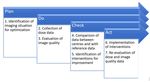
The methodology used for optimization was, in principle, a straightforward process, following a relevant analogue of the PDCA (plan-do-check-act) process, in seven discrete steps, as summarized in Figure 1.
A total of 1445 anonymized patient data were collected, relatively well distributed between examination types studied (radiography: thorax and abdomen, fluoroscopy: MCU, CT: chest and head) and age groups (0-1y, 1y-5y, 5y-10y, 10y-16y), as shown in Figure 2.
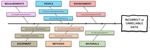
Results demonstrated that in order to effectively carry out the comparison stage, as a pre-requisite to identifying and implementing optimization strategies, it is necessary to have a high degree of confidence in the data obtained. There are, however, several reasons why such confidence may be lacking, in relation to both dosimetric and image quality parameters. Some of the main causes that may lead to unreliable data, as identified during the project are summarized in Figure 3, in the form of an Ishikawa diagram.
The dose results used were collated in a number of different ways across participants, reflecting the spread of methodologies generally available. Increasingly prevalent was the use of electronically recorded parameters, determined by the imaging equipment itself, transferred to the hospital’s patient or imaging data management system, and automatically downloaded into a spreadsheet or by commercial software. At the other end of the spectrum, the usual dosimetry parameters were calculated manually from more easily acquired data relating to individual patient exposures. In between these extremes, electronic dose data was transcribed manually, or semi-automatically, either from a dose measuring device, the imaging equipment itself, or from a data management system.
Electronic collection of data is certainly preferable, as it prevents human transcription errors, although it can be the source of other systematic errors, for instance due to lack of standardisation of dosimetry quantities.
During the data collection process, most issues identified were related to the conventional/planar procedures compared to CT procedures. This was both due to the lack of harmonization of the relevant quantities and units and to the data collection procedure. In certain cases, KAP data and exposure index data were switched round during transfer of data from downloaded spreadsheet to project spreadsheet. In addition, incorrect conversion factors were applied from one KAP unit to another, and incorrect calculation of KAP through using inconsistent units was observed. These issues were able to be resolved once identified externally by reference to the raw data and notification of the centers.
Calibration of all related dose monitoring equipment or electronic display is also a prerequisite for the collection of reliable data. As a minimum, the accuracy of displayed dose parameters should be checked during commissioning and quality assurance of equipment and corrected if outside an acceptable tolerance level.
Adequate diagnostic information is the most important requirement during any imaging procedure and this needs to be emphasized during any optimization process, despite the emphasis given to the dose levels. Image quality evaluation should be an integral part of the optimization process and in certain cases, the optimization process must be driven by the image quality evaluation rather than the dose results. This was especially evident in one institute where MCUs were performed, with KAP values of approximately half of the DRLs, with automatic brightness control compensating for attenuation of the contrasted filled bladder resulting in extremely overexposed images. Low dose levels were due to short fluoroscopy time, appropriately low frame rates and detector dose level, and high added filtration. In this case these images were graded as excellent and no optimization was initiall performed, emphasizing the need for appropriate training for all specialists involved, medical radiology technologists, medical physicists and radiologists.