General presentation of data
An online database was created by the EFSUMB, where different institutions from Europe and across the World can contribute cases where CEUS was performed in paediatric patients. The database is online-based within the website of EFSUMB ( https://www.efsumb.org/blog/) and can be accessed using the following link:
https://www.efsumb.org/blog/) and can be accessed using the following link:  http://www.efsumb-data.org/v1/login.asp. After a brief authorization process, each registered physician can contribute cases and data. Information requested is categorized in different types and includes demographic data (age, gender), indication for CEUS (organ examined, type of pathology, past medical history etc.), technical parameters (type of scanner, dose of contrast agent, single or multiple injections), safety (presence of any side effects), CEUS findings (size of lesion and enhancement pattern) and further details (histologic confirmation or assessment of diagnostic value of CEUS). Ethics committee approval was provided for this study (European Paediatric Contrast-enhanced Ultrasound Database 16/SW/0042 IRAS Project ID 164664 and funding included EFSUMB support for the database and data analysis.
http://www.efsumb-data.org/v1/login.asp. After a brief authorization process, each registered physician can contribute cases and data. Information requested is categorized in different types and includes demographic data (age, gender), indication for CEUS (organ examined, type of pathology, past medical history etc.), technical parameters (type of scanner, dose of contrast agent, single or multiple injections), safety (presence of any side effects), CEUS findings (size of lesion and enhancement pattern) and further details (histologic confirmation or assessment of diagnostic value of CEUS). Ethics committee approval was provided for this study (European Paediatric Contrast-enhanced Ultrasound Database 16/SW/0042 IRAS Project ID 164664 and funding included EFSUMB support for the database and data analysis.
Until early 2019, in total 1151 children were recorded within the online registry. The average age of patients was 11-years with a full spectrum of paediatric age group represented in the dataset, with a range of age from 0 to 17 years of age. An equal distribution between boys (n=598, 52%) and girls (n=554, 48%) was observed.
Safety of paediatric CEUS
No side effect to the agent was reported in 1136 (98.69%) cases, while 13 (1.12%) children complained of mild symptoms. Symptoms recorded included tachycardia, erythema, nausea, vomiting and hypotension. Only one child (0.086%) presented with an anaphylactic reaction managed in intensive care successfully (0.17%). In two cases gas was demonstrated inside the portal vein as an incidental finding without a recognized explanation.
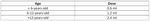
With regards to dose of UCA administered, a range from 0.1 ml to 4.8 ml was documented. In 1049 (91%) cases a single dose of UCA was administered and was sufficient for a diagnostically efficient examination, while in only 103 (9%) of cases a second dose was necessary. The UCA reported to be used in all cases was SonoVue™, also marketed in the USA as Lumason™. According to the manufacturer recommendations, 0.03 ml/kg of body weight should be administered for children, with an upper limit of 2.4 ml for a single dose. This recommendation has been slightly adapted in institutions performing paediatric CEUS examinations, with a widely adopted dosing scheme shown in Table 1. According to the data contributed, the dose physicians chose depended on the child’s age and body weight but variations were also noted depending on equipment’s sensitivity and the organ examined. A full dose of 4.8 mLs (normally 2.4 mLs for the liver in adult patients) of the UCA was used in superficial organ examinations, as the inherent technique requires a 3-4MHz transducer for best results, but less sensitive at the higher frequencies used for superficial structures.
|
Age
|
Dose
|
|
< 6-years-old
|
0.6 ml
|
|
6-12-years-old
|
1.2 ml
|
|
>12-years-old
|
2.4 ml
|
Table 1. A potential dosing scheme for paediatric CEUS
Applications of paediatric CEUS
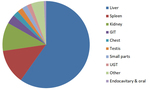
Organs examined included: liver (n=711 cases, 61.8%), spleen (n=153, 13.3%), kidneys (n=126, 10.9%), gastro-intestinal tract (n=41, 3.5%), chest (n=25, 2.1%) and testis (n=24, 2.0%). Less common applications reported included small parts (n=23, 1.9%), urogenital organs like ovaries, pelvic structures and bladder (n=20, 1.6%), adrenal glands (n=18, 1.5%) and other indications not grouped. According to the physician who performed the examination, CEUS was regarded overall useful in 941 applications (81.6%).
