Background/introduction
Introduction:
The 33-month European Commission tender project EUCLID (European Study on Clinical Diagnostic Reference Levels for X-ray Medical Imaging) (Figure 1) was launched in August 2017 and submitted its draft final report in January 2020 (with an update, if applicable, due in April 2020). In December 2019 a workshop was held in Luxembourg to discuss the outcomes of the project. This poster will summarize the main results and outcomes of that workshop and the project in general.
Background:
Diagnostic Reference Levels (DRLs) were developed as...
Description of activity and work performed
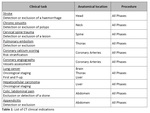
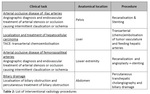
The EUCLID project worked with competent national authorities in 31 different countries to ascertain the current status of DRLs in each country. Subsequently, 20 Hospitals/clinics chosen among the Eurosafe stars network in 14 different European countries contributed data for 10 CT indications and 4 IR indications (see table 1 & 2) with a view to establishing new Clinical DRLs for those procedures. The outcomes were presented and discussed in a workshop, which took place in Luxembourg on 9-10th December 2019. The workshop was attended by...
Conclusion and recommendations
The EU guidance on the establishment and use of DRLs requires updating as it is 20 years old. The EUCLID project has provided a framework for this. Indeed, the workshop discussion revealed that the EUCLID project’s list of clinical indications has already been adopted by some US colleagues. The EUCLID project’s list may furthermore be complemented by a list of 5 clinical indications for CBCT that has been developed in Switzerland. The experience gained during the EUCLID project revealed the importance of data cleaning and...
Personal/organisational information
J. Clark; Vienna/AT - nothing to disclose J. Damilakis; Heraklion/GR - nothing to disclose G. P. Frija; Paris/FR - nothing to disclose