Background
Definition and Classification
Acute aortic dissection (AAD) is part of a complex similar pathologies defined as Acute Aortic Syndrome, the other two being intra-mural hematoma (IMH) and penetrating atherosclerotic ulcer (PAU).
Aortic dissection is defined as a pathologic process that allows bloods to enter in the media due to an intimal tear or a rupture of tunica media’s vasa vasorum. Subsequently, this process leads to the formation of a true lumen (TL) and a false lumen (FL) that communicate through at least an intimal tear.
Currently, the most used classifications of aortic dissections are Stanford’s and DeBakey’s:
|
Stanford
|
A
|
B
|
|
|
Involves ascending aorta.
|
Involves descending aorta only, commencing after the origin of the left subclavian artery.
|
|
DeBakey
|
I
|
II
|
III
|
|
|
Involves ascending and descending aorta.
|
Involves ascending aorta only.
|
Involves descending aorta only, commencing after the origin of the left subclavian artery.
|
Furthermore, other than Stanford type A and B, there is also another nosological entity, known as “Non-A Non-B dissection: it is defined as an aortic dissection which involves the arch alone or is the result of retrograde propagation of a dissection originating distal to the left subclavian artery’s origin, without extending to the ascending tract. It is a quite rare type of dissection with different outcome and therapeutic management compared to Stanford A and B types of dissection.
Pathophysiology and Epidemiology
AAD is the most common of acute aortic syndromes and it occurs more frequently in male patients who are >60 years old. Female patients are more likely to be affected by AAD at an older age when compared to male patients, often with atypical symptoms.
Several risk factors are associated with this pathology, the most important being long-term hypertension, atherosclerosis, sudden increase of arterious blood pressure (e.g. in cocaine abuse), smoking. AAD could also be associated with connective tissue disorders (e.g. Marfan syndrome, Ehlers-Danlos syndrome) and inflammatory processes (e.g. Horton’s disease, Takayasu Syndrome, syphilis) that could possibly affect aortic wall’s integrity. At last, aortic dissection could be also the consequence of a trauma or a iatrogenic lesion (for example, it could be a complication of cardiac catheterization procedures).
At a molecular level, aortic dissection is the result of aortic wall remodeling due to inflammatory process and extra-cellular matrix degradation: activated macrophages infiltrate the tunica media releasing pro-inflammatory factors and metalloproteinases. The disbalance between the metalloproteinases and their tissue inhibitors leads to a proteolytic process and ultimately to aortic wall remodeling. Another important factor is vascular endothelial growth factor (VEGF), which has a pivotal role concerning neo-angiogenesis, but also a pro-inflammatory action.
Physical factors such as chronic hypertension are very important regarding the pathogenesis of AAD. Hypertension, causing an increased shear stress on the aortic wall acts directly as a stress factor towards the wall itself and indirectly as a pro-inflammatory stimulus, being responsible for macrophage recruitment and activation.
AAD starts with an intimal tear that allows the tunica media to be exposed to pulsatile blood flow. Subsequently, the blood separates the two layers of the wall creating a TL and a FL. The latter could expand further along the vessel’s course or it could even lead to an aortic rupture in case of avventitial disruption, a high mortality complication. The blood could also re-enter in the vessel’s TL through a second intimal tear, hence leading to a variety of symptoms or even to an asymptomatic condition if the patients doesn’t have perfusion impairment. The different flow dynamics between FL and TL could possibly lead to thrombotic phenomena, that obviously occur in the lumen characterized by a slower flow, which is often the FL. Thrombotic phenomena could also not occur if the flow dynamics are equivalent between the two lumens, likely due to the presence of many fenestrations between the two.
Clinical spectrum
Since aortic dissection can ideally involve any tract of the aorta and thus the vessels originating by it, the spectrum of clinical manifestations is quite wide, varying from a complete absence of clinical symptoms to acute life-threatening conditions.
The most common symptom associated with AAD is acute chest pain, which usually has an abrupt onset and a very high intensity, being described as knife-like, sharp or ripping. The pain could also radiate to back and/or abdomen although less frequently (respectively 40% and 25%). Furthermore, patients with AAD could possibly show signs of multiple or single organ failure (the most common being heart failure), neurological symptoms (15-40%) and syncope due to peripheral ischemia secondary to the dissection. Cardiac complications are the most frequent: among them the most important are aortic regurgitation (40-75% of cases, the second cause of death after aortic rupture), myocardial ischemia. Further complications could also occur, depending on the extent of aortic dissection, since an acute occlusion of abdominal vessels originating from the aorta could possibly lead to a variety of clinical conditions (e.g. intestinal ischemia, kidney ischemia, etc.).
