The most important predictors of malignancy are patient risk factors (smoking status,
prior history of malignancy) and nodule size,
chronicity,
density and growth
When a nodule is identified,
all previous CT scans should be reviewed to determine its chronicity. CT can also provide information about the location,
size,
shape,
margins and attenuation characteristics (12).
Size and growth rate represent the main indicators for determining the nature of a pulmonary nodule (13,
14). Nodule size closely relates to likelihood of malignancy in a nonlinear relationship (2).
In the NELSON trial,
the probability of cancer in a nodule under 5 mm was 0.4%.
In nodules 5 to 10 mm,
the probability was 1.3%,
and in nodules 10 mm or greater,
the probability rose to 15.2%,
with nodules at least 20 mm having a probability of cancer ranging in the literature from 64-82% (10,
14).
The risk of nodules in never-smokers is not as well known,
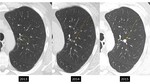
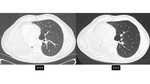
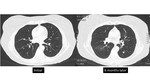
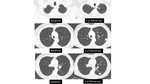
however the likelihood of cancer would be less compared to this higher risk population. It should be noted that the usual size rules do not apply to solid pulmonary nodules under 6 mm which are new,
which have a risk of approximately 3% (4,
10) [figure 1].
Assessment of a nodule’s growth is also important. In one study of 561 screening participants (15),
growth on at least two CT scans increased the malignancy rate from less than 1% to 75%. In this study,
the mean diameter of malignant nodules on detection was 12.8 mm (range of 3 to 36.4 mm).
However,
18% of malignant nodules were less than or equal to 4 mm in diameter when first seen.
The type of pulmonary nodule (solid,
part-solid,
ground glass) is another important factor.
ELCAP was an early lung cancer screening study which assessed malignancy rates of different types of nodules.
In tissue-proven subcentimeter nodules,
a higher rate of bronchogenic carcinoma was found in subsolid compared to solid nodules (16).
Similarly,
in McWilliam’s study (2),
part-solid nodules as compared with solid nodules have a greater risk,
even at sizes of less than 10 mm.
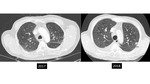
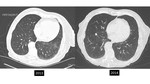
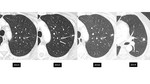
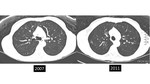
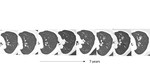
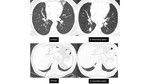
Small groundglass nodules (under 6 mm) often represent atypical adenomatous hyperplasia (AAH) [figure 14].
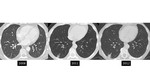
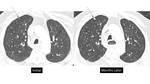
Approximately 10% of groundglass nodules less than 6 mm in diameter will grow and 1% will develop into invasive adenocarcinoma or minimally-invasive adenocarcinoma,
with a mean period between baseline CT screening and appearance of a solid component of 3.6 years (17) [figures 15-17]. Therefore,
in the case of tiny pulmonary nodules,
it is important to obtain CT with thin slices to accurately assess attenuation of a nodule.
Because of volume averaging,
CT with thick slices (5 mm) can make a solid nodule appear as groundglass attenuation.
In addition to size,
growth and density,
other important imaging characteristics include location and morphology. In a study of high-risk asymptomatic patients undergoing CT screening,
a larger proportion of cancers were observed in the upper lobes as opposed to the right middle and lower lobes (2).
Spiculation is also a significant predictor of lung cancer.
In one study,
it was found that a malignant diagnosis was five times more common for nodules with spiculated margins (LR 5.5),
whereas malignancy was five times less likely for smooth or polygonal margins (LR 0.2) (12).
An oval or triangular perifissural nodule represents a minimal risk of lung cancer and probably does not require CT follow-up (2).
These are thought to represent intrapulmonary lymph nodes and can wax and wane as they react to inflammatory change in the lungs.
A benign diagnosis can also be established when there is a characteristic benign pattern of calcification including diffuse,
central,
laminated and popcorn.
Evaluating the growth,
density and margins of tiny nodules is challenging
However,
the traditional approach to the assessment of pulmonary nodules,
including evaluation of growth,
density,
and margins,
is often not useful in tiny nodules.
Intrinsic error associated with measurement and variability are more evident in this subset of nodules.
In one retrospective study of solid noncalcified nodules < 2 cm using manual measurements by means of an electronic caliper (largest transverse diameter),
the best intra-reader repeatability coefficient (5% error rate) was 1.32 and among readers was +/- 1.73.
Clinically,
this means that using the 1D method,
measurement values <1.32 mm and < 1.73 mm cannot be distinguished from errors (14,
18).
