Learning objectives
Review the clinical course and the haemodynamic changes in splenic steal syndrome after liver transplant
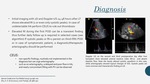
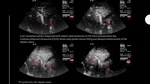
Describe the imaging characteristics of the Doppler ultrasound,
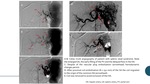
the contrast enhanced ultrasound and the digital subtraction angiography of the disease
Describe the indications,
techniques,
results and potential complications of endovascular treatment
Background
Liver transplantation (LT) is the preferred curative treatment of irreversible chronic and acute end stage liver disease and selected cases of hepatocellular carcinoma.
Although the improvement of pre-transplant evaluation,
patient selection,
transplant procedure and technique extend the survival after LT,
post-transplantation complications may still cause life threatening graft failure.
Potential complications: malignancy,
graft rejection,
biliary lesions and the most important: vascular complications
Venous complications are less frequent and less severe then the arterial ones:
Portal vein thrombosis – incidence 1-2 %
IVC stenosis/thrombosis - 1%...
Findings and procedure details
Postoperative Doppler-US
• After LT a routine Doppler-ultrasound (US) should be performed in the following 24-48 h in order to:
– confirm vascular permeability (HA,
PV,
hepatic veins,
IVC)
–detect flow in the common hepatic artery and main intrahepatic branches
– determine waveform and Resistive Index (RI) (peak systolic velocity– end- diastolic velocity/peak systolic velocity)
– measure portal flow
–rule out other complications
•During the first post-transplantation days elevated RI (>0,80) is relatively common without any pathologic relevance. Doppler US follow-up should be performed daily...
Conclusion
Conclusion
Splenic steal syndrome is an uncommon,
probably under-recognized vascular complication after liver transplant.
Clinical suspicion is raised in case of liver enzyme elevation,
altered arterial resistive index detected on post-transplant follow up Doppler ultrasound with patent hepatic artery and portal permeability in the absence of other clinical or histopathological cause of graft failure.
In such cases,
prompt diagnostic/therapeutic arteriography should be performed of the celiac axis.
Findings of delayed hepatic arteriogram,
large splenic artery diameter,
decreased HA/SA diameter ratio,
splenomegaly could support diagnostic suspicion...
References
GarcĂa-Criado A,
et al.Doppler Ultrasound Findings in the Hepatic Artery Shortly After Liver TransplantationAmerican Journal of Roentgenology2009;193:1,128-135
GarcĂa-Criado A,
Gilabert R,
Nicolau C,
et al.
Early detection of hepatic artery thrombosis after liver transplantation by Doppler ultrasonography.J Ultrasound Med2001; 20:51-58
NĂĽssler N,
Settmacher U,
Haase R,
Stange B,
Heise M,
Neuhaus P.
Diagnosis and treatment of arterial steal syndromes in liver transplant recipients.Liver Transpl2003; 9:596-602
Nishida S,
Kadono J,
De Faria W,
Levi D,
Moon J,
Tzakis A.
Gastroduodenal artery steal syndrome during liver transplantation:...