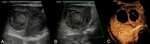
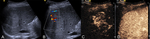
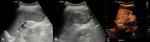
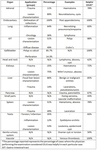
A sub-group analysis of the percentages of different types of applications for every organ examined can be found in Table 2. It is evident that CEUS is helpful in most organs examined, although in some instances this was not the case. For example, CEUS was deemed helpful in only 43% of cases related to small parts and half of cases involving the genito-urinary system (primarily ovaries). Nevertheless, percentages ≥70% have been recorded in other applications, reaching 100% in the case of endocavitary CEUS, vascular pathology and gallbladder applications. Regarding the types of various applications, these can be grouped in certain categories, to better appreciate the indications encountered in various organs. The data contributed showed that CEUS is used for different indications in different organs. For instance in liver applications, CEUS is predominantly used for lesion characterization (86%) and secondly for trauma (14%); an observation possibly attributable to the well-established enhancement patterns and criteria of the various focal liver lesions (Fig. 1, Fig. 2). On the contrary, splenic applications of CEUS are equally divided between lesion characterization and trauma; possibly due to the less well-defined diagnostic criteria available for splenic lesions (Fig. 3). In kidneys, CEUS was mostly used for lesion characterization (75%) and less for trauma (25%); an observation explained by the added value of CEUS in Bosniak classification of renal cysts. A pie-chart showing the ratio of various organs examined with CEUS in children on the EFSUMB Paediatric Registry is shown in Fig. 4.
|
Organ
|
Application (groups)
|
Percentage
|
Examples
|
Helpful CEUS*
|
|
Adrenal
|
-Trauma
-Lesion characterization
|
11%
89%
|
Haematoma
Neuroblastoma, cysts
|
78%
|
|
Endocavitary
|
-Delineation of collections
|
100%
|
Post appendectomy
|
100%
|
|
Lung
|
-Inflammation
-Oncology
|
64%
36%
|
Necrotizing pneumonia/empyema
lymphoma
|
83%
|
|
GIT
|
-Lesion characterization
-Diffuse disease
|
51%
49%
|
Polyp
Crohn’s
|
88%
|
|
Gallbladder
|
-Polyp vs calculi
-Perforation
|
85.7%
14.3%
|
N/A
|
100%
|
|
Head and neck
|
N/A
|
N/A
|
Lymphoma, abscess, cyst
|
62%
|
|
Kidneys
|
-Trauma
-Lesion characterization
|
25%
75%
|
Haematoma
WIlm’s tumour, abscess
|
72%
|
|
Liver
|
-Focal liver lesion characterization
-Trauma
|
86%
14%
|
Benign or malignant entities
Lacerations, pseudoaneurysms
|
85%
|
|
Pancreas
|
N/A
|
N/A
|
trauma, pancreatitis
|
67%
|
|
Small parts
|
N/A
|
N/A
|
Ewing sarcoma, vascular malformation
|
43%
|
|
Spleen
|
-Lesion characterization
-Trauma
|
51%
49%
|
Hamartoma, abscess
Laceration
|
78%
|
|
Testis
|
-Torsion / infarction
-Inflammation
-Lesion characterization
|
35%
12%
53%
|
-
Epididymo-orchitis
Leukemia, epidermoid cyst
|
83%
|
|
Genito-urinary system
|
N/A
|
N/A
|
Ovarian cyst or torsion
|
50%
|
|
Vascular
|
N/A
|
N/A
|
Venous thrombosis
|
100%
|
|
*The percentage reported represents the percentage of cases where the physician performing the examination considered CEUS was helpful in each particular case.
GIT: Gastro-Intestinal tract
|
Table 2. Sub-group analysis of percentages of different application types for organs examined with CEUS in paediatric patients, according to the data contributed to the EFSUMB Paediatric Registry.
Diagnostic accuracy for focal liver lesion characterization
In subgroup analysis for focal liver lesion characterization, CEUS was found 88.5% sensitive and 92.0% specific for diagnosing malignancy. For this type of analysis, only cases with available histologic confirmation of the final diagnosis were analyzed. The index test was the assessment of physician performing CEUS classifying the lesion as benign or malignant and histology was the reference method. Examples of benign diagnoses include hemangioma, FNH, adenoma and focal fatty infiltration or sparing. Malignancies studied include HCC and metastasis.
Limitations of study
Despite the large scale of this project and the plurality of data contributed, this study has some limitations, which need to be taken into consideration before drawing conclusions. These include the retrospective study design, lack of histologic confirmation in many cases, lack of information regarding the inter-observer agreement for evaluation of CEUS findings and the subjective data assessment.
Ethics committee approval (European Paediatric Contrast-enhanced Ultrasound Database 16/SW/0042 IRAS Project ID 164664
Funding: EFSUMB supports the database and data analysis