Materials and Methods
The CT scanners included in the study are described in Table 1. Data on scanner manufacturer, model, year of manufacture, number of detector rows, use of iterative reconstruction and availability of automatic tube voltage selection are provided. Four Siemens SOMATOM Definition AS+ scanners with automatic tube voltage selection and tube current modulation (TCM), three of them with iterative reconstruction (IR), two Philips Ingenuity and one Philips Brilliance iCT 256 (all with TCM and IR) were included. Patient data were retrieved from the picture archiving and communication system (PACS) of the Trust using the Radimetrics (Bayer) dose management software for a 6-month period from October 2018 until April 2019. Thirteen examinations as required by Public Health England (PHE) for the purpose of the national survey were considered. The list of examinations and clinical indications is shown in Table 2. Protocol settings for the same types of examinations were compared between scanners. Patient data were initially filtered to select the correct examination, protocol name and equipment. DICOM tags Procedure and Description were used to reject other examinations sometimes performed under the same CT protocol. Additional filtering on DICOM tags Series Description, Rotation Time, Slice Thickness, Pitch, and Acquisition Type (axial or helical) was used to select the appropriate phases of the examinations and to reject non-typical ones. Pre-monitoring and monitoring phases were excluded for simplification, as they were estimated to contribute less than 1% of the total patient dose. Total dose-length product (DLP) for multi-phase examinations was calculated as a sum of DLP from separate phases, as the total DLP was not provided by Radimetrics. Descriptive statistical data was calculated for the volume computed tomography dose index (CTDIvol) and the DLP for the patient samples for each examination and on each scanner. These included minimum, maximum, mean, standard deviation, median, 1st and 3rd quartiles, 5th and 95th percentiles of the dosimetry quantities distributions. Since information on patient weight wasn’t available, all dose data (CTDIvol and DLP) below the 5th and above the 95th percentiles were removed and statistical parameters were recalculated, as recommended by ICRP Publication 135. Mean values of the reduced samples were compared with the UK national DRLs (based on means and established during the 2011 review [6]), but median values were chosen as typical doses as considered to be more representative. LDRLs were calculated based on third quartiles of room medians, excluding data from the CT scanner without IR (denoted as CT3 in Table 1). For examinations not performed on all scanners, due to the small number of systems considered, LDRLs were established based on the mean of room medians instead (Neck/chest/abdomen/pelvis (NCAP); Urogram; Virtual Colonoscopy).
Results
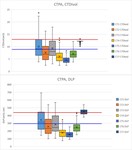
Data was analysed for 24,529 exams. The protocols settings, including tube voltage, TCM settings, the use and setting of IR, total beam collimation, reconstructed primary image slice thickness, tube rotation time, pitch, use of contrast medium, and mean values of CTDIvol and DLP of the reduced samples, are presented in Table 3. The TCM settings of the Siemens scanners are in the order Type of modulation (axial, longitudinal or combined)/Quality Reference mAs (QRM)/Effective mAs/Strength. For Philips scanners the order of the TCM settings is Type of modulation/DoseRight Index (DRI)/mAs for Average Adult/Absolute Max mAs. The Absolute Min mAs is ‘None’ for all Philips protocols. The symbol ‘N’ denotes fixed tube current. Two of the examinations are routinely performed with two acquisitions (phases), namely Virtual Colonoscopy and Urogram. Data are separately presented for each acquisition. The protocols were supposed to be identical on all scanners of the same type (apart CT3 without IR), but several differences were found, mainly due to erroneous data input. The differing settings are bold and underlined in the table. Median values of CTDIvol and DLP of the reduced samples are graphically presented in Figure 1. LDRLs and NDRLs are presented for CTDIvol and DLP in Figure 2. All typical doses were below NDRLs except for Chest – High Resolution DLP on CT1 and Pulmonary Angiography DLP on CT7. The higher TCM settings on both scanners (Table 3) can explain this. Some of the examinations were associated with significant dose differences, with the highest median ratio between scanners 2.88, 2.12, 2.39, 2.90 and 2.86 for Sinuses, Cervical spine, Chest – High Resolution, Pulmonary Angiography and Urogram, respectively. Box plots of CTDIvol and DLP of patient samples for those examinations on all scanners are presented in Figures 3-7, including the LDRL as a blue and the NDRL as a red line, where available. The box is determined by the 1st and the 3rd quartiles, the horizontal line inside the box is the median, the cross is the mean, the whiskers extend up to the smallest/largest data element that is within 1.5 times the interquartile range and the dots represent outlier values outside this range. Urogram is performed on CT1 and CT2 with two different protocols, prone and supine, the supine one usually used for larger patients. For this reason, LDRLs are calculated based on the prone protocol only, although all data are included in the graph (Figure 7). This can also explain the higher doses for the supine protocol in comparison to the prone on the same CT scanners.
