IPF is a chronic fibrotic lung disease limited to the lungs characterized by patchy,
peripheral lobular fibrosis that progresses to “honeycombing” and end-stage fibrosis over the course of years.
Updates to diagnostic guidelines for IPF
The updated ATS/ERS/JRS/ALAT guideline and Fleischner Society statement reach similar conclusions and recommendations with only small differences.
We review the main points of the updated diagnostic guidelines for IPF.
1- Clinical aspects
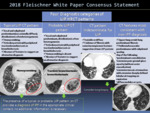
Both documents emphasize the pivotal role of the clinician in evaluating patients suspected to have IPF,
particularly excluding other forms of interstitial lung disease (ILD).
It is important to identify the “clinical context of IPF” which includes a patient older than 60 years,
generally male,
with progressive exertional dyspnea and/or dry cough in the setting of bilateral inspiratory crackles.
Additional important clinical information is the exclusion of significant environmental or medication exposure and no evidence of connective tissue disease (CTD).
These features increase the clinical probability of IPF.
A systematic assessment for CTD is necessary for a patient with suspected IPF,
particularly in adults younger than 60 years.
In every patient with fibrosing ILD,
identification of exposure to antigens that might result in hypersensitivity pneumonitis is important.
Patient with a history of occupational or medication exposures should be interpreted in a multidisciplinary discussion.

Fig. 1: Clinical assessment according to Fleischner guideline. It is essential to determine the clinical context of IPF and to exclude alternative causes of fibrosing ILD.
2- Imaging interpretation
2.1 Evaluation of imaging quality- HRCT technique
Both documents emphasize the relevance of high-quality HRCT images in the evaluation of patients suspected to have IPF.
Recommendations for CT protocol are summarized:
Volumetric CT acquisition is preferred to sequential CT scanning because it improves the detection of all abnormalities.
Optimal quality requires the thinnest collimation (<2 mm) and high spatial resolution reconstruction.
Images should be obtained at full inspiration in supine position.
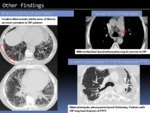
Expiratory images (end-expiration volumetric or sequential acquisition) can be useful to identify air trapping,
suggesting an alternative diagnosis as chronic hypersensitivity pneumonitis (CHP) or CTD.
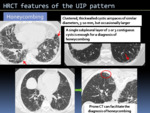
Prone CT (volumetric or sequential acquisition) can be added and it is particularly useful in clearing depending opacifications on supine CT images and also to facilitates the diagnosis of honeycombing.
ATS/ERS/JRS/ALAT guideline recommend scanning to evaluate acute respiratory worsening in a patient with a known ILD.
In this clinical context,
the goal is to rule out acute pulmonary embolism or an acute exacerbation,
performing a chest CT angiography either alone or in addition to a noncontrast HRCT.

Fig. 2: HRCT scanning recommendations for the accurate diagnosis of IPF.
2.2 Interpretation of key HRCT features
When IPF is considered in the clinical context,
the radiologist must indicate if the UIP pattern is present and the level of confidence it has in the presence of this pattern.
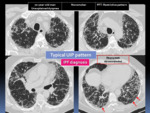
HRCT features seen in UIP pattern include honeycombing,
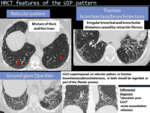
traction bronchiectasis and traction bronchiolectasis,
reticular pattern and ground-glass opacities.
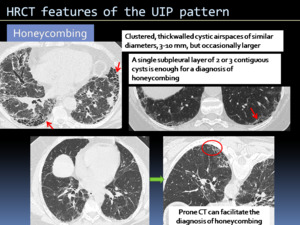
Fig. 3: HRCT features of the UIP pattern.
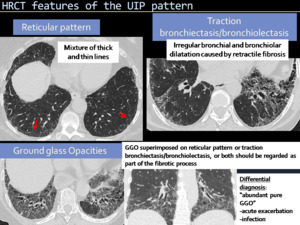
Fig. 4: HRCT features of the UIP pattern.
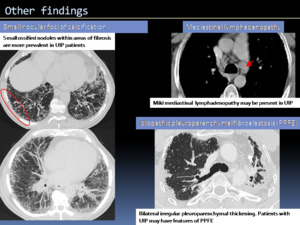
Fig. 5: HRCT features of the UIP pattern.
2.3 HRCT Diagnostic categories of UIP pattern
Regarding the HRCT diagnostic criteria,
both documents incorporate four diagnostic criteria of UIP,
instead of the three previous 2011 categories: 1.
“Usual Interstitial Pneumonia (UIP) pattern”,
2.
“probable UIP pattern”,
3.
“indeterminate for UIP” pattern and 4.
"alternative diagnosis"1/"most consistent with non-IPF diagnosis"2 .
