Keywords:
Toxicity, Radiotherapy techniques, Treatment effects, Intensity Modulated Radiotherapy (IMRT), Brachytherapy, CT, Genital / Reproductive system male, Oncology
Authors:
N. Mitina, K. Gogna, A. Gibbs, G. Wood; QLD/AU
DOI:
10.1594/ranzcr2016/R-0048
Results
Only eight patients had a documented recurrent prostate cancer location and Gleason score.
For one patient the recurrence Gleason score was lower than at the time of the initial diagnosis,
in two cases Gleason score was not assigned and in all other cases it was higher than at the initial diagnosis.
That could be due to the natural disease progression or due to sampling uncertainties (all patients initially had mapping biopsies but no dominant nodule sampling).
There was no visible correlation between the initial mapping biopsies’ positive core locations and the recurrence locations.
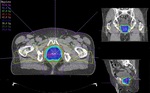
Review of the nine available post-implant dosimetric studies revealed satisfactory target coverage in all patients with mean D90 equalling 130.5Gy.
There was no excessive dose to OARs on post-implant dosimetry.
No correlation was identified between post-implant dosimetric coverage and recurrence location.
Only one patient was treated using 3D conformal technique while 9 patients were treated with IMRT.
The median prostate dose for EBRT was 50.4Gy (range 50.4 – 54Gy) (Table 1).
Nine patients received prostate and seminal vesicles treatment in 2 phases and one had only anterior portion of the prostate treated to 54Gy.
Medial rectal Dmax was 51.04Gy (range 14.11 – 55.60Gy) (Table 1).
EBRT was delivered using daily image guidance with the match to the LDR seeds.
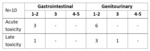
No patients experienced grade 3-5 acute gastrointestinal/genitourinary (GI/GU) toxicities.
Three patients (30%) had grade 1 and one (10%) developed grade 2 acute GI toxicity.
Six patients experienced grade 1-2 acute GU side effects (Table 2).
In regards to the late side effects,
the patient treated with 3D conformal technique was the only one who developed grade 3 GU toxicity (gross haematuria requiring transfusions and cystoscopies).
Four other patients (40%) reported grade 1-2 late GU / GI effects.
There were no grade 4 or 5 toxicities (Table 2).
Six patients (60%) developed biochemical relapse with average biochemical failure free survival of 19 months (median 15,
range 10 – 45 months).
One patient was lost to follow up and other three (30%) remain in remission (14,
41 and 48 months since EBRT completion).
PubMed and Embase systematic literature search returned 375 records.
All records were reviewed and only one case report and one case series published as a poster abstract were identified as relevant to our study [2,
3].
No other published research regarding salvage EBRT after LDR brachytherapy for prostate cancer treatment was found.