) Enhanced CT scan shows more dilatation of the pulmonary trunk in comparison of the diameter of aorta at the level of bifurcation of both right and left main pulmonary arteries. (B) Area of hypoattenuated lung (white star), larger than lobular extent, can be considered as vasoconstriction of PAH, comparing of pulmonary vasculatures within hypoatttenuated area to surrounding normal lung.
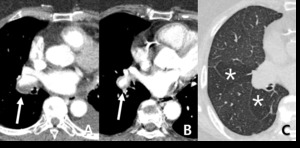
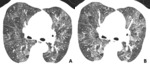
Fig. 10: 73-year-old woman with chronic thromboembolic pulmonary hypertension (CTEPH). (A,B) Crescent filling defects within dilated right lower lobar arteries (arrow), as chronic thromboembolic pulmonary arterial hypertension. (c) hypoattenuated area of lung (star), more extensive than lobular region caused by vascular origin rather than airway abnormality.
Analysis airways
Abnormality of the large airway is bronchi wall thickening and bronchiectasis.
Distribution of abnormal airway helps differential diagnosis.
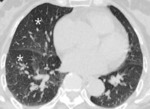
If abnormal airways are distributed in areas of low attenuation,
it suggests small airway disease such as constrictive bronchiolitis (Fig.
11,12).
Otherwise,
dilated airways seen in the areas of increased attenuation suggest parenchymal disease such as organizing pneumonia,
organizing phase of diffuse alveolar damage,
NSIP.
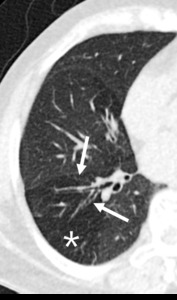
Fig. 11: 60-year-old woman with bronchiectasis. Hypoattenuated areas (star) of lung distal to segmental broncheal wall thickening and mild dilatation can be explained as combined small airway disease of constrictive bronchiolitis.
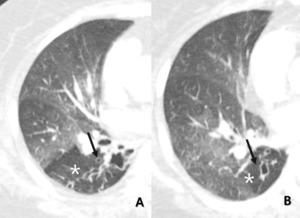
Fig. 12: Other, 71-year-old woman with bronchiectasis. Subsegmental or larger than lobular hypoattenuated region (star) of the lung reveals combined bronchiectasis (arrow) as indirectly visualized.
Direct visualization of small airway is not possible on normal condition.
Centrilobular nodules or tree-in-bud nodules are good clue of small airway disease (Fig.
13).
If there finding are accompanied to the mosaic attenuation,
you can consider small airway disease such as infectious bronchiolitis,
which is direct visualization of exudative form of bronchiolitis.
However,
hazy ill-defined centrilobular nodule with diffuse GGO could be a mixed decreased and increased pattern of lung attenuation.
These can be due to hypersensitivity pneumonitis,
smoking-related lung disease (respiratory bronchiolitis,
RB-ILD,
DIP).
In case of pulmonary vascular disease,
airways are usually normal.
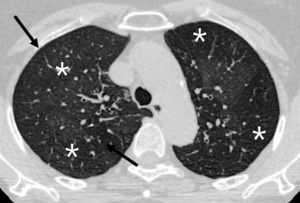
Fig. 13: 69-year-old woman with small airway disease, infectious bronchiolitis.
Well defined centrilobular nodules (arrow) are accompanied with lobular hypoattenuation (star) is directly visualized exudative form of small airway disease.
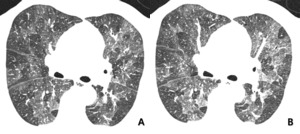
Fig. 14: 53-year-old man with subacute hypersensitivity pneumonitis. (A) patchy or mixed density differences reflecting both interstitial infiltrates and coexiting small airway disease. (B) End-expiratory CT showing enhancing or more prominence of patchy density differences, reflecting small airway involvement.
Analysis the heart
Features of right heart strain are dilated ventricle and bulging of interventricular septum.
IVC dilatation with contrast reflux to hepatic vein indicated RV failure.
These findings imply pulmonary hypertension.
Therefore,
mosaic attenuation with RVH could be due to pulmonary vascular cause,
PAH (Fig.
