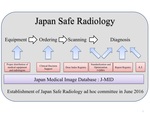
The concept of Japan Safe Radiology is summarized in Figure 1.
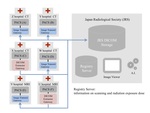
The maximal goal of Japan Safe Radiology is the unified management of medical resources, i.e., the establishment of the J-MID itself, a national database of diagnostic imaging. Collected data can be shared, analyzed and utilized as big data. We have already started to design and develop a small-sized system using data from seven medical institutions. Our goal for the distant future is also to construct an environment in which the image presentation service can be used via the Cloud. The design and development have been performed for a client's gateway system, which can respond to every picture archiving and communication system (PACS) maker. Figure 2 describes the J-MID schematically. Consideration will be given to the integration of this database with other domains such as endoscopic, pathological, ultrasonographic, fundus and cutaneous images. Below, we will report the progress of the construction of the J-MID system.
In December 2017, we established the J-MID server at Kyushu University and then set up a gateway server at each of the seven collaborative medical facilities (university hospitals) to be connected. These facilities were connected by a high-speed and highly secure network called the Science Information NETwork 5 (SINET 5)-virtual private network (VPN).
In March 2018, we started sending CT images and their diagnostic report data to the J-MID server for generating the database. In October 2019, 301,000 CT examinations (76,500,000 images) and 331,000 diagnostic reports have been accumulated in the J-MID. By March 2020, we plan to collect MR images to the J-MID system according to the purpose of multi-institutional joint research.
We describe the progress of the six items described above for each one.
① The optimal distribution of radiologists and medical equipment
The numbers of CT and MRI systems have increased more rapidly in Japan compared to other technologically advanced nations [1], but the number of radiologists in Japan is inadequate. Approximately twice the number of radiologists now employed in Japan is needed to interpret all CT and MRI findings (Figure 3) [2]. Medical equipment is also unevenly distributed in Japan, and the regional gaps in radiological medical practice are large. The radiologist potential workload in Japan was 2.78–4.17 times higher than those in other countries (Figure 4) [3]. A maximum prefecture-to-prefecture variation was 3.88 (Figure 4) [3]. In order to narrow these regional gaps, both medical equipment and radiologists should be distributed across the country more appropriately. To achieve this purpose the data collected in the J-MID may be useful.
② The development of a CDS system for the best use of medical equipment
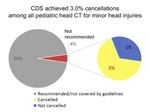
Imaging not recommended by the guidelines is ordered from physicians for modalities and/or body parts, especially for breast cancer-related imaging and for head CT and MRI [4]. Radiologists have to contract physicians who request not recommended examinations for changing in modality and scan range in the intervals of interpretation. A clinical decision support (CDS) system contributes for the optimal use of medical equipment. When the CDS system is online, a diagnostic examination that is suitable for each patient's condition is automatically selected in clinical practice. The guidelines for the choice of a diagnostic examination are also stored in this system. On the occasion of an initial examination order a physician calls up relevant patient data from the server, and a proper examination for the patient is selected automatically. A reduction in the number of orders of duplicated examinations, an improvement in diagnostic accuracy, reduced medical radiation exposure and medical expenses, improved quality of radiologists' reports, and overall advances in the efficiency of clinical practice are expected. We have developed a CDS system for a limited disease set and tested the system at a small number of facilities to verify the feasibility of a CDS in Japan. A pilot study has been started at Juntendo University Hospital for minor head injuries in children. 4% of head CT examinations were not recommended, and 3% of them were cancelled using this CDS system (Figure 5). Another pilot study is performed for lumbar MRI examinations for lumbago at eight facilities.
