Background of the Condition
Cryptococcosis is a systemic mycosis, caused predominantly by Cryptococcus gattii in immunocompetent individuals, acquired from inhalation of spores from pigeon droppings and certain eucalypts[2,3]. Clinical features are variable but predominantly involve a combination of meningitis (67%) [1], papilloedema (15%) [4], and cryptococcomas within the lung (67%) that can cause general respiratory symptoms or be asymptomatic altogether [5]. Cutaneous involvement may be rarely present with different morphologies including ulcers and subcutaneous masses, and may be the first manifestation of disseminated cryptococcosis in endemic areas [6]. Raised intracranial pressure due to mass effect or obstructive hydrocephalus remains a poor prognostic factor, despite symptoms responding well to pressure relief via a drainage shunt [7]. Immune reconstitution inflammatory syndrome (IRIS)-like disease may occur in 7% of patients after starting anti-fungal therapy [8] and is likely due to switching from a Th2 anti-inflammatory response to a Th1 pro-inflammatory response [9]. Imaging, described in detail below, may provide the first indications of disease, noting that cryptococcomas masquerade as neoplastic lesions and indeed up to 42% of patients have a provisional diagnosis of malignancy prior to diagnosis [10]. Definitive diagnosis is made by positive culture and histopathology of CSF and/or fine needle aspirates of a lesion.
Treatment involves initial induction therapy with amphotericin B plus flucytosine for at least 6 weeks followed by consolidation therapy with fluconazole or voriconazole for up to 18 months [12]. Cerebral cryptococcomas are associated with poor response to therapy, and intracranial pressure needs to be closely managed with a shunt as required [12]. Adjuvant glucocorticoid is recommended if there is surrounding oedema and associated neurological deficits, particularly if there is concern of an IRIS-like reaction [13]. Interferon-gamma may be trialled in those unresponsive to prolonged courses of multiple anti-fungals, though its results are mixed [14]. These anti-fungals cause substantial side effects and weekly monitoring of renal function, liver enzymes and serum electrolytes is essential. Even with treatment, mortality may be as high as 20% in Australia [4].
Case
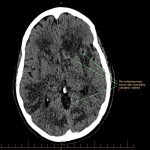
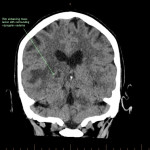
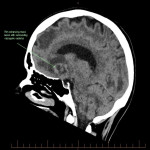
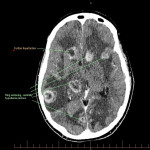
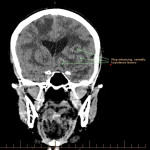
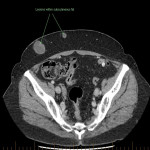
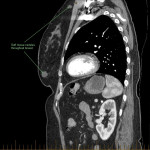
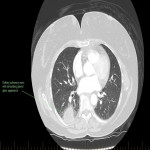
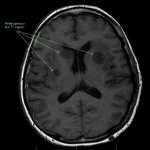
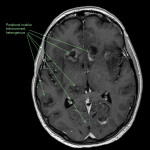
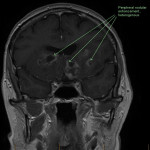
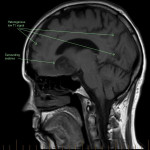
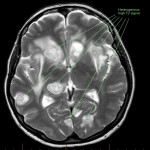
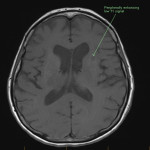
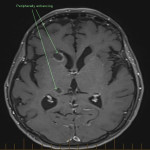
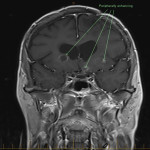
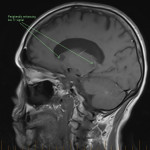
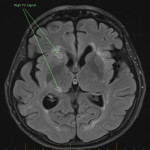
SP, a 63yo immunocompetent female, presented with several weeks of intermittent confusion, night sweats and right visual field defect in her right eye. She had an outpatient CT brain (fig 1-5) with her GP and was referred to the hospital for further workup of metastatic cancer. CT CAP (fig 6-8) showed numerous lesions in the breast, subcutaneous nodules in the abdomen, and a solitary lung lesion with surrounding ground-glass appearance concerning for haemorrhage. US biopsy of the largest abdominal lesion was performed, with pathology favouring C. gattii infection. A lumbar puncture was also performed with CSF growing C. gattii. MRI brain (fig 9-13) showed innumerable cryptococcomas throughout the cerebrum and cerebellum, with particular affinity for the basal ganglia. There was no leptomeningeal enhancement or dilation of the perivascular spaces. Under care of the infectious diseases team, SP was started on amphotericin and flucytosine, but changed to voriconazole due to critical side effects. She required multiple lumbar punctures to relieve intracranial pressure and an external ventricular drain was placed by the neurosurgical team, subsequently removed. SP developed IRIS-like reaction. For the following two years, SP was treated with varying doses of amphotericin, flucytosine, fluconazole, voriconazole, prednisolone, thalidomide and interferon-gamma. She had almost bi-weekly admissions to hospital with critical electrolyte imbalances and renal impairment. However, by May 2022 she had recovered sufficiently to return to work and live independently. Her MRI brain at this time (fig 14-18) showed significant improvement in the size and quantity of the cryptococcomas, though several were still present. The treatment plan for SP was to continue voriconazole and prednisolone life long, however she continued to experience side affects by way of severe weight loss and thinning red skin. Shared decision making was to cease the voriconazole and monitor closely. At the time of this writing, the adverse affects were improving but SP had felt three new subcutaneous nodules and will likely need to restart treatment.