15).
On the other hands,
left vertricle hypertrophy or left atrium dilatation with mosaic pattern implies pulmonary edema,
more likely (Fig.
16).
In case of small airway,
the heart features are non-specific.
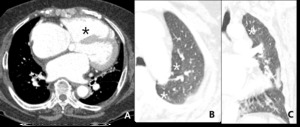
Fig. 15: 77–year-old woman with hypertrophic cardiomyopathy. Right ventricle hypertrophy and dilated right ventricle (black star) and bulging of interventricular septum reflecting pulmonary hypertension. Mosaic attenuation (white star) with RVH can be due to PAH.
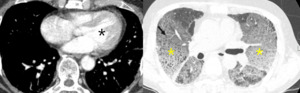
Fig. 16: 75-year-old man with pulmonary edema due to left ventricle hypertrophy (black star). Mosaic attenuation cannot be explained as small airway disease.
Additional clues
In the case of CTEPH,
peripheral fibrotic lesion on the low attenuation area could be seen (Fig.
17).
It is result from peripheral scarring form the prior lung infarction.
In the cases of small airway disease and PAH,
fibrosis is not common.
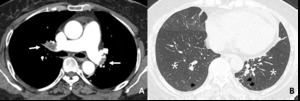
Fig. 17: 59-year-old woman with CTEPH. (A) Thromboembolic filling defects (arrow), bilateral proximal lower lobar arteries.
(B) Hypoattenuated areas of bothlungs more extensive than lobular in distribution and linear scarrings, peripheral area of both lower lobe (arrowhead), reflecting evidence of old infarction. Hypoattenuated area, non-lobular, subsegmental in distribution and old infarction scars imply mosaic attenuation caused by vascular origin.
Connective tissue disease,
such as rheumatic arthritis could present lung disease as small airway diseases,
such as constrictive bronchiolitis (Fig.
18).
The findings of arthritis in the joint space such as shoulder joint or rib joint could be a clue of the small airway disease.
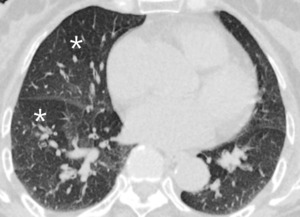
Fig. 18: 67-year-old woman with RA. On expiratory CT scan notes extensive patchy air trapping (white star) reflecting constrictive bronchiolitis.
Small airway disease (constrictive bronchiolitis) is common complication in lung transplantation patient as a chronic rejection.
Almost half of patients occur in 5 years.
Findings of lung transplantation,
such as clamshell median sternotomy wire could be a clue of the small airway disease (Fig.
19).
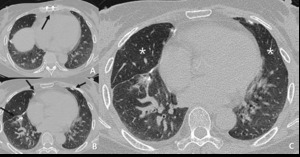
Fig. 19: 60-year-old woman who received lung transplantation 2 weeks ago. (A.B) Surgical clips are noted (arrow). (C) Areas of hypoattenuation (star) are seen in the right middle lobe, reflecting bronchiolitis obliterans as an acute rejection phenomenon.
References: courtesy of Dr. Yoon-young Choi.
To differential diagnosis of parenchymal disease.
Ground glass opacity (GGO) is increased attenuation in the lung with preserved bronchial and vascular markings.
This finding is non-specific sign with a wide etiology.
Acute causes of GGO are pulmonary edema,
pulmonary hemorrhage,
infections (Pneumocystis pneumonia,
Cytomegalovirus pneumonia),
diffuse alveolar damage,
exudative phase (ARDS,
acute interstitial pneumonia) and acute eosinophilic pneumonia.
Subacute to chronic causes are organizing pneumonia,
hypersensitivity pneumonia,
infections (PCP),
diffuse alveolar damage,
organizing and fibrotic phase,
nonspecific interstitial pneumonia
To differential diagnosis of mixed increased and decreased attenuation disease.
Common mixed diseases are hypersensitivity pneumonia,
respiratory bronchiolitis,
desquamative interstitial pneumonia (DIP),
pulmonary capillary hemangiomatosis.
It is difficult to differential diagnosis by imaging.
So other finding such as emphysema or smoking history are needed to differential diagnosis.