Table 1. Parameters of CT scanners.
| System |
Manufacturer |
Model |
Year of manufacture |
Number of detector rows |
Iterative reconstruction |
Automatic tube voltage selection |
| CT1 |
Siemens |
SOMATOM Definition AS+ |
2010 |
64 & flying focal spot |
SAFIRE |
Yes |
| CT2 |
Siemens |
SOMATOM Definition AS+ |
2011 |
64 & flying focal spot |
SAFIRE |
Yes |
| CT3 |
Siemens |
SOMATOM Definition AS+ |
2010 |
64 & flying focal spot |
No |
Yes |
| CT4 |
Siemens |
SOMATOM Definition AS+ |
2010 |
64 & flying focal spot |
SAFIRE |
Yes |
| CT5 |
Philips |
Ingenuity |
2014 |
64 |
iDose |
No |
| CT6 |
Philips |
Ingenuity |
2014 |
64 |
iDose |
No |
| CT7 |
Philips |
Brilliance iCT 256 |
2009 |
128 & smart focal spot |
iDose |
No |
Table 2. List of examinations and relevant clinical indications included in the study.
| PHE CT protocol |
Clinical indication |
| Head |
Acute stroke |
| Paranasal sinuses |
Paranasal sinuses |
| Cervical spine (C-spine) |
Fracture |
| Neck, chest, abdomen and pelvis |
Query Cancer |
| Chest |
Query Lung cancer |
| Chest – high resolution |
Interstitial lung disease |
| Chest and abdomen |
Query Lung cancer |
| Chest-abdomen-pelvis (CAP) |
Query Cancer |
| CT pulmonary angiography (CTPA) |
Pulmonary embolism |
| Abdomen and pelvis |
Abscess |
| Colonography/Virtual colonoscopy (VC) |
Polyps/tumour |
| Kidney-ureters-bladder (KUB) |
Stones/colic |
| Urogram |
Stones/colic or tumour |
Table 3. Protocols settings and mean values of CTDIvol and DLP for reduced patient samples. TCM settings for Siemens scanners are in the order Type of modulation/QRM/Effective mAs/Strength. TCM settings for Philips scanners are in the order Type of modulation/DRI/mAs for Average Adult/Absolute Max mAs.
| System |
Tube voltage (kV) |
TCM |
IR |
Beam collimation (mm) |
Primary image slice thickness (mm) |
Rotation time (s) |
Pitch |
Contrast |
CTDIvol (mGy) |
DLP (mGy cm) |
| Head |
| CT1 |
120 |
Z/290/290/Normal |
SAFIRE 3 |
38.4 |
1 |
1 |
0.55 |
N |
32.1 |
548 |
| CT2 |
120 |
Z/290/290/Normal |
SAFIRE 3 |
38.4 |
1 |
1 |
0.55 |
N |
31.2 |
535 |
| CT3 |
120 |
Z/350/350/Weak |
N |
12 |
3 |
1 |
0.55 |
N |
49.0 |
777 |
| CT4 |
120 |
Z/350/350/Weak |
N |
12 |
1 |
1 |
0.55 |
N |
48.3 |
770 |
| CT5 |
120 |
Z/34/240/250 |