Since the symptoms described are not specific for AAD, it is important to rule out other pathologies with similar symptomatology (e.g. IMH, PAU, pulmonary embolism, myocardial infarction, etc.).
Diagnosis
The first step in diagnosis is an accurate clinical examination and blood test for differential diagnosis and complications’ detection. Furthermore, ECG and chest X-ray have a limited value in AAD diagnosis, because the findings are often nonspecific or could even be negative.
Transthoracic echocardiography (TTE) could also prove useful but lacks of sensitivity. At last transoesophageal echocardiography (TOE), Magnetic Resonance Imaging and Computed Tomography Angiography (CTA) have the best diagnostic performances in terms of sensitivity and specificity (almost 95-100%), the latter being the most used in the clinical settings due to availability and scan rapidity.
Typical findings of AAD in CT scans are:
- Evidence of FL and TL lumen (figure 1, 2). The first one is usually smaller and more intensely opacified in early angiographic phases, while the second is often crescent shaped and could contain thrombotic material and residual cobwebs (due to an incomplete separation between the intimal layer and the tunica media). A three-phase scan approach should be applied when an AAD is suspected: a non-enhanced phase that could be useful in the evaluation of the aortic wall (to assess the possible presence of intra-mural haematoma or misplaced intimal calcification), an early angiographic phase to discern the TL from FL (the latter being often less opacified in this phase due to a slower flow), a venous phase to further study the two lumens end verify the presence of thrombotic material in FL.
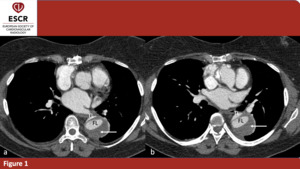
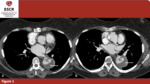
Fig. 1: 46yo F – Patient affected by Stanford A AAD with involvement of thoracic and abdominal aorta. The FL is bigger than the TL and partially thrombosed. (a)(b)axial planes; true (TL) and false (FL) lumina. Thrombotic material (arrow).
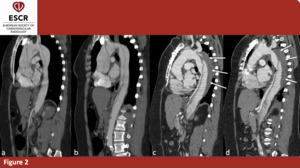
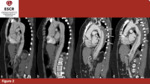
Fig. 2: 46yo F – Baseline and postoperative scans in a patient affected by Stanford A AAD involving thoracic and abdominal aorta. Both the scans were acquired without ECG-gating, thus the intimal flap seems interrupted in many points due to motion artifact. In post operative evaluation, it is not possible to see FL in ascending aorta anymore, but it is still patent in aortic arch, descending and abdominal aorta although partially thrombosed. (a)(b)baseline scan; sagittal planes (c)(d) postoperative scan; sagittal planes; thrombotic material (arrow)
- Evidence of one or more intimal tears, that appear as an interruption of the intimal layer (figure 3). The two most common site of intimal layer rupture are the lateral wall of the ascending aorta and the proximal descending aorta (distal to the origin of left subclavian artery). The regions described are the most affected by increased shear stress, thus being the most frequent locations of entry intimal tear. It could be also possible to see fenestration distal to the entry intimal tear: they allow the blood to re-enter from the FL to the TL. These fenestrations also allow the FL and the vessels originating from it to stay patent.
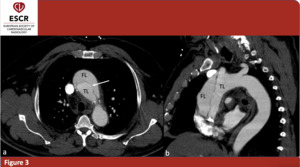
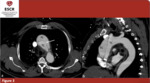
Fig. 3: 62yo M - CTA scans in a patients affected by Stanford A AAD. The images shows involvement of the ascending aorta; aortic arch and descending aorta are not involved. (a) axial plane; intimal tear (arrow) (b) sagittal plane; false (FL) and true lumina (TL).
- Evidence of vessels obstruction and ischemia concerning the organs involved by the dissection (e.g. myocardial ischemia, epi-aortic vessels obstruction, intestinal ischemia, etc.).
Atypical findings of AAD include the presence of more than two lumens, leading to the visualization of multiple channels in the vessel (“Mercedes-Benz sign”, common in dissections secondary to Marfan syndrome) or an incomplete separation between the aortic layers (incomplete tear).
However, the assessment of TL, FL and of and intimal tear could be challenging. Motion artifacts, proximity of great vessels lumens, beam-hardening artifacts caused by severely calcified aortic walls could lead to a misinterpretation of the images.
ECG-gated CTA allows to overcome these limits, providing an accurate representation of the heart and ascending aorta. This technique allows to synchronize the scan to the heartbeat, thus the images are still, clear and of high diagnostic value (figure 4). ECG-gated CTA could be acquired with a retrospective or with a prospective protocol. The first one allows to acquire scans of the aorta during the whole cardiac cycle, administrating a considerable radiation dose to the patient. The second one, on the other hand, is performed only during a desired phase of the cardiac cycle, with a decrease in the radiation dose; mainly, the chosen phase is the left ventricle diastole, because it is the least affected by motion artifacts of the ascending aorta.
