Congress:
EuroSafe Imaging 2021
Keywords:
Breast, Cardiovascular system, Radioprotection / Radiation dose, CT, Radiation effects, Radiation safety, Radiobiology, Epidemiology, Workforce
Authors:
G. Frija, E. Cardis, M. Benderitter, R. Coppes, J. Damilakis, G. Flux, I. THIERRY-CHEF, U. MAYERHOFER-SEBERA
DOI:
10.26044/esi2021/ESI-14476
Results or findings
In the following, the key achievements of the MEDIRAD project are presented.
WP2:
- An imaging and radiation dose repository and integrated structured reporting templates to collect clinical information, radiological findings and radiation dose have been developed.
- Α software system is being produced that provides a) image quality information and b) accurate estimation of patient organ doses from chest CT examinations (CTRad).
- Individual voxel phantoms have been developed using high-resolution chest CT image data.
- A real time patient dose monitoring system for interventional procedures has been improved and validated for specific clinical set-up.
- The efficiency and effectiveness of different shielding devices has been assessed.
WP3:
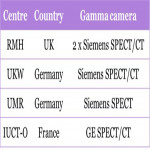
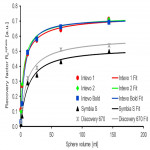
- A European imaging network for quantitative 131I imaging for dosimetry of thyroid cancer patients was set up.
- A Clinical study is open for recruitment at four clinical sites: 92/100 patients have been recruited so far. Dosimetry is performed on patients recruited.
- A radioiodine bio-kinetic model was developed.
- An investigation into the induction and repair of DNA damage in blood cells during radioiodine therapy using blood samples collected as part of the clinical study is underway.
- The initial assessment of the correlation between induction and repair of DNA damage in blood cells and absorbed dose to the blood was performed.
WP4:
- EARLY-HEART, a multi-center prospective cohort study, aims at identifying the most important cardiac imaging and circulating biomarkers of radiation-induced cardiovascular changes arising in the first 2 years after breast cancer radiotherapy and at developing prediction models integrating these biomarkers combined with the dose metrics of cardiac substructures based on 3D-dosimetry. 257 patients were included in the study, and cardiac echo, MRI and CTCA data are currently being analysed.
- BRACE focuses on the development and validation of a multivariable NTCP-model for acute coronary events (ACE). It is a multi-centre retrospective cohort study with two parts: 1. model development on a test cohort: 2. external model validation on the validation cohort. It is the largest cohort study for the development of a multivariable prediction model for ACE. 4,366 patients were analysed. The prediction model based on Left Ventricle-V5, Mean Heart Dose and 7 clinical variables was developed, and optimisation and external validation are currently in progress.
- The preclinical studies to identify mechanism of radiation-induced cardiovascular toxicity aim to investigate the biological mechanisms of heart damage as a function of dose in rodents, for radiation qualities used in radiology and radiotherapy. It was shown that dose and global longitudinal strain correlate, and changes in vascular density are observed with increasing doses. Further analysis is ongoing.
- All irradiations of animals have been performed in the course of the preclinical and clinical identification of standard and innovative biomarkers of radiation-induced cardiovascular toxicity. The first analysis shows no significant correlation with any biomarker, but only with multiple ones. Further analysis is also ongoing.
- A biologically-based model on development of atherosclerosis and subsequent myocardial infarction has been developed.
WP5:
- The follow-up of the Dutch, French, Spanish and UK EPI-CT cohorts by about 5 years is underway with ethics approvals obtained in all countries and data collection on vital status and cancer incidence underway.
- Dosimetric work is underway including 1) a determination of the likelihood the children having received additional CT examinations based on an analysis of data obtained from health insurances with national coverage; and 2) an assessment of doses from CT scanning in recent years based on a sample of PACS data combined with a literature review of the evaluation of CT technology in recent years.
- Case-control study of brain and haematological malignancies nested within EPI-CT cohort: all study documents and approvals are finalised. Recruitment of cases and controls is ongoing in France and Spain. In Sweden, data was collected from medical files, which are currently analysed.
- Biomarkers: tests and validations of kits have been conducted and the strategy for analysis is ready, awaiting receipt of the first samples from the case-control study.
WP6:
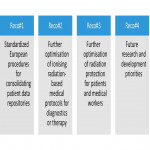
- Based on the results of WP2-5, WP6 is formulating a series of four overarching science-based policy recommendations (“Recos”), for the effective protection of patients, workers and the general public. Together with the Stakeholder Forum, 16 topics have been identified for inclusion in the recommendation documents.
- Two dissemination seminars will be organised at the end of 2021 to present and discuss the recommendation, which will be made publicly available early 2022.