iDose 2 |
40 |
3 |
0.4 |
0.392 |
N |
30.0 |
676 |
| CT6 |
120 |
Z/34/240/None |
iDose 2 |
40 |
3 |
0.4 |
0.392 |
N |
31.1 |
691 |
| CT7 |
120 |
Z/34/240/250 |
iDose 2 |
40 |
3 |
0.4 |
0.39 |
N |
31.5 |
638 |
| Paranasal sinuses |
| CT1 |
120 |
Z/50/50/Normal |
SAFIRE 3 |
38.4 |
1.5 |
1 |
0.8 |
N |
6.0 |
87 |
| CT2 |
120 |
Z/50/50/Normal |
SAFIRE 3 |
38.4 |
1.5 |
1 |
0.8 |
N |
5.7 |
86 |
| CT3 |
120 |
N/ fixed 80 |
N |
38.4 |
1.5 |
1 |
0.8 |
N |
11.6 |
157 |
| CT4 |
120 |
N/fixed 80 |
N |
38.4 |
1.5 |
1 |
0.8 |
N |
11.4 |
154 |
| CT5 |
120 |
N/25 |
iDose 4 |
40 |
1 |
0.4 |
0.399 |
N |
3.2 |
55 |
| CT6 |
120 |
N/25 |
iDose 4 |
40 |
1 |
0.4 |
0.399 |
N |
3.2 |
55 |
| CT7 |
120 |
N/25 |
iDose 4 |
80 |
1 |
0.4 |
0.383 |
N |
3.2 |
53 |
| Cervical spine (C-spine) |
| CT1 |
120 |
4D/275/275/Normal |
N |
38.4 |
1 |
1 |
0.8 |
N |
19.8 |
414 |
| CT2 |
120 |
4D/275/275/Normal |
N |
38.4 |
1 |
1 |
0.8 |
N |
20.1 |
419 |
| CT5 |
120 |
Z/20/127/400 |
iDose 4 |
40 |
1 |
0.5 |
1.4 |
N |
9.6 |
291 |
| CT6 |
120 |
Z/20/127/400 |
iDose 4 |
40 |
1 |
0.5 |
1.4 |
N |
7.4 |
214 |
| CT7 |
120 |
Z/20/127/400 |
iDose 4 |
80 |
1 |
0.4 |
0.925 |
N |
11.0 |
336 |
| Chest |
| CT1 |
Auto |
4D/110/110/Normal |
SAFIRE 3 |
38.4 |
1.5 |
0.5 |
1.2 |
IV |
8.4 |
300 |
| CT2 |
Auto |
4D/110/65/Normal |
SAFIRE 3 |
38.4 |
1.5 |
0.3 |
0.6 |
IV |
9.0 |
317 |
| CT3 |
Auto |
4D/130/110/Weak |
N |
38.4 |
1.5 |
0.5 |
1.2 |
IV |
9.9 |
335 |
| CT4 |
Auto |
4D/170/210/Weak |
SAFIRE 3 |
38.4 |
1.5 |
0.5 |
0.6 |
IV |
9.0 |
283 |
| CT5 |
120 |
Z&3D/16/81/250 |
iDose 3 |
40 |
2 |
0.5 |
0.798 |
IV |
6.0 |
241 |
| CT6 |
120 |
Z&3D/16/81/400 |
iDose 3 |
40 |
2 |
0.5 |
0.798 |
IV |
5.9 |
239 |
| CT7 |
120 |
Z&3D/16/81/400 |
iDose 3 |
80 |
2 |
0.4 |
0.804 |
IV |
6.3 |
266 |
| Chest - high resolution (Chest-HR) |
| CT1 |
120 |
4D/130/110/Normal |
SAFIRE 3 |
38.4 |
1.5 |
0.5 |
1.2 |
N |
11.6 |
404 |
| CT2 |
Auto |
4D/80/65/Normal |
SAFIRE 3 |
38.4 |
1.5 |
0.3 |
0.6 |
N |
7.4 |
261 |
| CT3 |
Auto |
4D/130/110/Weak |
N |
38.4 |
1.5 |
0.5 |
1.2 |
N |
10.1 |
335 |
| CT4 |
Auto |
4D/90/110/Weak |
SAFIRE 3 |
38.4 |
1 |
0.5 |
1.2 |
N |
6.9 |
231 |
| CT5 |
120 |
Z&3D/14/64/90 |
iDose 4 |
40 |
1 |
0.5 |
1.015 |
N |
4.3 |
164 |
| CT6 |
120 |
Z&3D/16/81/90 |
iDose 4 |
40 |
1 |
0.4 |
