Keywords:
Genital / Reproductive system male, Oncology, Other, Brachytherapy, Outcomes, Toxicity
Authors:
W. L. Ong, J. Millar
DOI:
10.26044/ranzcr2022/R-0140
Methods and materials
Study population: The study comprised men with low to intermediate risk prostate cancer, who had LDR-BT in a single Australia institution. As part of standard of care in our deparment, all men completed the Expanded Prostate Cancer Index Composite short form (EPIC-26) prior to treatment, and at regular intervals post treatment (at 3- and 6-month post-treatment, then 6-monthly for 2 years, and annually thereafter to 10 years). Only men who had EPIC-26 data at baseline and at least three follow-up timepoint (at least 6 months) post LDR-BT were included in this study.
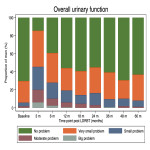
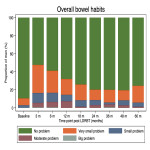
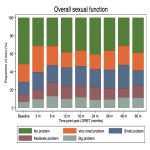
Outcome definition: (1) LAPERS: 'Substantial' symptoms were defined as ‘moderate’ (score 25) to ‘big’ problem (score 0) in EPIC-26 urinary, bowel, and sexual function domain questions. ‘Persistent’ symptoms were defined as ‘substantial’ symptoms that present in at least half of the follow-up EPIC-26 questionnaires. This provides a binary LAPERS outcomes (yes/ no). Baseline adjusted LAPERS takes into account presence of substantial symptoms at baseline. (2) Prevalence at specified time point: Proportion of men who had 'substantial' symptoms at each follow-up time point. (3) Cumulative incidence: Time to the first reported 'substantial' symptoms.
Statistical analyses: Descriptive statistics were used to report proportion of men with (1) LAPERS and baseline-corrected LAPERS, and (2) 'Substantial' symptoms at specified time point. The Kaplan-Meier method was used to estimate (3) cumulative incidence of ‘substantial’ symptoms