The project is managed via a steering committee with representatives from all healthcare providers involved. The steering committee holds open meetings twice per year, at the Swedish Radiology Conference (Röntgenveckan) and the annual Swedish Medical Physicist Meeting (Nationellt möte om sjukhusfysik). At these open meetings, manufacturer representatives, the Swedish Radiation Safety Authority and medical physics colleagues from other disciplines attend and contribute. The steering committee assigned a project manager to deliver the project and the work was structured into six main work packages (fig. 1), each with its own team and team leader.
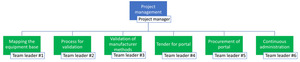
Fig. 1: The structure of the operational management of the project. The project manager divided the project into six well-defined work packages. Work package #1 and #2 were developed and delivered first, as they provided the foundations for work package #3 in terms of providing standardized processes for validation and peer review, as well as reporting templates.
Work package #1 – National inventory of radiological equipment
The outcome of work package #1 was an inventory list of all x-ray equipment that at the time was installed and in use in Sweden, together with a prioritization of which radiological equipment to include for validation in work package #3. The project currently covers 19 manufacturers and 73 medical imaging modalities, which represents the majority of the installed base of radiology equipment in Sweden. The types of modalities include conventional radiography, computed tomography, fluoroscopy labs, mobile c-arms, extra- and intra oral equipment. This work package was completed and delivered to the project management in 2019.
Work package #2 – A quality management system (QMS) for the project
A QMS was devised in work package #2, which consists of a set of standardized processes for validation of manufacturer methods and peer-review of results as well as other core processes to achieve the project aim and goal. This work package was completed and delivered to the project management in 2019. The core process is shown in fig 2.
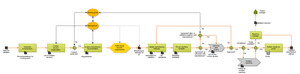
Fig. 2: The core validation process in the project. Green boxes illustrate major activities, black document symbols represent documented routines, recommendations, repositories, lists, reporting templates etc., and grey boxes illustrate sub-processes like peer-review and incident reporting.
Work package #3 – Validation of manufacturer QC methods
Work package #3 is the major work package in the project, comprised of about 25 working groups consisting of medical physicists and engineers distributed throughout Sweden. These working groups were tasked with investigating and understanding how manufacturers are implementing IEC regulations in their quality assurance and control for installation and maintenance of radiology equipment. Once a given equipment had been investigated, properly understood, and quality control tests were documented according to the reporting templates, a second team of medical physicists and engineers performed peer review validation of the outcome for quality management. The validation process is described below in subsequent sections. Work package #3 is presently ongoing.
The validation process
Local validation groups were assembled within each healthcare provider organisation, typically consisting of a team leader, medical physicists and medical engineers. The validation groups were assigned one or several radiology equipment for validation of manufacturer QC methods. The validation groups contacted their respective manufacturers, which in turn assisted with documentation for QC methods used for installation and service as well as engineering expertise within their Swedish and/or Global organizations. In order to share service and QC method documentation, some manufacturers demanded that the healthcare provider should have completed service engineering courses for their own personnel. Once the local validation groups had access to the service and QC documentation, methods were validated according to IEC standards for the respective types of radiology equipment. The findings of the validation process were documented in templates, together with frequency of tests and comments from each validation group, see figure 3 (in Swedish). When validation was completed for an equipment, the documented findings were sent for peer review for quality assurance of the outcome.

Fig. 3: An excerpt of the template produced for validation of a conventional x-ray modality. Testing frequency of relevant IEC standards are described as well as type of test (1 for direct measurement, 2 for indirect measurement), comments on potential weaknesses in the measurement, comments on how the test is performed (manual, automatic, etc.) and lastly the manufacturers own tolerance level (if different from IEC standard).
Peer review of QC method findings
Each validation group sent their documented findings to a quality assessment group, consisting of medical physicists that have the validated radiology equipment in their service. The peer review quality assessment groups had three tasks: (1) determine whether the validation outcome was reasonable, understandable, and that the manufacturer was in agreement with the findings; (2) determine whether the descriptions of manufacturer methods were clearly described in the validation report; and (3) if they agree with the findings of the validation group.
Management of findings that deviate from IEC standards
The TG instated a process for handling deviation from IEC standards and a general lack of quality in manufacturer methods for QC. Included in this process is the reporting of any such findings to the Swedish Radiation Safety Authority and the Swedish Medical Products Agency. No major deviations from IEC standards or general quality deficiencies have been found in the Swedish national quality assurance project for radiological equipment. Minor deviations discovered by the TG has been received well by the manufacturers who look to revise their methods in accordance with the findings of the TG.
Work package #4 and #5 – Tendering and procurement of a web portal
The Swedish national quality assurance project for radiological equipment has a stated goal of delivering a national resource (web portal) for quality management of radiology equipment. This includes financing, tendering and procurement of the technical solution. Work package #4 and #5 are tasked with documenting the requirements of such a portal and to secure funding from the participating healthcare providers. Once a modality has been validated and peer-reviewed, the documentation can be accessed by the members of the project in order to quickly gain an understanding of how the manufacturer conducts QC. This documentation will act as a complement to understanding the manufacturer QC process. Access to the manufacturer’s documentation is still necessary for a complete understanding of the measurements involved. These work packages are presently ongoing.
Work package #6 – Continuous management of the project
For continuous quality management of radiology equipment, evaluation of manufacturer QC methods needs to be undertaken as new equipment is released on the market. Work package #6 develops processes for supplying the community with formal demands in the procurement process with regards to QC for radiology equipment. The continuous management also include yearly follow-up and dialogue with representatives from manufacturers (through the Swedish manufacturer association), suppliers in case of third-party sales, and the healthcare providers that participate in this effort to improve the quality and cost efficiency of handling radiology equipment.