1.015 |
N |
5.1 |
199 |
| CT7 |
120 |
Z&3D/13/57/90 |
iDose 4 |
40 |
1 |
0.5 |
0.984 |
N |
4.4 |
163 |
| Chest - abdomen - pelvis (CAP) |
| CT1 |
Auto |
4D/150/210/Normal |
SAFRIRE 3 |
38.4 |
1.5 |
0.5 |
0.6 |
IV |
6.3 |
413 |
| CT2 |
Auto |
4D/150/210/Normal |
SAFRIRE 3 |
38.4 |
1.5 |
0.5 |
0.6 |
IV |
6.6 |
433 |
| CT3 |
Auto |
4D/210/210/Weak |
N |
38.4 |
1.5 |
0.5 |
0.6 |
IV |
9.1 |
591 |
| CT4 |
Auto |
4D/170/210/Weak |
SAFRIRE 3 |
38.4 |
1.5 |
0.5 |
0.6 |
IV |
7.4 |
477 |
| CT5 |
120 |
Z&3D/19/113/400 |
iDose 4 |
40 |
2 |
0.5 |
0.798 |
IV |
9.1 |
649 |
| CT6 |
120 |
Z&3D/16/81/400 |
iDose 4 |
40 |
2 |
0.5 |
0.798 |
IV |
6.6 |
470 |
| CT7 |
120 |
Z&3D/16/81/400 |
iDose 4 |
80 |
2 |
0.4 |
0.804 |
IV |
6.7 |
490 |
| CT pulmonary angiography (CTPA) |
| CT1 |
Auto |
4D/110/110/Normal |
SAFIRE 3 |
38.4 |
1 |
0.5 |
1.2 |
IV |
10.0 |
343 |
| CT2 |
Auto |
4D/80/110/Normal |
SAFIRE 3 |
38.4 |
1 |
0.5 |
1.2 |
IV |
7.6 |
258 |
| CT3 |
Auto |
4D/140/110/Weak |
N |
38.4 |
1 |
0.5 |
1.2 |
IV |
9.4 |
285 |
| CT4 |
Auto |
4D/100/110/Weak |
SAFIRE 3 |
38.4 |
1 |
0.5 |
1.2 |
IV |
6.0 |
179 |
| CT5 |
100 |
Z&3D/17/95/190 |
iDose 4 |
40 |
2 |
0.5 |
0.798 |
IV |
4.6 |
156 |
| CT6 |
100 |
Z&3D/17/150/400 |
iDose 4 |
40 |
2 |
0.5 |
0.798 |
IV |
7.1 |
249 |
| CT7 |
100 |
Z&3D/17/237/400 |
iDose 4 |
80 |
1 |
0.33 |
0.763 |
IV |
12.1 |
448 |
| Abdomen and pelvis |
| CT1 |
Auto |
4D/150/210/Normal |
SAFIRE 3 |
38.4 |
1.5 |
0.5 |
0.6 |
IV |
8.2 |
394 |
| CT2 |
Auto |
4D/150/210/Normal |
SAFIRE 3 |
38.4 |
1.5 |
0.5 |
0.6 |
IV |
7.7 |
352 |
| CT3 |
Auto |
4D/240/210/Weak |
N |
38.4 |
2 |
0.5 |
0.8 |
IV |
12.5 |
604 |
| CT4 |
Auto |
4D/170/210/Weak |
SAFIRE 3 |
38.4 |
2 |
0.5 |
0.8 |
IV |
8.7 |
412 |
| CT5 |
120 |
Z&3D/18/101/400 |
iDose 5 |
40 |
2 |
0.5 |
0.89 |
IV |
8.6 |
463 |
| CT6 |
120 |
Z&3D/18/101/300 |
iDose 5 |
40 |
2 |
0.5 |
0.89 |
IV |
8.5 |
447 |
| CT7 |
120 |
Z&3D/17/90/400 |
iDose5 |
80 |
2 |
0.5 |
0.804 |
IV |
7.8 |
435 |
| Kidney - Ureter - Bladder (KUB) |
| CT1 |
Auto |
4D/100/210/Normal |
SAFIRE 3 |
38.8 |
1.5 |
0.5 |
0.6 |
N |
5.6 |
261 |
| CT2 |
Auto |
4D/100/210/Normal |
SAFIRE 3 |
38.8 |
1.5 |
0.5 |
0.6 |
N |
4.5 |
202 |
| CT3 |
Auto |
4D/100/210/Weak |
N |
38.8 |
2 |
0.5 |
0.6 |
N |
6.3 |
265 |
| CT4 |
Auto |
