Keywords:
Anatomy, Genital / Reproductive system male, Pelvis, Cone beam CT, CT, Radiation effects, Radiation therapy / Oncology, Image guided radiotherapy, Image verification, Toxicity
Authors:
J. Ward, S. Gill, K. Armstrong, T. Fogarty, A. Scott, D. Tan, A. Yahya, S. S. Dhaliwal, A. Jacques, C. Tang; Perth,, WA/AU
DOI:
10.26044/ranzcr2021/R-0407
Methods and materials
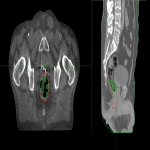
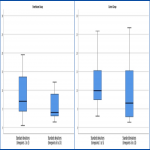
This trial was a prospective, single centre, non-blinded, randomised controlled trial. It included prostate cancer patients being treated with external beam radiation therapy radically to an intact prostate, as well as salvage or adjuvant radiation therapy to the prostate bed, with or without the inclusion of the pelvic lymph nodes. 30 patients were randomised 1:1 to the simethicone or no intervention arm. The simethicone arm had a dose of 100mg three times per day for three days prior to their planning scan, restarted simethicone three days prior to treatment commencement and continued throughout treatment. Cone beam computed tomography (CBCT) scans were performed daily for the first 3 fractions, then weekly until completion. RV and GV were measured using volume delineation on Varian Eclipse on each CBCT (Figure 1). Standard deviations were calculated for each patient for timepoints 1-10. To assess the effect of time on the outcome, these were grouped into timepoints 1-5 and 6-10 for comparison. Toxicity data was collected at baseline and weekly from week 4 using the International Prostate Symptom Score (IPSS), EORTC Quality of Life Questionnaire Prostate Cancer Module (EORTC QLQ PR25) and EORTC QLQ C30 Quality of Life Questionnaire (1,2)