Both documents have removed the previous “Possible usual interstitial pneumonia (UIP)” and two new patterns have been described: "Probable UIP" and "indeterminate for UIP pattern". They included a fourth category with features indicative of an "alternative diagnosis"1/"most consistent with non-IPF diagnosis"2.
UIP pattern
The UIP pattern is the radiographic hallmark of IPF.
A typical UIP pattern is sufficient to ensure a diagnosis of IPF without performing a surgical lung biopsy or other invasive interventions.
The presence of honeycombing is mandatory in the UIP pattern.
The UIP pattern is characterized by honeycombing and reticular opacities,
usually associated with traction bronchiectasis.
GGO superimposed on the other features can be present.
The typical distribution of UIP is subpleural with basal predominance,
although upper lobe involvement is common.
Asymmetric disease may occur in a quarter of the cases.
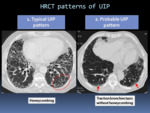
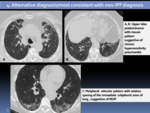
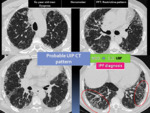
Probable UIP pattern
Subpleural,
basal predominant reticular abnormalities with peripheral traction bronchiectasis or bronchiolectasis but without honeycombing is regarded as a “probable UIP” pattern.
On the basis that some studies have shown that the absence of honeycombing should not exclude a diagnosis of UIP if all other features of UIP are present,
particularly subpleural and basal predominance and traction bronchiectasis,
Fleischner guideline recommend that in a consistent clinical context of IPF,
a lung biopsy is not necessary for the diagnosis of IPF.
While the ATS guideline recommends as a conditional recommendation to perform a surgical lung biopsy in a patient with a probable UIP pattern on CT.
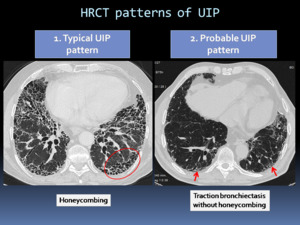
Fig. 6: HRCT patterns of UIP: Typical UIP pattern and Probable UIP pattern.
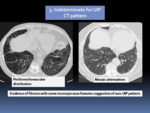
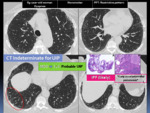
Indeterminate for UIP pattern
This pattern includes subtle subpleural and basilar predominant reticulation with or without mild ground-glass opacities or architectural distortion.
It demonstrates features of fibrosis but without UIP or probable UIP criteria and does not explicitly suggest an alternative diagnosis.
A UIP pattern can still be found on histological testing of patients who do not have typical or probable UIP patterns.
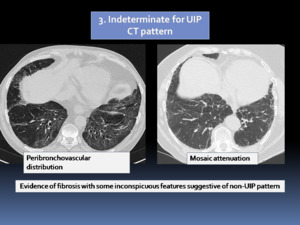
Fig. 7: HRCT patterns of UIP: Indeterminate for UIP CT pattern.
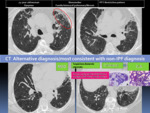
Alternative diagnosis1/Most consistent with non-IPF diagnosis2
In this category,
there are atypical radiographic features suggesting an alternative diagnosis,
including upper lobe bronchocentric fibrosis,
air trapping,
and extensive ground glass opacification with subpleural sparing.
The presence of ancillary findings suggesting an alternative diagnosis,
even in the presence of UIP,
probable UIP,
or indeterminate pattern, should reconsider an alternative diagnosis to IPF.
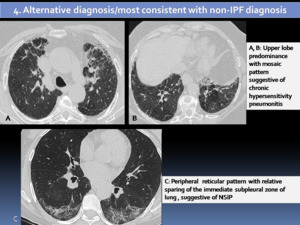
Fig. 8: HRCT patterns of UIP: Alternative diagnosis/most consistent with non-IPF diagnosis.
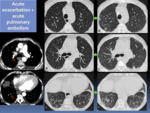
CT findings in an acute exacerbation
CT findings in the presence of an acute exacerbation are described in ATS guideline.
Bilateral GGO with or without consolidation on a background of lung fibrosis,
even in the absence of a previous HRCT study is highly suggestive of acute exacerbation and can be used to confirm an underlying IPF diagnosis in the appropriate clinical context.
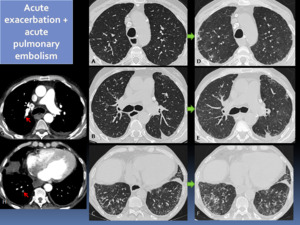
Fig. 9: 75 year-old-man with a probable UIP pattern (A-C) who presented with acute respiratory worsening. HRCT and chest CT angiography were performed showing new bilateral GGOs (D-F) and segmentary acute pulmonary embolism (G-H).