4D/80/210/Weak |
SAFIRE 3 |
38.8 |
2 |
0.5 |
0.6 |
N |
4.4 |
185 |
| CT5 |
120 |
Z&3D/16/81/150 |
iDose 4 |
40 |
2 |
0.4 |
1.49 |
N |
7.1 |
372 |
| CT6 |
120 |
Z&3D/16/81/150 |
iDose 4 |
40 |
2 |
0.4 |
1.49 |
N |
7.0 |
367 |
| CT7 |
120 |
Z&3D/16/81/150 |
iDose 4 |
80 |
2 |
0.5 |
0.804 |
N |
6.9 |
368 |
| Virtual colonoscopy (VC) |
| CT2 Acquis.1 |
Auto |
4D/150/210/Normal |
SAFIRE 3 |
38.4 |
1 |
0.5 |
0.6 |
Case based |
6.9 |
311 |
| CT2 Acquis.2 |
Auto |
4D/30/210/Normal |
SAFIRE 3 |
38.4 |
1 |
0.5 |
1 |
N |
1.4 |
65 |
| CT3 Acquis.1 |
Auto |
4D/230/210/Weak |
N |
38.4 |
1 |
0.5 |
0.6 |
Case based |
10.7 |
507 |
| CT3 Acquis.2 |
Auto |
4D/30/210/Weak |
N |
38.4 |
1 |
0.5 |
0.6 |
N |
1.4 |
63 |
| CT6 Acquis.1 |
120 |
Z&3D/18/101/250 |
iDose 5 |
40 |
2 |
0.5 |
0.89 |
Case based |
9.4 |
486 |
| CT6 Acquis.2 |
120 |
N/40 |
iDose 5 |
40 |
2 |
0.4 |
1.49 |
N |
2.6 |
137 |
| Urogram |
| CT1 Prone Acquis.1 |
Auto |
4D/160/210/Normal |
SAFIRE 3 |
38.4 |
1.5 |
0.5 |
0.6 |
N |
8.7 |
412 |
| CT1 Prone Acquis.2 |
Auto |
4D/150/210/Normal |
SAFIRE 3 |
38.4 |
1.5 |
0.5 |
0.6 |
IV |
6.8 |
322 |
| CT1 Supine Acquis.1 |
Auto |
4D/150/210/Normal |
SAFIRE 3 |
38.4 |
1.5 |
0.5 |
0.6 |
N |
10.6 |
490 |
| CT1 Supine Acquis.2 |
Auto |
4D/150/210/Normal |
SAFIRE 3 |
38.4 |
1.5 |
0.5 |
0.6 |
IV |
10.5 |
508 |
| CT2 Prone Acquis.1 |
Auto |
4D/160/210/Normal |
SAFIRE 3 |
38.4 |
1.5 |
0.5 |
0.6 |
N |
8.7 |
396 |
| CT2 Prone Acquis.2 |
Auto |
4D/160/210/Normal |
SAFIRE 3 |
38.4 |
1.5 |
0.5 |
0.6 |
IV |
7.2 |
331 |
| CT2 Supine Acquis.1 |
Auto |
4D/160/210/Normal |
SAFIRE 3 |
38.4 |
1.5 |
0.5 |
0.6 |
N |
11.3 |
501 |
| CT2 Supine Acquis.2 |
Auto |
4D/160/210/Normal |
SAFIRE 3 |
38.4 |
1.5 |
0.5 |
0.6 |
IV |
10.2 |
481 |
| CT3 Acquis.1 |
120 |
N/160/160/Weak |
N |
38.4 |
2 |
0.5 |
0.6 |
N |
10.8 |
445 |
| CT3 Acquis.2 |
Auto |
4D/230/160/Weak |
N |
38.4 |
2 |
0.5 |
0.6 |
IV |
10.5 |
424 |
| CT4 Acquis.1 |
Auto |
4D/80/210/Weak |
SAFIRE 3 |
38.4 |
2 |
0.5 |
0.6 |
N |
4.3 |
179 |
| CT4 Acquis.2 |
Auto |
4D/80/210/Weak |
SAFIRE 3 |
38.4 |
2 |
0.5 |
0.6 |
IV |
4.4 |
183 |
| CT6 Acquis.1 |
120 |
ACS/18/101/180 |
iDose 5 |
40 |
2 |
0.5 |
0.89 |
N |
10.4 |
533 |
| CT6 Acquis.2 |
120 |
Z&3D/19/113/400 |
iDose 5 |
40 |
2 |
0.5 |
0.89 |
IV |
9.9 |
503 |