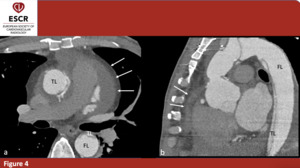
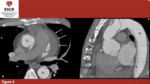
Fig. 4: 40yo M - ECG-gated postoperative evaluation in patient affected by Stanford A AAD with involvement of thoracic and abdominal aorta. The patient underwent prosthetic replacement of the ascending aorta but FL is still patent in the arch (with involvement of epi-aortic vessels) descending and abdominal aorta. It is also possible to see hemopericardium. (a) axial plane; (b) sagittal plane; true (TL) and false (FL) lumina. Hemopericardium (arrow)
Post-surgery imaging features of the aorta may vary depending on the procedure the patient underwent. For example, there are techniques that consist in prosthetic substitution of aorta with or without aortic valve replacement (e.g. Bentall procedure, Yacoub procedure), debranching and re-implant of epi-aortic vessels and even endo-vascular procedures. Therefore, clinical history has a pivotal role concerning a correct evaluation of the post-operative outcome.
Among the most important features that should be evaluated there are possible prosthesis misplacement, aortic valve misplacement and para-valvular leak, patency of the false lumen and thrombosis of the latter.
Therapy
Following the European Society of Cardiology (ESC) guidelines, type A aortic dissections are usually managed surgically, due to the very high rate of major complications and death (50% of mortality in the first 48 hours if not treated). Medical management is reserved for patients of advanced age and/or with significant comorbidity. The adequate surgery performed on the patients is chosen depending of the dissection extent, aortic valve involvement, etc. Nevertheless, the aim of the different surgery techniques is the same and it is to restore the normal flow dynamics of the aorta by closing the entry intimal tear, thus obliterating the FL, or by substituting the aortic segment involved by the dissection with a prosthesis.
Type B aortic dissections, on the other hand, usually respond well to anti-hypertensive therapy which limits or even stops the progression of disease. Surgical management is reserved in case of malperfusion syndrome or evidence of periaortic hematoma.
The treatment of non-A non-B dissections is challenging and debated at length among the experts, being a rare type of dissection. Endo-vascular procedures, conservative approaches have been proposed in certain cases but the main indication is a patient-tailored procedure.
Methods and Materials
We retrospectively evaluated 20 patients who underwent CTA in our Emergency Radiology Department from 2010 to 2018, referring clinical symptoms suggestive for Stanford A AAD, which was later confirmed in the scans. The patients we chose underwent at least one post-surgery CTA after the first scan in the Emergency Department to evaluate the surgical outcome and the development of late complications.
CT scans were acquired with GE Lightspeed Pro 16 slice (GE Healthcare – Milwaukee USA) concerning baseline evaluation at the Emergency Department. Follow-up exams were acquired with Philips iCT 256 Brilliance (Philips – the Nederlands), often with ECG-gated retrospective protocol. Contrast media was administered at an injection rate of 3 to 5 ml/s with a iodine delivery rate >1,6 gI/s, when it was feasible.
The main features of the AAD dissection were evaluated at baseline (coronary arteries’ ostia and aortic valve involvement, epi-aortic vessels involvement, extension of the dissection distal to the arch and possible involvement of abdominal arteries, etc.).
Post-Surgery CTA scans were evaluated to assess the presence of late complications. Among them, the most important features we evaluated were false lumen residual patency or thrombosis (figure 5), new intimal tears, para-valvular leak in cases of combined aortic valve replacement, aortic prosthesis misplacement, hemopericardium or periaortic hematoma (figure 6).
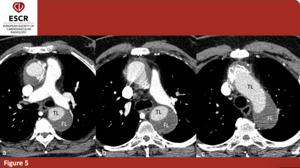
Fig. 5: 64yo M – Succesful surgery outcome in patient affected by Stanford A AAD. FL is not enhanced. (a)(b)(c) axial plane only; true (TL) and false (FL) lumina.
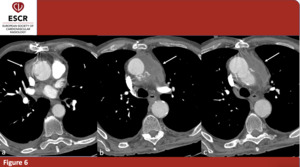
Fig. 6: 78yo M – Postoperative CTA in a patient affected by Stanford A AAD involving the ascending aorta. Although the surgery procedure was successful, it is evident a periaortic hematoma. (a)(b)(c) axial plane only; periaortic hematoma (arrow).