Keywords:
Lung, CT, Audit and standards, Outcomes
Authors:
A. Ajjikuttira, M. Peters, K. Dobeli, P. Sharma, K. Steinke
DOI:
10.26044/ranzcr2023/C-170
Methods and materials
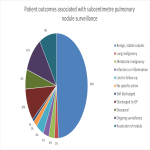
Exemption from full ethics review was obtained. A chronological list of patients was generated through the radiology information system (RIS) used at our facility. Suitable patients were defined as those who had subcentimetre pulmonary nodule(s) incidentally identified after undergoing thoracic-related imaging in the last 4 years. After duplicates were filtered, the unit record number (URN) of the first 100 patients was recorded and their medical records perused. Data parameters of interest included type of imaging performed, Medicare benefit payable per study, dose length product (DLP) of each study and clinical outcome from surveillance. Estimated effective dose was calculated by multiplying DLP by a chest k-factor of 0.014.5