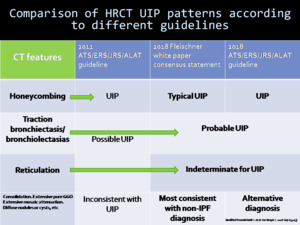
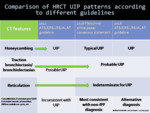
Fig. 10: Comparison of HRCT UIP patterns with the characteristic CT features according to different guidelines.
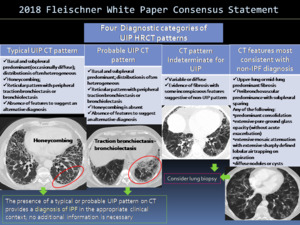
Fig. 11: Four diagnostic categories of UIP based on CT patterns according to 2018 Fleischner White Paper Consensus Statement.

Fig. 12: Four diagnostic categories of UIP based on CT patterns according to 2018 ATS/ERS/JRS/ALAT clinical practice guideline.
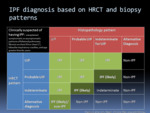
3- Histopathology UIP patterns
Both guidelines recommend categorizing histopathologic findings of biopsies into four categories: “UIP,” “probable UIP,” “indeterminate for UIP,” and “alternative diagnosis”1/ "features most consistent with an alternative diagnosis"2.
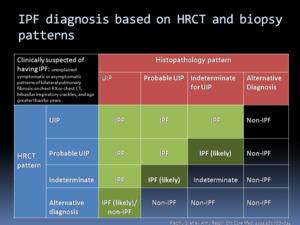
Fig. 13: IPF diagnosis based on HRCT and histopathology patterns.
4- Diagnostic criteria for IPF
A confident diagnosis of IPF can be made in a clinical context of IPF (all: >60 yrs,
absence of clinically significant environmental or medication exposure,
no evidence of connective tissue disease) + CT pattern of typical UIP (also with a probable CT pattern of UIP according to Fleischner guideline),
or with specific combinations of HRCT patterns and histopathology patterns in patients subjected to lung tissue sampling.
According to Fleischner guideline,
the presence of typical or probable UIP pattern on CT provides a diagnosis of IPF in the appropriate clinical context and no additional information is necessary.

Fig. 14: Confident working multidisciplinary diagnosis of IPF according to Fleischner guideline.
5- Diagnostic Interventions
Recommendations for diagnostic interventions intended for patients who are clinically suspected of having IPF without a typical UIP pattern.
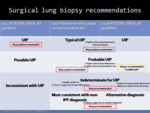
The main difference between the two documents is related to the need for a surgical lung biopsy in patients with a probable UIP pattern on HRCT,
as previously mentioned.
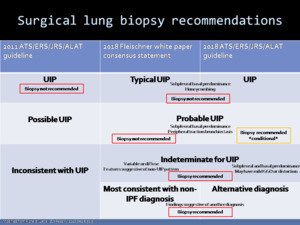
Fig. 15: Comparison of HRCT UIP patterns according to different guidelines and the corresponding recommendations of surgical lung biopsy.
Both documents confirm surgical biopsy as the gold standard for obtaining a tissue sample.
The decision to perform a surgical lung biopsy should be individualized on the basis of risk factors and the patient's decision.
The guidelines provide recommendations about other diagnostic interventions that can be performed instead of surgical biopsy (i.e.
patients who cannot or choose not to undergo a surgical procedure).
They agree that bronchoscopy with transbronchial lung biopsy and bronchoscopic alveolar lavage could provide information to increase the likelihood of an IPF diagnosis or of an alternative diagnosis.
One of the points in which guidelines differ is about the utility of cryobiopsy.
Cryobiopsy had a higher yield than transbronchial lung biopsy alone,
establishing a definitive diagnosis in nearly 80% of cases,
albeit with no direct comparison to samples obtained by surgical lung biopsy.
The ATS/ERS/JRS/ALAT guidelines do not make a recommendation for or against transbronchial cryobiopsy due to the lack of standardization of the procedure but they supported continuing cryobiopsy at experienced centers in patients with probable UIP,
indeterminate,
or an alternative diagnosis HRCT.
On the other hand,
Fleischner Society statement describes transbronchial cryobiopsy as a procedure with an unclear role given the variable levels of clinical experience and postulate that further studies of cryobiopsy and correlation with surgical biopsy and outcome should clarify the role of this technique in the diagnosis of IPF.

Fig. 16: Recommendations for the diagnosis of IPF detailed in the 2018 ATS/ERS/JRS/ALAT guideline.
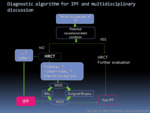
6-Multidisciplinary Discussion
Both documents conclude that multidisciplinary discussion (MDD) among experienced experts are necessary to make an accurate diagnosis of IPF,
particularly for patients with newly detected ILD of apparently unknown cause who are clinically suspected of having IPF.