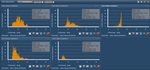
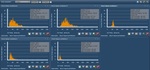
③ The network-type management of radiation exposure doses (DIR)
In addition to imaging data, radiation exposure doses are collected in the J-MID, with the aim of developing a system that performs automatic statistical interpretation; that is, a dose index registry, or DIR. Eventually the optimization of scanning protocols and radiation exposure doses is performed. The exposure doses for chest enhanced CT at five university hospitals in Japan are shown in Figures 6 and 7. As shown in the figure, the exposure dose differs markedly among institutions. The diagnostic reference level (DRL), which is a standard of the upper limit of radiation exposure in a CT examination, is set at the 75th percentile of the dose distribution from a survey conducted across a broad user base in Japan. In cooperation with the Japan Network for Research and Information on Medical Exposures (J-RIME), which is aiming to standardize radiation exposure across large-scale imaging systems, new evidence of DRL could be obtained by the J-MID. At this time, the necessary hardware and software for the DIR have been introduced. The information on weights of patients is missing for a specific vendor though we analyze the data from different CT scanners. Now we try to use Radiation Dose Structure Report (RDSR), instead of a combination of axial images and weights, for analysis of exposure doses. In addition, there is a problem that the names of scanning protocols are different from the actual scanning range for some patients. We aim for the correction of such cases by using AI.
④ The proper quantitation of medical imaging data based on the establishment of the Japan Quantitative Imaging Biomarker Alliance (J-QIBA)
An imaging biomarker is an indicator that can be used to identify a disorder noninvasively and quantitatively. Many researchers have undertaken clinical studies of imaging biomarkers, and various useful biomarkers have been reported globally. However, due to differences in medical equipment, scanning protocols and calculation methods, it is difficult to make direct comparisons of imaging biomarkers obtained at different institutions. In the J-MID project, the standardization of quantitative imaging biomarkers will be attempted based on the imaging information collected in the J-MID. A platform for advancing accuracy and standardizing imaging biomarkers will be structured so that they do not differ among various vendors and equipment. Currently, as members of the Japan Quantitative Imaging Biomarker Alliance (J-QIBA), we are attempting to standardize various imaging biomarkers related to diffusion tensor image; visualization of nerve fiber tract, MR
elastography; evaluation of liver fibrosis, and T1 / T2 value measurement. In the future we will expand this standardization widely. With the standardization of images, we can build a high-quality J-MID that could enable the development of more accurate diagnostic imaging support systems using AI.
⑤ The unification of diagnostic reports
Not only imaging data but also diagnostic reports have been collected, as much as possible, for the J-MID. This will enable us to compare new data with a large dataset of previously obtained images and reports at individual institutions in daily practice. By integration with the database of pathological imaging and reports, we will be able to share rare cases, contributing to the education of radiologists and the improvement of their diagnostic performance. In the future, a part of this database could also be used to fine-tune the JRS registration system and to evaluate medical treatment fees and hospital evaluation systems, for example. We have already constructed a system delivering diagnostic CT reports from a report server at each collaborating facility to the J-MID server after the anonymization of the patient data. We plan to construct the structured reports for the development of a diagnostic imaging support system using AI. Furthermore, we established a system to add a final pathological diagnosis in reports of pre-operative CT when radiologists interpret follow-up CT of the same patient. This system could be useful to the development of AI because of the precise collection of target diseases.
⑥ The clinical applications of artificial intelligence (AI)
Diagnostic imaging support with AI has been improving. In the J-MID project, an automatic diagnosis system based on deep learning is constructed using accurate annotations including the type of diagnostic imaging, the site of disease, the final diagnosis, and other medical data (e.g., findings and diagnostic information). Various new applications or services can be developed using big imaging and reporting data with anonymity. Moreover, this environmental management may enable the training of humans for analytics in the field of diagnostic imaging. Imaging data in the J-MID will be used for the establishment and validation of such support systems as well. Now we address the detection of subarachnoid hemorrhage on unenhanced CT and renal cell carcinoma on the nephrongraphic phase of enhanced CT and collaborate with National Institute of Informatics, which is only the general academic research institution seeking to create future value in the new discipline of informatics in Japan. This collaboration will enhance the improvement of AI systems. For these purpose we established the original annotation system.