Keywords:
Head and neck, Ultrasound, Biopsy, Pathology
Authors:
G. Hinton, S. Prowse
DOI:
10.26044/ranzcr2022/R-0080
Methods and materials
Approval was obtained from the NALHN clinical governance committee to perform a de-identified quality control audit into thyroid FNA in the radiology department at LMH.
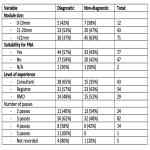
We collected de-identified patient data on all thyroid FNA’s performed at the LMH radiology department over a 7 month period between September 2020 and March 2021. The data collected included: nodule dimensions, maximum diameter, TIRADS classification (if reported), needle gauge (if recorded), number of passes (if recorded), Bethesda category, diagnostic (yes or no), FNA indicated based on TIRADS score and nodule size (yes, no), level of experience of the performing clinician and any subsequent pathology results. The primary aim of this audit was to assess the diagnostic rate of thyroid FNA. Secondary aims of the audit included assessment of potential factors contributing to the diagnostic rate such as procedural factors and suitability for thyroid FNA.
On review of operative histology we can also observe the effectiveness of the TIRADS classification system in predicting the risk of malignancy by comparing the final histology results from FNA to the TIRADs score given on initial diagnostic ultrasound. Unfortunately given the study design we will not be able to tell how many cancers have been missed in this cohort.
Non-diagnostic FNA was defined as Bethesda category 1. Diagnostic FNA was defined as Bethesda categories 2-6.
Assessment of adequate indication for FNA was based on the ACR TIRADS recommendations (Tessler FN et al 2017): this is based on the maximum diameter of the nodule and the TIRADS score reported on the previous diagnostic ultrasound (if available).