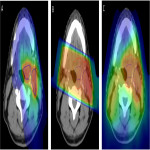
Eight patients diagnosed with head and neck cancer who were referred for radiotherapy at the Central Coast Cancer Centre between 2017 and 2020 were randomly selected for inclusion in this study.
Target volumes and organs at risk (OAR) were independently contoured by a head and neck radiation oncologist. Three radiotherapy treatment plans were generated for each patient: SABR, CPRT, and CD-VMAT.
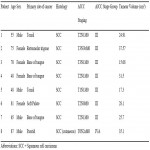
For SABR plans, a dose of 45 Gy in 5 fractions with an overall treatment time of 10 days was prescribed. The gross tumour volume (GTV) was defined based on all available clinical and imaging information. A clinical target volume, 30 Gy (CTV30) was created by expanding the GTV by 5mm, and clipping it at anatomical boundaries. A planning target volume, 45 Gy (PTV45) was created by expanding the GTV isotropically by 3mm. A planning target volume, 30 Gy (PTV30) was created by expanding the CTV30 by 3mm. The target objectives and OAR constraints are listed in Figure 1.
The two dose levels (45 Gy and 30 Gy) were treated using a simultaneous integrated boost (SIB) technique with volumetric-modulated arc therapy (VMAT) with 1-2 coplanar arcs.
For CPRT plans, a dose of 30 Gy in 5 fractions with an overall treatment time of 10 days was prescribed. The GTV was defined the same way as for SABR plans. The CTV30 was also defined the same way as for SABR plans. The PTV30 was created by expanding the CTV30 by 5mm. Simple two- or three-dimensional radiotherapy plans were created, covering as much of the PTV as possible, while excluding the spinal cord to limit its dose to 25.3 Gy.
For CD-VMAT plans, a dose of 70Gy in 35 fractions with an overall treatment time of 47 days was prescribed. The GTV was defined the same way as for SABR plans. The clinical target volume, 70Gy (CTV70) was defined by expanding the GTV by 5mm, and clipping it at anatomical boundaries. The clinical target volume, 56Gy (CTV56) was defined as per consensus guidelines for elective nodal volumes[7,8]. The two dose levels (70 Gy and 56 Gy) were treated using a SIB technique with VMAT with a single coplanar arc. Target objectives and OAR constraints were as per the TROG 12.01 protocol [9].
TCP values were calculated using a modified Poisson model [10]. Parameters used were α/β = 10, α = 0.396, σα = 0.07 and clonogenic cell density = 107 clonogens/cm3. Tumour kinetics were also considered in the calculation with tumour kick-off time (Tk) of 28 days and potential doubling time (Tpot) of 3 days.
Two NTCP models were selected for the biological modelling in this study. NTCPsaliva for patient-rated xerostomia and sticky saliva were calculated using the Beetz logistic regression formula: NTCP = (1 + e-S)-1, where S = -1.443 + (mean dose contralateral parotid gland × 0.047) + (baseline xerostomia score × 0.720) [11]. Baseline xerostomia score was standardised as 0 for all patients. NTCPswallow for swallowing dysfunction was calculated using the Christianen formula: NTCP = (1 + e-S)-1, where S = -6.09 + (mean dose superior pharyngeal constrictor muscle × 0.057) + (mean dose supraglottic larynx × 0.037) [12]. Doses were converted to 2 Gy per fraction using the linear-quadratic model with an α/β ratio of 3 for the parotid gland, superior pharyngeal constrictor muscle and supraglottic larynx.
The Agren model was used to calculate the uncomplicated tumour control probability (UTCP) [13]. This calculation combined the TCP and NTCP values for the two organs using σ = 0.2.
Analysis of variance (ANOVA) with repeated measures was performed on the various radiobiological metrics for the three treatment plans. Statistical significance was taken at P<0.05.