Congress:
EuroSafe Imaging 2020
Keywords:
Not applicable, Outcomes, Dosimetric comparison, Safety, Radiation safety, Nuclear medicine conventional, CT, Conventional radiography, Radioprotection / Radiation dose, Management, Action 2 - Clinical diagnostic reference levels (DRLs)
Authors:
J. Damilakis, G. Frija
DOI:
10.26044/esi2020/ESI-00131
Description of activity and work performed
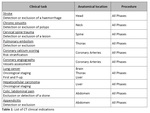
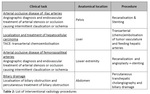
The EUCLID project worked with competent national authorities in 31 different countries to ascertain the current status of DRLs in each country. Subsequently, 20 Hospitals/clinics chosen among the Eurosafe stars network in 14 different European countries contributed data for 10 CT indications and 4 IR indications (see table 1 & 2) with a view to establishing new Clinical DRLs for those procedures. The outcomes were presented and discussed in a workshop, which took place in Luxembourg on 9-10th December 2019. The workshop was attended by experts, including from National Competent Authorities (including the Heads of the European Radiological Protection Competent Authorities (HERCA)), representatives of specialist societies (e.g. the American College of Radiology, European Association of Nuclear Medicine, European Society of Cardiology), international bodies (e.g. World Health Organization, International Atomic Energy Agency, International Commission on Radiological Protection, etc.), and industry experts (e.g. COCIR).
As part of the workshop, all European Union Member States were given the opportunity to present national DRLs for adult patients through their Competent authorities.