The MDD consists of an interaction between a pulmonologist (and rheumatologist on a case-by-case basis),
radiologist,
and pathologist.

Fig. 17: MDD: multidisciplinary discussion. Recommendations for MDD conferences according to Fleischner guideline.
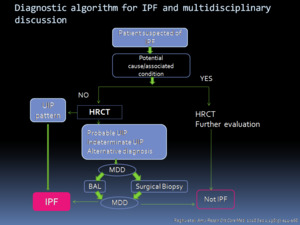
Fig. 18: Diagnostic algorithm for IPF proposed in the 2018 ATS/ERS/JRS/ALAT guideline.
We show the IPF diagnostic algorithm proposed in our hospital with the systematic use of cryobiopsy before performing surgical lung biopsy in selected cases.
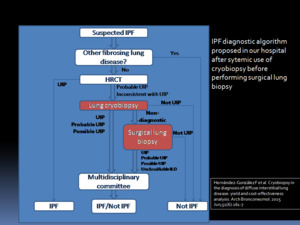
Fig. 19: IPF diagnostic algorithm proposed in our hospital after sytemic use of cryobiopsy before performing surgical lung biopsy.
Clinical cases with histopathological correlation are shown,
demonstrating indications of biopsy.
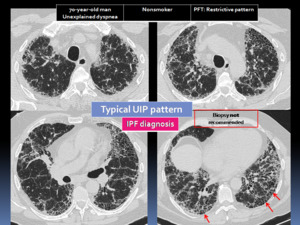
Fig. 20: 70-year-old man with unexplained dyspnea. Axial HRCT images show the presence of honeycombing (red arrows) with basal and subpleural reticular pattern and peripheral traction bronchiectasis. Theses features correspond to a typical UIP pattern, representing an IPF diagnosis. Biopsy is not recommended.
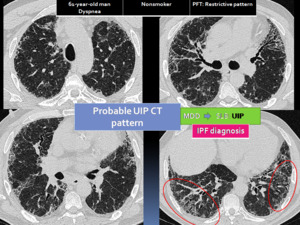
Fig. 21: 61-year-old man with dyspnea. Probable UIP pattern: Axial HRCT images show peripheral traction bronchiectasias and bronchiolectasis (red circles) with a reticular pattern. Honeycombing is not present. A surgical lung biopsy (SLB) was performed and UIP was proven at histology.
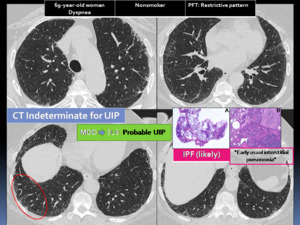
Fig. 22: 69-year-old woman with dyspnea. Indeterminate for UIP CT pattern (reticular opacities, red circle) . A surgical lung biopsy (SLB) showed probable UIP (A: subpleural and paraseptal distribution of fibrosis with architectural distortion; B: higher power evaluation showing patchy fibrosis without honeycomb change. Based on HRCT and biopsy pattern proposed in the 2018 ATS/ERS/JRS/ALAT guideline, the patient was diagnosis with IPF (likely), considered in this case an “Early usual interstitial pneumonia”.
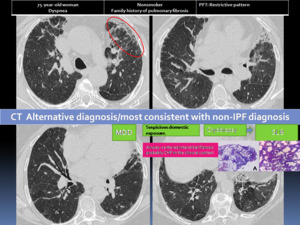
Fig. 23: 75-year-old woman with dyspnea. HRCT with features of an alternative diagnosis: reticular opacities, traction bronchiolectasias in upper lobe predominance (red circle) and mosaic attenuaction pattern. Relevant clinical information suggestive of chronic hypersensitivity pneumonitis (CHP) was highlighted in the MDD: Suspicious domestic exposure; Elevated IgG for Aspergillus, Cladosporium, Penicillium; Bronchioloalveolar lavage: M 62% N 35% L 3%. A cryobiopsy was non-diagnostic. A surgical lung biopsy (SLB) was performed demonstrating A: interstitial fibrosis centered and extending around the bronchioles without non-necrotising granulomas, suggesting a CHP.
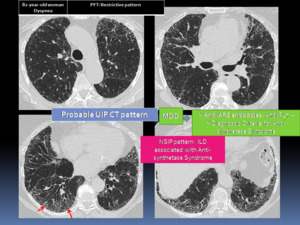
Fig. 24: A 82-year-old woman with dyspnea and a probable CT pattern was presented in a MDD. Additional clinical information found the presence of specific anti-synthetase antibodies (anti-PL7) and diagnostic criteria for Anti-synthetase Syndrome. The HRCT pattern was considered as a Nonspecific interstitial pneumonia (NSIP) related to the autoimmune disease.