Congress:
EuroSafe Imaging 2020
Keywords:
Action 4 - Dose management systems, Computer applications, Radioprotection / Radiation dose, PACS, RIS, Computer Applications-General, Dosimetry, Safety, Image registration, Not applicable, Performed at one institution
Authors:
A. Pérez Del Barrio , P. Menéndez Fernández-Miranda, P. Sanz Bellon, E. Marqués Fraguela, E. M. Marco De Lucas, N. Ferreiros Vázquez
DOI:
10.26044/esi2020/ESI-06435
Background/introduction
The objectives of this work are as follows:
- To understand how the study description of an imaging test is transmitted from RIS (Radiology Information System) to DIS (Dosimetric Information System).
- To analyze the problems that arise with the DICOM workflow for this goal.
- To propose a solution to these problems through the use of HL7 messaging and a Business Intelligence (BI) tool such as “QlikView”.
The correct description of imaging tests is essential in a DIS in order to establish diagnostic reference levels (DRLs) and to configure the alert notification system.
In the Cantabrian Health Service, SCS (Servicio Cántabro de Salud), these descriptions are based on the catalog of the Spanish Society of Medical Radiology, SERAM (Sociedad Española de Radiología Médica).
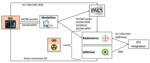
DICOM (Digital Imaging and Communication On Medicine) standard is the usual way of sending the study description from the RIS to the DIS.
The DICOM workflow is as follows:
-
First, study description is transferred from RIS to the modality through the DICOM worklist service.
-
Then, it is sent, either directly from the modality or through the PACS (Picture Archiving and Communication System), to DIS on DICOM headers or on radiation dose structured reports (RDSR).
In the following situations, all of them very usual in routine clinical practice, the DICOM workflow fails to transfer the full RIS description to DIS:
- When a study is started by hand in the modality, without using the DICOM worklist service.
- When the modality does not extract the study description from the DICOM worklist in the proper way (the extracted data depend on the modality manufacturer and configuration).
- When the study description is changed in the RIS after the validation of the order or after having performed the imaging test.