Even a doubling of volume is below the resolution of CT (eg,
diameter increase from 4 to 5 mm).
In addition,
although nearly all carcinomas exhibit growth over a follow-up period of two years,
it may be difficult to detect enlargement of small lepidic adenocarcinomas because of their slow growth.
Volume doubling times among lung cancers that manifest as pure groundglass nodules (mean 813 days) and part-solid nodules (mean 457 days) are significantly higher compared to solid nodules (mean 149 days) (19).
Density and margins can also be difficult to assess in sub-6-mm nodules.
Because of these limitations,
the clinical context should not be overlooked.
In the case of tiny nodules,
patient risk factors including age,
smoking history,
and prior malignancy are more important predictors of malignancy (2,
5,
9,
13,
20).
Although there is a lack of evidence regarding cancer probability in tiny nodules in patients who are at low clinical risk,
in general the risk of lung cancer in patients who have never smoked is significantly lower,
with a relative risk of approximately 0.15 in the United States compared with heavy smokers (20).
Given that the risk of cancer in solid nodules under 6 mm in heaver smokers is less than 1%,
it is reasonable to assume an even lower risk in non-smokers (13).
However it is important to keep in mind that approximately 10 to 15% of all lung cancers arise in never smokers (20).
In addition,
the follow-up of groundglass nodules is much less dependent on smoking history,
as AAH and AIS are increasingly recognized in never-smokers.
Guidelines: are they as useful for tiny nodules?
Follow-up CT is the only practical method for evaluating the significance of a tiny nodule.
Although the best approach remains a challenge,
data from lung cancer screening studies have helped indicate what imaging strategies may be reasonable.
In Lung-RADS (and LU-RADS and NELSON protocols),
tiny nodules are labelled as a negative screen (21-23). The Fleischner Society guidelines (13) also states that follow-up is either optional or not indicated in this subset of nodules.
Optional follow-up may be appropriate when there are certain features that raise suspicion.
For example,
follow-up CT at 12 months may be warranted if a tiny nodule has a suspicious morphology,
upper lobe location,
or both depending on patient comorbidity and preferences,
as these features may further increase cancer risk into the 1-5% range (13).
These guidelines are not intended for patients with known primary malignancy (who are at risk for metastases) or immunocompromised patients,
in which case management should be according to the clinical situation.
In addition,
because lung cancer is rare in adults younger than 35,
the guidelines are not appropriate for these patients.
In this situation,
the clinician should tailor management on a case-by-case basis,
recognizing that infectious causes are more likely than cancer and minimizing the use of serial CT (13). Because new solid nodules under 6 mm have a higher risk of malignancy (4,
10),
they generally warrant a more aggressive follow-up strategy.
For example,
the Lung-RADS guidelines recommend a repeat CT chest in 6 months for new solid nodules 4 mm to < 6 mm (categorized as probably benign).
It has been suggested that this management strategy should also be used in routine clinical practice for high-risk patients outside of CT screening programs,
if the nodule is known to have newly developed within 1-2 years (4).
There remain gaps in our knowledge,
however. Experience has shown that tiny solid nodules,
when malignant,
seldom advance in stage over 12 months (13,
24).
For focal GGO’s,
particularly when small,
2 years of stability may not exclude adenocarcinoma-in-situ or the potential to become an invasive lesion.
If these nodules may take years to demonstrate a worrisome CT appearance,
then research following patients at most for 4 years,
often only 2,
may not be sufficient.
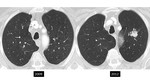
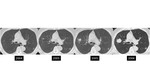
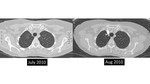
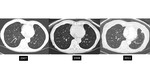
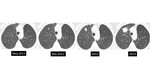
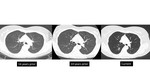
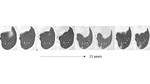
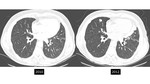
There is also lack of data regarding when to follow tiny nodules in patients with known malignancy [figures 18-22]. Another limitation of the current approach is that nodule risk calculators only apply to nodules seen on a baseline scan.
An additional challenge is the high prevalence of pulmonary nodules and the low incidence of lung cancer.
Decreasing the number of repeat CT’s for nodules would reduce harms associated with exposure to radiation,
costs,
and diagnostic procedures,
but may also decrease the sensitivity for detecting cancerous nodules.
Another issue is the effect of imaging on patient anxiety.
In one study,
approximately half of participants reported psychological distress in connection with having to wait for the results of the CT scan and dreading those results (25).
Therefore,
the frequency and duration of follow-up should take into account both the patient’s risk for developing lung cancer as well as individual preferences.